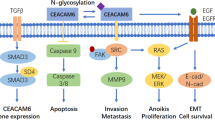
Abstract
Carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) is a broadly-expressed immunoglobulin-like cell adhesion molecule with a wide range of biological functions to regulate cell signaling. The present article mainly focuses on the role of CEACAM1 as a therapeutic target in lung diseases and discusses the potential of therapeutic strategies targeting CEACAM1. The article overviews the structure and its sub-types, biological function, and potential roles of CEACAM1 in lung diseases. Alterations of CEACAM1 expression and CEACAM1-S/CEACAM1-L ratio promote the growth and metastasis of non-small cell lung carcinoma (NSCLC). Moreover, CEACAM1 mediates bacterial adherence and transcellular transcytosis, resulting in the suppression of immune cell activities and inflammatory responses, which may trigger acute exacerbation of chronic obstructive pulmonary disease (AECOPD). CEACAM1 plays a critical role in the development of NSCLC and AECOPD and can be a diagnostic biomarker and therapeutic target in lung diseases.






Similar content being viewed by others
References
Kuespert, K., Pils, S., & Hauck, C. R. (2006). CEACAMs: their role in physiology and pathophysiology. Current Opinion in Cell Biology, 18(5), 565–571.
Obrink, B. (2008). On the role of CEACAM1 in cancer. Lung Cancer, 60(3), 309–312.
Ullric, N., Heinemann, A., Nilewski, E., Scheffrahn, I., Klode, J., Scherag, A., Schadendorf, D., Singer, B. B., Helfrich, I. (2015). CEACAM1-3S drives melanoma cells into NK cell-mediated cytolysis and enhances patient survival. Cancer Research, 75(9), 1897–1907.
Beauchemin, N., & Arabzadeh, A. (2013). Carcinoembryonic antigen-related cell adhesion molecules (CEACAMs) in cancer progression and metastasis. Cancer and Metastasis Reviews, 32(3–4), 643–671.
Gray-Owen, S. D., & Blumberg, R. S. (2006). CEACAM1: contact-dependent control of immunity. Nature Reviews Immunology, 6(6), 433–446.
Berardi, R., Morgese, F., Onofri, A., Mazzanti, P., Pistelli, M., Ballatore, Z., et al. (2013). Role of maspin in cancer. Clinical and Translational Medicine, 2, 8.
Chen, C. J., Kirshner, J., Sherman, M. A., Hu, W., Nguyen, T., & Shively, J. E. (2007). Mutation analysis of the short cytoplasmic domain of the cell–cell adhesion molecule CEACAM1 identifies residues that orchestrate actin binding and lumen formation. Journal of Biological Chemistry, 282(8), 5749–5760.
Gaur, S., Shively, J. E., Yen, Y., & Gaur, R. K. (2008). Altered splicing of CEACAM1 in breast cancer: identification of regulatory sequences that control splicing of CEACAM1 into long or short cytoplasmic domain isoforms. Molecular Cancer, 7, 46.
Dery, K. J., Gaur, S., Gencheva, M., Yen, Y., Shively, J. E., & Gaur, R. K. (2011). Mechanistic control of carcinoembryonic antigen-related cell adhesion molecule-1 (CEACAM1) splice isoforms by the heterogeneous nuclear ribonuclear proteins hnRNP L, hnRNP A1, and hnRNP M. Journal of Biological Chemistry, 286(18), 16039–16051.
Klaile, E., Vorontsova, O., Sigmundsson, K., Svensson, S., Skoglund, U., & Obrink, B. (2009). The CEACAM1 N-terminal Ig domain mediates cis- and trans-binding and is essential for allosteric rearrangements of CEACAM1 microclusters. Journal of Cell Biology, 187(4), 553–567.
Müller, M. M., Klaile, E., Vorontsova, O., Singer, B. B., & Obrink, B. (2009). Homophilic adhesion and CEACAM1-S regulate dimerization of CEACAM1-L and recruitment of SHP-2 and c-Src. Journal of Cell Biology, 187(4), 569–581.
Brümmer, J., Neumaier, M., Göpfert, C., & Wagener, C. (1995). Association of pp60c-src with biliary glycoprotein (CD66a), an adhesion molecule of the carcinoembryonic antigen family downregulated in colorectal carcinomas. Oncogene, 11(8), 1649–1655.
Huber, M., Izzi, L., Grondin, P., Houde, C., Kunath, T., Veillette, A., & Beauchemin, N. (1999). The carboxyl-terminal region of biliary glycoprotein controls its tyrosine phosphorylation and association with protein-tyrosine phosphatases SHP-1 and SHP-2 in epithelial cells. Journal of Biological Chemistry, 274(1), 335–344.
