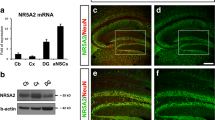
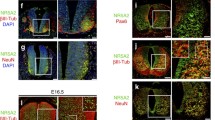
Abstract
Neural stem cells (NSCs) serve as the source of both neurons and support cells, and neurogenesis is reportedly linked to the circadian clock. This study aimed to clarify the functional role of the circadian rhythm-related nuclear receptor, REV-ERBβ, in neurogenesis of NSCs from adult brain. Accordingly, Rev-erbβ expression and the effect of Rev-erbβ gene-specific knockdown on neurogenesis in vitro was examined in adult rodent NSCs. Initial experiments confirmed REV-ERBβ expression in cultured adult NSCs, while subsequent gene expression and gene ontogeny analyses identified functional genes upregulated or downregulated by REV-ERBβ. In particular, expression levels of factors associated with proliferation, stemness, and neural differentiation were affected. Knockdown of Rev-erbβ showed involvement of REV-ERBβ in regulation of cellular proliferation and self-renewal of cultured adult NSCs. Moreover, Rev-erbβ-knockdown cells formed neurons with a slightly shrunken morphology, fewer new primary neurites, and reduced length and branch formation of neurites. Altogether, this suggests that REV-ERBβ is involved in neurite formation during neuronal differentiation of cultured adult NSCs. In summary, REV-ERBβ is a known circadian regulatory protein that appears to be involved in neurogenesis via regulation of networks for cell proliferation and neural differentiation/maturation in adult NSCs.





Similar content being viewed by others
Change history
12 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10571-021-01076-5
References
Aimone JB, Deng W, Gage FH (2011) Resolving new memories: a critical look at the dentate gyrus, adult neurogenesis, and pattern separation. Neuron 70(4):589–596. https://doi.org/10.1016/j.neuron.2011.05.010
Akers KG, Martinez-Canabal A, Restivo L, Yiu AP, De Cristofaro A, Hsiang HL, Wheeler AL, Guskjolen A, Niibori Y, Shoji H, Ohira K, Richards BA, Miyakawa T, Josselyn SA, Frankland PW (2014) Hippocampal neurogenesis regulates forgetting during adulthood and infancy. Science 344(6184):598–602. https://doi.org/10.1126/science.1248903
Akle V, Stankiewicz AJ, Kharchenko V, Yu L, Kharchenko PV, Zhdanova IV (2017) Circadian kinetics of cell cycle progression in adult neurogenic niches of a diurnal vertebrate. J Neurosci 37(7):1900–1909. https://doi.org/10.1523/JNEUROSCI.3222-16.2017
Altman J, Das GD (1965) Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124(3):319–335
Alvarez-Buylla A, Lim DA (2004) For the long run: maintaining germinal niches in the adult brain. Neuron 41(5):683–686
Artigiani S, Conrotto P, Fazzari P, Gilestro GF, Barberis D, Giordano S, Comoglio PM, Tamagnone L (2004) Plexin-B3 is a functional receptor for semaphorin 5A. EMBO Rep 5(7):710–714. https://doi.org/10.1038/sj.embor.7400189
Bancroft J, Auckland P, Samora CP, McAinsh AD (2015) Chromosome congression is promoted by CENP-Q- and CENP-E-dependent pathways. J Cell Sci 128(1):171–184. https://doi.org/10.1242/jcs.163659
