Abstract
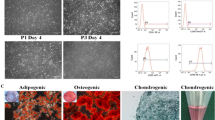
Mesenchymal Stem Cells (MSCs) are important in regenerative medicine and tissue engineering and will be a very sensible choice for repair and regeneration of tendon. New biological practices, such as cellular therapy using stem cells, are promising for facilitating or expediting tendon therapy. Before using these cells clinically, it is best to check and confirm the optimal conditions for differentiation of these cells in the laboratory. Hence, in the present study, the impacts of PDGF-BB and GDF-6 supplementation on adipose-derived MSCs (ASCs) culture were studied. The frozen ASC were recovered and expanded in basic culture medium (DMEM with 10%FBS). The cells after passage five (P5) were treated with basic medium containing L-Prolin, Ascorbic Acid and only PDGF-BB or GDF-6 (20 ng/ml) or both of them (mix) as 3 groups for 14 days to investigate efficiency of ASCs differentiation towards tenocytes. The cells culturing in basic medium were used as control group. To validate tenogenic differentiation, H&E and Sirius Red staining were used to assess cell morphology and collagen production, respectively. In addition, mRNA levels of collagen I and III, Scleraxis and Tenomodulin as tenogenic markers were analyzed using qPCR. In all test groups, cells appeared slenderer, elongated cytoplasmic attributes compared to the control cells. The intensity of Sirius Red staining was significantly higher in GDF-6, PDGF-BB alone, than in group without supplements. The optical density was higher in the GDF-6 than PDGF-BB and mix-group. QPCR results showed that Col I and III gene expression was increased in all groups compared to the control. SCX expression was significantly increased only in the PDGF-BB group. TNMD mRNA expression was not significant among groups. In this study, we have corroborated that human ASCs are reactionary to tenogenic induction by GDF-6 and PDGF-BB alone or in combination. These outcomes will help greater insight into GDF-6 and PDGF-BB driven tenogenesis of ASCs and new directions of discovery in the design of ASC-based treatments for tendon healing.



Similar content being viewed by others
Data availability
The data are available from the corresponding author upon reasonable request.
References
Anitua E, Sanchez M, De la Fuente M et al (2012) Plasma rich in growth factors (PRGF-Endoret) stimulates tendon and synovial fibroblasts migration and improves the biological properties of hyaluronic acid. Knee Surg Sport Traumatol Arthrosc 20:1657–1665. https://doi.org/10.1007/s00167-011-1697-4
Bernard-Beaubois K, Hecquet C, Houcine O et al (1997) Culture and characterization of juvenile rabbit tenocytes. Cell Biol Toxicol 13:103–113. https://doi.org/10.1023/B:CBTO.0000010395.51944.2a
Bi Y, Ehirchiou D, Kilts TM et al (2007) Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med 13:1219–1227. https://doi.org/10.1038/nm1630
Butler DL, Juncosa-Melvin N, Boivin GP et al (2008) Functional tissue engineering for tendon repair: a multidisciplinary strategy using mesenchymal stem cells, bioscaffolds, and mechanical stimulation. J Orthop Res 26:1–9. https://doi.org/10.1002/jor.20456
Caliari SR, Harley BAC (2011) The effect of anisotropic collagen-GAG scaffolds and growth factor supplementation on tendon cell recruitment, alignment, and metabolic activity. Biomaterials 32:5330–5340. https://doi.org/10.1016/j.biomaterials.2011.04.021