Nagaishi, T., Pao, L., Lin, S. H., Iijima, H., Kaser, A., Qiao, S. W., Chen, Z., Glickman, J., Najjar, S. M., Nakajima, A., Neel, B. G., & Blumberg, R. S. (2006). SHP1 phosphatase-dependent T cell inhibition by CEACAM1 adhesion molecule isoforms. Immunity, 25(5), 769–781.
Najjar, S. M. (2002). Regulation of insulin action by CEACAM1. Trends in Endocrinology and Metabolism, 13(6), 240–245.
Poy, M. N., Ruch, R. J., Fernstrom, M. A., Okabayashi, Y., & Najjar, S. M. (2002). Shc and CEACAM1 interact to regulate the mitogenic action of insulin. Journal of Biological Chemistry, 277(2), 1076–1084.
Liu, W., Wei, W., Winer, D., Bamberger, A. M., Bamberger, C., Wagener, C., Ezzat, S., & Asa, S. L. (2007). CEACAM1 impedes thyroid cancer growth but promotes invasiveness: a putative mechanism for early metastases. Oncogene, 26(19), 2747–2758.
Abou-Rjaily, G. A., Lee, S. J., May, D., Al-Share, Q. Y., Deangelis, A. M., Ruch, R. J., Neumaier, M., Kalthoff, H., Lin, S. H., & Najjar, S. M. (2004). CEACAM1 modulates epidermal growth factor receptor–mediated cell proliferation. Journal of Clinical Investigation, 114(7), 944–952.
Ergün, S., Kilik, N., Ziegeler, G., Hansen, A., Nollau, P., Götze, J., Wurmbach, J. H., Horst, A., Weil, J., Fernando, M., & Wagener, C. (2000). CEA-related cell adhesion molecule 1: a potent angiogenic factor and a major effector of vascular endothelial growth factor. Molecular Cell, 5(2), 311–320.
Lohela, M., Bry, M., Tammela, T., & Alitalo, K. (2009). VEGFs and receptors involved in angiogenesis versus lymphangiogenesis. Current Opinion in Cell Biology, 21(2), 154–165.
Kilic, N., Oliveira-Ferrer, L., Wurmbach, J. H., Loges, S., Chalajour, F., Neshat-Vahid, S., Weil, J., Fernando, M., & Ergun, S. (2005). Pro-angiogenic signaling by the endothelial presence of CEACAM1. Journal of Biological Chemistry, 280(3), 2361–2369.
Horst, A. K., Ito, W. D., Dabelstein, J., Schumacher, U., Sander, H., Turbide, C., Brümmer, J., Meinertz, T., Beauchemin, N., & Wagener, C. (2006). Carcinoembryonic antigen-related cell adhesion molecule 1 modulates vascular remodeling in vitro and in vivo. Journal of Clinical Investigation, 116(6), 1596–1605.
Gurda, G. T., Zhang, L., Wang, Y., Chen, L., Geddes, S., William, C., Cho, W. C., Askin, F., Gabrielson, E., & Li, Q. (2015). Utility of five commonly used immunohistochemical markers TTF-1, Napsin A, CK7, CK5/6 and P63 in primary and metastatic adenocarcinoma and squamous cell carcinoma of the lung: a retrospective study of 246 fine needle aspiration cases. Clinical and Translational Medicine, 4, 16.
Nouvion, A. L., Oubaha, M., Leblanc, S., Davis, E. C., Jastrow, H., Kammerer, R., Breton, V., Turbide, C., Ergun, S., Gratton, J. P., & Beauchemin, N. (2010). CEACAM1: a key regulator of vascular permeability. Journal of Cell Science, 123(Pt 24), 4221–4230.
Kilic, N., Oliveira-Ferrer, L., Neshat-Vahid, S., Irmak, S., Obst-Pernberg, K., Wurmbach, J. H., Loges, S., Kilic, E., Weil, J., Lauke, H., Tilki, D., Singer, B. B., & Ergün, S. (2007). Lymphatic reprogramming of microvascular endothelial cells by CEA-related cell adhesion molecule-1 via interaction with VEGFR-3 and Prox1. Blood, 110(13), 4223–4233.
Oliveira-Ferrer, L., Tilki, D., Ziegeler, G., Hauschild, J., Loges, S., Irmak, S., Kilic, E., Huland, H., Friedrich, M., & Ergün, S. (2004). Dual role of carcinoembryonic antigen-related cell adhesion molecule 1 in angiogenesis and invasion of human urinary bladder cancer. Cancer Research, 64(24), 8932–8938.
Tilki, D., Irmak, S., Oliveira-Ferrer, L., Hauschild, J., Miethe, K., Atakaya, H., Hammerer, P., Friedrich, M. G., Schuch, G., Galalae, R., Stief, C. G., Kilic, E., Huland, H., & Ergun, S. (2006). CEA-related cell adhesion molecule-1 is involved in angiogenic switch in prostate cancer. Oncogene, 25(36), 4965–4974.