Banerjee S, Wang Y, Solt LA, Griffett K, Kazantzis M, Amador A, El-Gendy BM, Huitron-Resendiz S, Roberts AJ, Shin Y, Kamenecka TM, Burris TP (2014) Pharmacological targeting of the mammalian clock regulates sleep architecture and emotional behaviour. Nat Commun 5:5759. https://doi.org/10.1038/ncomms6759
Borgs L, Beukelaers P, Vandenbosch R, Nguyen L, Moonen G, Maquet P, Albrecht U, Belachew S, Malgrange B (2009) Period 2 regulates neural stem/progenitor cell proliferation in the adult hippocampus. BMC Neurosci 10:30. https://doi.org/10.1186/1471-2202-10-30
Bouchard-Cannon P, Mendoza-Viveros L, Yuen A, Kaern M, Cheng HY (2013) The circadian molecular clock regulates adult hippocampal neurogenesis by controlling the timing of cell-cycle entry and exit. Cell Rep 5(4):961–973. https://doi.org/10.1016/j.celrep.2013.10.037
Brandt-Bohne U, Keene DR, White FA, Koch M (2007) MEGF9: a novel transmembrane protein with a strong and developmentally regulated expression in the nervous system. Biochem J 401(2):447–457. https://doi.org/10.1042/BJ20060691
Bugge A, Feng D, Everett LJ, Briggs ER, Mullican SE, Wang F, Jager J, Lazar MA (2012) Rev-erbalpha and Rev-erbbeta coordinately protect the circadian clock and normal metabolic function. Genes Dev 26(7):657–667. https://doi.org/10.1101/gad.186858.112
Burris TP (2008) Nuclear hormone receptors for heme: REV-ERBalpha and REV-ERBbeta are ligand-regulated components of the mammalian clock. Mol Endocrinol 22(7):1509–1520. https://doi.org/10.1210/me.2007-0519
Cho H, Zhao X, Hatori M, Yu RT, Barish GD, Lam MT, Chong LW, DiTacchio L, Atkins AR, Glass CK, Liddle C, Auwerx J, Downes M, Panda S, Evans RM (2012) Regulation of circadian behaviour and metabolism by REV-ERB-alpha and REV-ERB-beta. Nature 485(7396):123–127. https://doi.org/10.1038/nature11048
Chung S, Lee EJ, Yun S, Choe HK, Park SB, Son HJ, Kim KS, Dluzen DE, Lee I, Hwang O, Son GH, Kim K (2014) Impact of circadian nuclear receptor REV-ERBalpha on midbrain dopamine production and mood regulation. Cell 157(4):858–868. https://doi.org/10.1016/j.cell.2014.03.039
Doetsch F, Caille I, Lim DA, Garcia-Verdugo JM, Alvarez-Buylla A (1999) Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97(6):703–716
Duncan MJ, Prochot JR, Cook DH, Tyler Smith J, Franklin KM (2013) Influence of aging on Bmal1 and Per2 expression in extra-SCN oscillators in hamster brain. Brain Res 1491:44–53. https://doi.org/10.1016/j.brainres.2012.11.008
Eriksson PS, Perfilieva E, Bjork-Eriksson T, Alborn AM, Nordborg C, Peterson DA, Gage FH (1998) Neurogenesis in the adult human hippocampus. Nat Med 4(11):1313–1317. https://doi.org/10.1038/3305
Franken P, Dijk DJ (2009) Circadian clock genes and sleep homeostasis. Eur J Neurosci 29(9):1820–1829. https://doi.org/10.1111/j.1460-9568.2009.06723.x
Gage FH (2000) Mammalian neural stem cells. Science 287(5457):1433–1438
Goldberg JL, Vargas ME, Wang JT, Mandemakers W, Oster SF, Sretavan DW, Barres BA (2004) An oligodendrocyte lineage-specific semaphorin, Sema5A, inhibits axon growth by retinal ganglion cells. J Neurosci 24(21):4989–4999. https://doi.org/10.1523/JNEUROSCI.4390-03.2004
Gunnersen JM, Kim MH, Fuller SJ, De Silva M, Britto JM, Hammond VE, Davies PJ, Petrou S, Faber ES, Sah P, Tan SS (2007) Sez-6 proteins affect dendritic arborization patterns and excitability of cortical pyramidal neurons. Neuron 56(4):621–639. https://doi.org/10.1016/j.neuron.2007.09.018