Chai W, Ni M, Rui YF et al (2013) Effect of growth and differentiation factor 6 on the tenogenic differentiation of bone marrow-derived mesenchymal stem cells. Chin Med J (engl) 126:1509–1516. https://doi.org/10.3760/cma.j.issn.0366-6999.20123351
Chen X, Song XH, Yin Z et al (2009) Stepwise differentiation of human embryonic stem cells promotes tendon regeneration by secreting fetal tendon matrix and differentiation factors. Stem Cells 27:1276–1287. https://doi.org/10.1002/stem.61
Cheng MT, Yang HW, Chen TH, Lee OK-S (2009) Isolation and characterization of multipotent stem cells from human cruciate ligaments. Cell Prolif 42:448–460. https://doi.org/10.1111/j.1365-2184.2009.00611.x
Dale TP, Mazher S, Webb WR et al (2018) Tenogenic differentiation of human embryonic stem cells. Tissue Eng Part A 24:361–368. https://doi.org/10.1089/ten.tea.2017.0017
Docheva D, Hunziker EB, Fassler R, Brandau O (2005) Tenomodulin is necessary for tenocyte proliferation and tendon maturation. Mol Cell Biol 25:699–705. https://doi.org/10.1128/mcb.25.2.699-705.2005
Docheva D, Müller SA, Majewski M, Evans CH (2015) Biologics for tendon repair. Adv Drug Deliv Rev 84:222–239. https://doi.org/10.1016/j.addr.2014.11.015
Evrova O, Buschmann J (2017) In vitro and in vivo effects of PDGF-BB delivery strategies on tendon healing: a review. Eur Cells Mater 34:15–39. https://doi.org/10.22203/eCM.v034a02
Evrova O, Houska J, Welti M et al (2016) Bioactive, elastic, and biodegradable emulsion electrospun DegraPol tube delivering PDGF-BB for tendon rupture repair. Macromol Biosci. https://doi.org/10.1002/mabi.201500455
Evrova O, Kellenberger D, Calcagni M, Vogel V, Buschmann J (2020) Supporting cell-based tendon therapy: effect of PDGF-BB and ascorbic acid on rabbit Achilles tenocytes In Vitro. Int J Mol Sci 21(2):458. https://doi.org/10.3390/ijms21020458
Favata M, Beredjiklian PK, Zgonis MH et al (2006) Regenerative properties of fetal sheep tendon are not adversely affected by transplantation into an adult environment. J Orthop Res 24:2124–2132. https://doi.org/10.1002/jor.20271
Francis-West PH, Parish J, Lee K, Archer CW (1999) BMP/GDF-signalling interactions during synovial joint development. Cell Tissue Res 296:111–119
Galatz LM, Ball CM, Teefey SA et al (2004) The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Jt Surg Ser A 86:219–224. https://doi.org/10.2106/00004623-200402000-00002
Gonçalves AI, Rodrigues MT, Lee SJ et al (2013) Understanding the role of growth factors in modulating stem cell tenogenesis. PLoS ONE 8:e83734. https://doi.org/10.1371/journal.pone.0083734
Heirani-Tabasi A, Toosi S, Mirahmadi M et al (2017) Chemokine receptors expression in MSCs: comparative analysis in different sources and passages. Tissue Eng Regen Med 14:605–615. https://doi.org/10.1007/s13770-017-0069-7
Hsu C, Chang J (2004) Clinical implications of growth factors in flexor tendon wound healing. J Hand Surg Am 29:551–563. https://doi.org/10.1016/j.jhsa.2004.04.020
Javanshir S, Younesi Soltani F, Dowlati G et al (2020) Induction of tenogenic differentiation of equine adipose-derived mesenchymal stem cells by platelet-derived growth factor-BB and growth differentiation factor-6. Mol Biol Rep 47:6855–6862. https://doi.org/10.1007/s11033-020-05742-7
** Q, Wei G, Lin Z et al (2008) Nanofibrous scaffolds incorporating PDGF-BB microspheres induce chemokine expression and tissue neogenesis in vivo. PLoS ONE 3:e1729. https://doi.org/10.1371/journal.pone.0001729
Lee JY, Zhou Z, Taub PJ et al (2011) BMP-12 treatment of adult mesenchymal stem cells In Vitro augments tendon-like tissue formation and defect repair In Vivo. PLoS ONE 6:e17531. https://doi.org/10.1371/journal.pone.0017531
