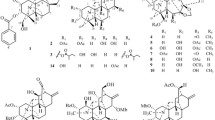
A new triterpene (1), named jaligonic acid B, together with three known triterpenoids, jaligonic acid (2), esculentoside H (3), and esculentoside B (4), was isolated from the roots of Phytolacca acinosa Roxb. The structure of compound 1 was determined on the basis of extensive spectroscopic (IR, MS, 1D and 2D NMR) analyses. All these compounds were evaluated for their cytotoxicity against three human cancer cell lines (BEL-7402, HL-60, HepG2.2.1.5). Compound 4 showed moderate cytotoxic activity. The effects of these compounds on the release of TNF-α from macrophage RAW264.7 cells were also studied. They were also screened for antimicrobial activity against Staphylococcus aureus and Escherichia coli. A triterpene containing a conjugated double bond is reported for the first time in the Phytolaccaceae family.

Similar content being viewed by others
References
M. Z. Getiya, M. A. Gabelaya, V. D. Mshvildadze, A Pichette, S. Lavoie, and G. E. Dekanosidze, Chem. Nat. Compd., 47, 764 (2011).
M. E. Galarraga, A. C. Mitaine-Offer, A. M. Amaro-Luis, T. Miyamoto, L. Pouysegu, S. Quideau, L. B. Rojas, and M. A. Lacaille-Dubois, Nat. Prod. Commun., 5, 775 (2010).
A. Johnson and Y. Shimizu, Tetrahedron, 30, 2033 (1974).
J. He, J. Ma, D. W. Lai, Y. M. Zhang, and W. J. Sun, Nat. Prod. Res., 25, 1771 (2011).
L. Y. Wang, L. M. Bai, T. Nagasawa, T. Hasegawa, X. Y. Yang, J. Sakai, Y. H. Bai, T. Kataoka, S. Oka, K. Hirose, A. Tomida, T. Tsuruo, and M. Ando, J. Nat. Prod., 71, 35 (2008).
B. Wieslawa and M. Irena, Acta. Pol. Pharm., 58, 69 (2001).
X. P. Ma, W. F. Zhang, P. Yi, J. J. Lan, B. **a, S. Jiang, H. Y. Lou, and W. D. Pan, Chem. Biodivers., 14, e1700361 (2017).
H. M. Gao, J. X. Liu, Z. M. Wang, and W. H. Wang, J. Asian Nat. Prod. Res., 11, 433 (2009).
C. L. Wang, H. S. Chen, Q. Y. Zheng, D. H. Qian, and J. P. Zhang, J. Second Mil. Med. Univ., 11, 56 (1990).
D. Zhang, T. Miyase, M. Kuroyanagi, and A. Ueno, Chem. Pharm. Bull., 43, 966 (1995).
W. S. Woo, Phytochemistry, 14, 1885 (1975).
Y. H. Yi and C. L. Wang, Planta Med., 55, 551 (1989).
H. Takahashi, Y. Namikawa, M. Tanaka, and Y. Fukuyama, Chem. Pharm. Bull., 49, 246 (2001).
T. Mosmann, J. Immunol. Methods, 65, 55 (1983).
H. L. Yu, L. Gong, X. Z. Wang, T. F. Zhao, K. L. Wang, X. B. Cui, and L. Chen, Pharm. Biol., 54, 1 (2015).
A. M. Asiri and S. A Khan, Molecules, 15, 6850 (2010).
Acknowledgment
This work was financially supported by the National Natural Science Foundation of China (81660580), the Ministry of Science and Technology of China (2017YFD0201402), the Science and Technology Department of Guizhou Province (QKHRCTD [2015]4026 and QKHRC [2016]4037), the Agricultural Commission of Guizhou Province (GZCYTX2015-0202), and the Science and Technology Department of Bijie City (BKLHZX [2016]06). The authors are grateful to Prof. Jian-**n Zhang and Prof. Dao-** Wang from the Key Laboratory of Chemistry for Natural Products of Guizhou Province and the Chinese Academy of Sciences, respectively, for NMR and mass spectrometric measurements.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 2, March–April, 2019, pp. 251–253.
Rights and permissions
About this article
Cite this article
Ma, XP., Lou, HY., Zhang, WF. et al. Triterpenoids from Phytolacca acinosa. Chem Nat Compd 55, 292–295 (2019). https://doi.org/10.1007/s10600-019-02670-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02670-2