Hwang HC, Clurman BE (2005) Cyclin E in normal and neoplastic cell cycles. Oncogene 24(17):2776–2786. https://doi.org/10.1038/sj.onc.1208613
Imayoshi I, Isomura A, Harima Y, Kawaguchi K, Kori H, Miyachi H, Fujiwara T, Ishidate F, Kageyama R (2013) Oscillatory control of factors determining multipotency and fate in mouse neural progenitors. Science 342(6163):1203–1208. https://doi.org/10.1126/science.1242366
Jagannath A, Peirson SN, Foster RG (2013) Sleep and circadian rhythm disruption in neuropsychiatric illness. Curr Opin Neurobiol 23(5):888–894. https://doi.org/10.1016/j.conb.2013.03.008
Jiang Y, Hsieh J (2014) HDAC3 controls gap 2/mitosis progression in adult neural stem/progenitor cells by regulating CDK1 levels. Proc Natl Acad Sci USA 111(37):13541–13546. https://doi.org/10.1073/pnas.1411939111
Johnston ST, Shtrahman M, Parylak S, Goncalves JT, Gage FH (2016) Paradox of pattern separation and adult neurogenesis: A dual role for new neurons balancing memory resolution and robustness. Neurobiol Learn Mem 129:60–68. https://doi.org/10.1016/j.nlm.2015.10.013
Kempermann G, Jessberger S, Steiner B, Kronenberg G (2004) Milestones of neuronal development in the adult hippocampus. Trends Neurosci 27(8):447–452. https://doi.org/10.1016/j.tins.2004.05.013
Khapre RV, Kondratova AA, Patel S, Dubrovsky Y, Wrobel M, Antoch MP, Kondratov RV (2014) BMAL1-dependent regulation of the mTOR signaling pathway delays aging. Aging (Albany NY) 6(1):48–57. https://doi.org/10.18632/aging.100633
Kimiwada T, Sakurai M, Ohashi H, Aoki S, Tominaga T, Wada K (2009) Clock genes regulate neurogenic transcription factors, including NeuroD1, and the neuronal differentiation of adult neural stem/progenitor cells. Neurochem Int 54(5–6):277–285. https://doi.org/10.1016/j.neuint.2008.12.005
Ko CH, Takahashi JS (2006) Molecular components of the mammalian circadian clock. Hum Mol Genet 15(Spec No 2):R271–277. https://doi.org/10.1093/hmg/ddl207
Kojetin DJ, Burris TP (2014) REV-ERB and ROR nuclear receptors as drug targets. Nat Rev Drug Discov 13(3):197–216. https://doi.org/10.1038/nrd4100
Kondratov RV, Kondratova AA, Gorbacheva VY, Vykhovanets OV, Antoch MP (2006) Early aging and age-related pathologies in mice deficient in BMAL1, the core componentof the circadian clock. Genes Dev 20(14):1868–1873. https://doi.org/10.1101/gad.1432206
Kondratova AA, Dubrovsky YV, Antoch MP, Kondratov RV (2010) Circadian clock proteins control adaptation to novel environment and memory formation. Aging (Albany NY) 2(5):285–297. https://doi.org/10.18632/aging.100142
Lacomme M, Liaubet L, Pituello F, Bel-Vialar S (2012) NEUROG2 drives cell cycle exit of neuronal precursors by specifically repressing a subset of cyclins acting at the G1 and S phases of the cell cycle. Mol Cell Biol 32(13):2596–2607. https://doi.org/10.1128/MCB.06745-11
Lee HJ, Son GH, Geum D (2013) Circadian rhythm hypotheses of mixed features, antidepressant treatment resistance, and manic switching in bipolar disorder. Psychiatry Investig 10(3):225–232. https://doi.org/10.4306/pi.2013.10.3.225