Léjard V, Brideau G, Blais F et al (2007) Scleraxis and NFATc regulate the expression of the pro-α1(I) collagen gene in tendon fibroblasts. J Biol Chem 282:17665–17675. https://doi.org/10.1074/jbc.M610113200
Maffulli N, Waterston SW, Squair J et al (1999) Changing incidence of Achilles tendon rupture in Scotland: a 15-year study. Clin J Sport Med 9:157–160. https://doi.org/10.1097/00042752-199907000-00007
Maffulli N, Ewen SWB, Waterston SW et al (2000) Tenocytes from ruptured and tendinopathic achilles tendons produce greater quantities of type III collagen than tenocytes from normal achilles tendons: an in vitro model of human tendon healing. Am J Sports Med 28:499–505. https://doi.org/10.1177/03635465000280040901
Martínez-Lorenzo MJ, Royo-Cañas M, Alegre-Aguarón E et al (2009) Phenotype and chondrogenic differentiation of mesenchymal cells from adipose tissue of different species. J Orthop Res 27:1499–1507. https://doi.org/10.1002/jor.20898
Marturano JE, Arena JD, Schiller ZA et al (2013) Characterization of mechanical and biochemical properties of develo** embryonic tendon. Proc Natl Acad Sci USA 110:6370–6375. https://doi.org/10.1073/pnas.1300135110
Molloy T, Wang Y, Murrell GAC (2003) The roles of growth factors in tendon and ligament healing. Sport Med 33:381–394
Naderi N, Combellack EJ, Griffin M et al (2017) The regenerative role of adipose-derived stem cells (ADSC) in plastic and reconstructive surgery. Int Wound J 14:112–124. https://doi.org/10.1111/iwj.12569
Nixon AJ, Dahlgren LA, Haupt JL et al (2008) Effect of adipose-derived nucleated cell fractions on tendon repair in horses with collagenase-induced tendinitis. Am J Vet Res 69:928–937. https://doi.org/10.2460/ajvr.69.7.928
Norelli JB, Plaza DP, Stal DN et al (2018) Tenogenically differentiated adipose-derived stem cells are effective in Achilles tendon repair in vivo. J Tissue Eng. https://doi.org/10.1177/2041731418811183
Pascucci L, Alessandri G, Dall’Aglio C et al (2014) Membrane vesicles mediate pro-angiogenic activity of equine adipose-derived mesenchymal stromal cells. Vet J 202:361–366. https://doi.org/10.1016/j.tvjl.2014.08.021
Pauly S, Klatte F, Strobel C et al (2010) Characterization of tendon cell cultures of the human rotator cuff. Eur Cells Mater 20:84–97. https://doi.org/10.22203/eCM.v020a08
Peister A, Mellad JA, Larson BL et al (2004) Adult stem cells from bone marrow (MSCs) isolated from different strains of inbred mice vary in surface epitopes, rates of proliferation, and differentiation potential. Blood 103:1662–1668. https://doi.org/10.1182/blood-2003-09-3070
Raabe O, Shell K, Fietz D et al (2013) Tenogenic differentiation of equine adipose-tissue-derived stem cells under the influence of tensile strain, growth differentiation factors and various oxygen tensions. Cell Tissue Res 352:509–521. https://doi.org/10.1007/s00441-013-1574-1
Schweitzer R, Chyung JH, Murtaugh LC et al (2001) Analysis of the tendon cell fate using Scleraxis, a specific marker for tendons and ligaments. Development 128:3855–3866
Shukunami C, Takimoto A, Oro M, Hiraki Y (2006) Scleraxis positively regulates the expression of tenomodulin, a differentiation marker of tenocytes. Dev Biol 298:234–247. https://doi.org/10.1016/j.ydbio.2006.06.036
Sieber C, Kopf J, Hiepen C, Knaus P (2009) Recent advances in BMP receptor signaling. Cytokine Growth Factor Rev 20:343–355