Louis SA, Rietze RL, Deleyrolle L, Wagey RE, Thomas TE, Eaves AC, Reynolds BA (2008) Enumeration of neural stem and progenitor cells in the neural colony-forming cell assay. Stem Cells 26(4):988–996. https://doi.org/10.1634/stemcells.2007-0867
Malik A, Kondratov RV, Jamasbi RJ, Geusz ME (2015) Circadian clock genes are essential for normal adult neurogenesis, differentiation, and fate determination. PLoS ONE 10(10):e0139655. https://doi.org/10.1371/journal.pone.0139655
McClung CA (2007) Circadian genes, rhythms and the biology of mood disorders. Pharmacol Ther 114(2):222–232. https://doi.org/10.1016/j.pharmthera.2007.02.003
McClung CA (2013) How might circadian rhythms control mood? Let me count the ways. Biol Psychiatry 74(4):242–249. https://doi.org/10.1016/j.biopsych.2013.02.019
Ming GL, Song H (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70(4):687–702. https://doi.org/10.1016/j.neuron.2011.05.001
Mira H, Andreu Z, Suh H, Lie DC, Jessberger S, Consiglio A, San Emeterio J, Hortiguela R, Marques-Torrejon MA, Nakashima K, Colak D, Gotz M, Farinas I, Gage FH (2010) Signaling through BMPR-IA regulates quiescence and long-term activity of neural stem cells in the adult hippocampus. Cell Stem Cell 7(1):78–89. https://doi.org/10.1016/j.stem.2010.04.016
Molinari F, Rio M, Meskenaite V, Encha-Razavi F, Auge J, Bacq D, Briault S, Vekemans M, Munnich A, Attie-Bitach T, Sonderegger P, Colleaux L (2002) Truncating neurotrypsin mutation in autosomal recessive nonsyndromic mental retardation. Science 298(5599):1779–1781. https://doi.org/10.1126/science.1076521
Musiek ES, Lim MM, Yang G, Bauer AQ, Qi L, Lee Y, Roh JH, Ortiz-Gonzalez X, Dearborn JT, Culver JP, Herzog ED, Hogenesch JB, Wozniak DF, Dikranian K, Giasson BI, Weaver DR, Holtzman DM, Fitzgerald GA (2013) Circadian clock proteins regulate neuronal redox homeostasis and neurodegeneration. J Clin Invest 123(12):5389–5400. https://doi.org/10.1172/JCI70317
Nigg EA, Gallant P, Krek W (1992) Regulation of p34cdc2 protein kinase activity by phosphorylation and cyclin binding. Ciba Found Symp 170:72–84 discussion 84–96
Pagano M, Pepperkok R, Verde F, Ansorge W, Draetta G (1992) Cyclin A is required at two points in the human cell cycle. EMBO J 11(3):961–971
Porlan E, Morante-Redolat JM, Marques-Torrejon MA, Andreu-Agullo C, Carneiro C, Gomez-Ibarlucea E, Soto A, Vidal A, Ferron SR, Farinas I (2013) Transcriptional repression of Bmp2 by p21(Waf1/Cip1) links quiescence to neural stem cell maintenance. Nat Neurosci 16(11):1567–1575. https://doi.org/10.1038/nn.3545
Rakai BD, Chrusch MJ, Spanswick SC, Dyck RH, Antle MC (2014) Survival of adult generated hippocampal neurons is altered in circadian arrhythmic mice. PLoS ONE 9(6):e99527. https://doi.org/10.1371/journal.pone.0099527
Ray J, Gage FH (2006) Differential properties of adult rat and mouse brain-derived neural stem/progenitor cells. Mol Cell Neurosci 31(3):560–573. https://doi.org/10.1016/j.mcn.2005.11.010
Schnell A, Chappuis S, Schmutz I, Brai E, Ripperger JA, Schaad O, Welzl H, Descombes P, Alberi L, Albrecht U (2014) The nuclear receptor REV-ERBalpha regulates Fabp7 and modulates adult hippocampal neurogenesis. PLoS ONE 9(6):e99883. https://doi.org/10.1371/journal.pone.0099883
Schuch JB, Genro JP, Bastos CR, Ghisleni G, Tovo-Rodrigues L (2017) The role of CLOCK gene in psychiatric disorders: evidence from human and animal research. Am J Med Genet B. https://doi.org/10.1002/ajmg.b.32599
Shi Y, Chichung Lie D, Taupin P, Nakashima K, Ray J, Yu RT, Gage FH, Evans RM (2004) Expression and function of orphan nuclear receptor TLX in adult neural stem cells. Nature 427(6969):78–83. https://doi.org/10.1038/nature02211
Shimozaki K (2017) Ten-eleven translocation 1 and 2 confer overlap** transcriptional programs for the proliferation of cultured adult neural stem cells. Cell Mol Neurobiol 37(6):995–1008. https://doi.org/10.1007/s10571-016-0432-6
Shimozaki K, Zhang CL, Suh H, Denli AM, Evans RM, Gage FH (2012) SRY-box-containing gene 2 regulation of nuclear receptor tailless (Tlx) transcription in adult neural stem cells. J Biol Chem 287(8):5969–5978. https://doi.org/10.1074/jbc.M111.290403
Shimozaki K, Clemenson GD Jr, Gage FH (2013) Paired related homeobox protein 1 is a regulator of stemness in adult neural stem/progenitor cells. J Neurosci 33(9):4066–4075. https://doi.org/10.1523/JNEUROSCI.4586-12.2013
Shors TJ, Miesegaes G, Beylin A, Zhao M, Rydel T, Gould E (2001) Neurogenesis in the adult is involved in the formation of trace memories. Nature 410(6826):372–376. https://doi.org/10.1038/35066584
Solt LA, Wang Y, Banerjee S, Hughes T, Kojetin DJ, Lundasen T, Shin Y, Liu J, Cameron MD, Noel R, Yoo SH, Takahashi JS, Butler AA, Kamenecka TM, Burris TP (2012) Regulation of circadian behaviour and metabolism by synthetic REV-ERB agonists. Nature 485(7396):62–68. https://doi.org/10.1038/nature11030
Song J, Zhong C, Bonaguidi MA, Sun GJ, Hsu D, Gu Y, Meletis K, Huang ZJ, Ge S, Enikolopov G, Deisseroth K, Luscher B, Christian KM, Ming GL, Song H (2012) Neuronal circuitry mechanism regulating adult quiescent neural stem-cell fate decision. Nature 489(7414):150–154. https://doi.org/10.1038/nature11306
Tamai S, Sanada K, Fukada Y (2008) Time-of-day-dependent enhancement of adult neurogenesis in the hippocampus. PLoS ONE 3(12):e3835. https://doi.org/10.1371/journal.pone.0003835
Yan J, Kuroyanagi H, Tomemori T, Okazaki N, Asato K, Matsuda Y, Suzuki Y, Ohshima Y, Mitani S, Masuho Y, Shirasawa T, Muramatsu M (1999) Mouse ULK2, a novel member of the UNC-51-like protein kinases: unique features of functional domains. Oncogene 18(43):5850–5859. https://doi.org/10.1038/sj.onc.1202988
Yu ZL, Jiang JM, Wu DH, **e HJ, Jiang JJ, Zhou L, Peng L, Bao GS (2007) Febrile seizures are associated with mutation of seizure-related (SEZ) 6, a brain-specific gene. J Neurosci Res 85(1):166–172. https://doi.org/10.1002/jnr.21103
Zenker J, Stettner M, Ruskamo S, Domenech-Estevez E, Baloui H, Medard JJ, Verheijen MH, Brouwers JF, Kursula P, Kieseier BC, Chrast R (2014) A role of peripheral myelin protein 2 in lipid homeostasis of myelinating Schwann cells. Glia 62(9):1502–1512. https://doi.org/10.1002/glia.22696
Zhang CL, Zou Y, He W, Gage FH, Evans RM (2008) A role for adult TLX-positive neural stem cells in learning and behaviour. Nature 451(7181):1004–1007. https://doi.org/10.1038/nature06562