Thomopoulos S, Zaegel M, Das R et al (2007) PDGF-BB released in tendon repair using a novel delivery system promotes cell proliferation and collagen remodeling. J Orthop Res 25:1358–1368. https://doi.org/10.1002/jor.20444
Uysal CA, Tobita M, Hyakusoku H, Mizuno H (2012) Adipose-derived stem cells enhance primary tendon repair: biomechanical and immunohistochemical evaluation. J Plast Reconstr Aesthetic Surg 65:1712–1719. https://doi.org/10.1016/j.bjps.2012.06.011
Wang EA, Rosen V, Cordes P et al (1988) Purification and characterization of other distinct bone-inducing factors. Proc Natl Acad Sci USA 85:9484–9488. https://doi.org/10.1073/pnas.85.24.9484
Wolfman NM, Hattersley G, Cox K et al (1997) Ectopic induction of tendon and ligament in rats by growth and differentiation factors 5, 6, and 7, members of the TGF-β gene family. J Clin Invest 100:321–330. https://doi.org/10.1172/JCI119537
Woo SL-Y, Debski RE, Zeminski J et al (2000) Injury and repair of ligaments and tendons. Annu Rev Biomed Eng 2:83–118. https://doi.org/10.1146/annurev.bioeng.2.1.83
Wozney JM (1992) The bone morphogenetic protein family and osteogenesis. Mol Reprod Dev 32:160–167. https://doi.org/10.1002/mrd.1080320212
Wozney JM, Rosen V, Celeste AJ et al (1988) Novel regulators of bone formation: molecular clones and activities. Science 242:1528–1534. https://doi.org/10.1126/science.3201241
Yang F, Richardson DW (2021) Comparative analysis of tenogenic gene expression in tenocyte-derived induced pluripotent stem cells and bone marrow-derived mesenchymal stem cells in response to biochemical and biomechanical stimuli. Stem Cells Int. https://doi.org/10.1155/2021/8835576
Yang G, Rothrauff BB, Tuan RS (2013) Tendon and ligament regeneration and repair: clinical relevance and developmental paradigm. Birth Defects Res Part C Embryo Today Rev 99:203–222. https://doi.org/10.1002/bdrc.21041
Yang G, Rothrauff BB, Lin H et al (2017) Tendon-derived extracellular matrix enhances transforming growth factor-β3-induced tenogenic differentiation of human adipose-derived stem cells. Tissue Eng Part A 23:166–176. https://doi.org/10.1089/ten.tea.2015.0498
Yin Z, Chen X, Chen JL, Ouyang HW (2010) Stem cells for tendon tissue engineering and regeneration. Expert Opin Biol Ther 10:689–700
Yin Z, Guo J, Wu T et al (2016) Stepwise differentiation of mesenchymal stem cells augments tendon-like tissue formation and defect repair in vivo. Stem Cells Transl Med 5:1108–1116
Yu Y, Bliss JP, Bruce WJM, Walsh WR (2007) Bone morphogenetic proteins and smad expression in ovine tendon-bone healing. Arthrosc J Arthrosc Relat Surg 23:205–210. https://doi.org/10.1016/j.arthro.2006.08.023
Acknowledgements
This study was financially supported by Ferdowsi University of Mashhad.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
All experiments were conducted with the approval of the Ethics Committee of Ferdowsi University of Mashhad, and Academic Center for Education, Culture and Research.
Informed consent
Human ASCs samples were obtained from the Stem Cell Bank in Academic Center for Education, Culture and Research (ACECR), Mashhad. Adipose tissue samples were previously collected for research purposes by the consent of all participants in ACECR.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Younesi Soltani, F., Javanshir, S., Dowlati, G. et al. Differentiation of human adipose-derived mesenchymal stem cells toward tenocyte by platelet-derived growth factor-BB and growth differentiation factor-6. Cell Tissue Bank 23, 237–246 (2022). https://doi.org/10.1007/s10561-021-09935-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-021-09935-7