Zhang RR, Cui QY, Murai K, Lim YC, Smith ZD, ** S, Ye P, Rosa L, Lee YK, Wu HP, Liu W, Xu ZM, Yang L, Ding YQ, Tang F, Meissner A, Ding C, Shi Y, Xu GL (2013) Tet1 regulates adult hippocampal neurogenesis and cognition. Cell Stem Cell 13(2):237–245. https://doi.org/10.1016/j.stem.2013.05.006
Zhao C, Teng EM, Summers RG Jr, Ming GL, Gage FH (2006) Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J Neurosci 26(1):3–11. https://doi.org/10.1523/JNEUROSCI.3648-05.2006
Zhao C, Deng W, Gage FH (2008) Mechanisms and functional implications of adult neurogenesis. Cell 132(4):645–660. https://doi.org/10.1016/j.cell.2008.01.033
Zhou X, Babu JR, da Silva S, Shu Q, Graef IA, Oliver T, Tomoda T, Tani T, Wooten MW, Wang F (2007) Unc-51-like kinase 1/2-mediated endocytic processes regulate filopodia extension and branching of sensory axons. Proc Natl Acad Sci USA 104(14):5842–5847. https://doi.org/10.1073/pnas.0701402104
Acknowledgements
I thank K. Akagi and the members of the Life Science Support Center at Nagasaki University for technical assistance and helpful discussions. Editage by Cactus and Edanz Group Japan are acknowledged for their assistance with preparation of the manuscript. I also thank Alison Sherwin, Ph.D., from Edanz Group (www.edanzediting.com/ac) for editing a draft of the manuscript. This work was supported by a Grant-in-Aid for Scientific Research (C) (Grant Number 15K06711) from the Japan Society for the Promotion of Science (JSPS) and a Grant-in-Aid from the Alumni Association of Nagasaki University School of Medicine.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised: the keyword "REV-ERBβ" has been corrected as "REV-ERB".
Electronic supplementary material
Below is the link to the electronic supplementary material.
10571_2018_576_MOESM1_ESM.pdf
Supplementary material 1 (PDF 1184 kb) Supplementary Material 1 Quantification of neural differentiation efficiency of mouse and rat cultured adult neural stem cells (NSCs) after gene-specific knockdown. Percentage of GFP and highly expressed GFAP (GFAPhigh) or βIII-tubulin double-positive cells in mouse (a) or rat (b) adult NSCs after neural differentiation was measured in three random microscopy fields (control [CTRL]: white bar; Rev-erbβ knockdown [Revβ-KD]: black bar). Error bars represent ± SEM (n = 4). P = 0.2582, P = 0.9856, P > 0.9999, P = 0.8715, Sidak’s multiple comparison test after two-way ANOVA
10571_2018_576_MOESM2_ESM.pdf
Supplementary material 2 (PDF 1158 kb) Supplementary Material 2 Expression of representative neural stem cell (NSC) marker genes after Rev-erbβ gene knockdown in cultured adult NSCs. Total RNA from NSCs was analyzed by qRT-PCR after Rev-erbβ knockdown and puromycin selection (control [CTRL]: white bars; Rev-erbβ knockdown [Revβ-KD]: black bars). Data represent mean ± SD (n = 4). *P < 0.05, **P < 0.01, ****P ≤ 0.0001, two-tailed unpaired t test. N.D.: (not detected)
Rights and permissions
About this article
Cite this article
Shimozaki, K. Involvement of Nuclear Receptor REV-ERBβ in Formation of Neurites and Proliferation of Cultured Adult Neural Stem Cells. Cell Mol Neurobiol 38, 1051–1065 (2018). https://doi.org/10.1007/s10571-018-0576-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-018-0576-7