Nittka, S., Günther, J., Ebisch, C., Erbersdobler, A., & Neumaier, M. (2004). The human tumor suppressor CEACAM1 modulates apoptosis and is implicated in early colorectal tumorigenesis. Oncogene, 23(58), 9306–9313.
Singer, B. B., Klaile, E., Scheffrahn, I., Müller, M. M., Kammerer, R., Reutter, W., Obrink, B., & Lucka, L. (2005). CEACAM1 (CD66a) mediates delay of spontaneous and Fas ligand-induced apoptosis in granulocytes. European Journal of Immunology, 35(6), 1949–1959.
Yu, Q., Chow, E. M., Wong, H., Gu, J., Mandelboim, O., Gray-Owen, S. D., & Ostrowski, M. A. (2006). CEACAM1 (CD66a) promotes human monocyte survival via a phosphatidylinositol 3-kinase- and AKT-dependent pathway. Journal of Biological Chemistry, 281(51), 39179–39193.
Li, Y., & Shively, J. E. (2013). CEACAM1 regulates Fas-mediated apoptosis in Jurkat T-cells via its interaction with β-catenin. Experimental Cell Research, 319(8), 1061–1072.
Lobo, E. O., Zhang, Z., & Shively, J. E. (2009). Pivotal advance: CEACAM1 is a negative coreceptor for the B cell receptor and promotes CD19-mediated adhesion of B cells in a PI3K-dependent manner. Journal of Leukocyte Biology, 86(2), 205–218.
Huang, J., Hardy, J. D., Sun, Y., & Shively, J. E. (1999). Essential role of biliary glycoprotein (CD66a) in morphogenesis of the human mammary epithelial cell line MCF10F. Journal of Cell Science, 112(Pt23), 4193–4205.
Kirshner, J., Chen, C. J., Liu, P., Huang, J., & Shively, J. E. (2003). CEACAM1-4S, a cell-cell adhesion molecule, mediates apoptosis and reverts mammary carcinoma cells to a normal morphogenic phenotype in a 3D culture. Proceedings of the National Academy of Sciences of the United States of America, 100(2), 521–526.
Chen, C. J., Nguyen, T., & Shively, J. E. (2010). Role of calpain-9 and PKC-delta in the apoptotic mechanism of lumen formation in CEACAM1 transfected breast epithelial cells. Experimental Cell Research, 316(4), 638–648.
Arabzadeh, A., & Beauchemin, N. (2012). Stromal CEACAM1 expression regulates colorectal cancer metastasis. Oncoimmunology, 1(7), 1205–1207.
Ieda, J., Yokoyama, S., Tamura, K., Takifuji, K., Hotta, T., Matsuda, K., Oku, Y., Nasu, T., Kiriyama, S., Yamamoto, N., Nakamura, Y., Shively, J. E., & Yamaue, H. (2011). Re-expression of CEACAM1 long cytoplasmic domain isoform is associated with invasion and migration of colorectal cancer. International Journal of Cancer, 129(6), 1351–1361.
Ebrahimnejad, A., Streichert, T., Nollau, P., Horst, A. K., Wagener, C., Bamberger, A. M., & Brümmer, J. (2004). CEACAM1 enhances invasion and migration of melanocytic and melanoma cells. American Journal of Pathology, 165(5), 1781–1787.
Bajaj, J., Maliekal, T. T., Vivien, E., Pattabiraman, C., Srivastava, S., Krishnamurthy, H., Giri, V., Subramanyam, D., & Krishna, S. (2011). Notch signaling in CD66+ cells drives the progression of human cervical cancers. Cancer Research, 71(14), 4888–4897.
Liu, J., Di, G., Wu, C. T., Hu, X., & Duan, H. (2013). CEACAM1 inhibits cell-matrix adhesion and promotes cell migration through regulating the expression of N-cadherin. Biochemical and Biophysical Research Communications, 430(2), 598–603.
Arabzadeh, A., Chan, C., Nouvion, A. L., Breton, V., Benlolo, S., DeMarte, L., Turbide, C., Brodt, P., Ferri, L., & Beauchemin, N. (2013). Host-related carcinoembryonic antigen cell adhesion molecule 1 promotes metastasis of colorectal cancer. Oncogene, 32(7), 849–860.
Virji, M., Watt, S. M., Barker, S., Makepeace, K., & Doyonnas, R. (1996). The N-domain of the human CD66a adhesion molecule is a target for Opa proteins of Neisseria meningitidis and Neisseria gonorrhoeae. Molecular Microbiology, 22(5), 929–939.
Hill, D. J., & Virji, M. (2003). A novel cell-binding mechanism of Moraxella catarrhalis ubiquitous surface protein UspA: specific targeting of the N-domain of carcinoembryonic antigen-related cell adhesion molecules by UspA1. Molecular Microbiology, 48(1), 117–129.
Hill, D. J., Toleman, M. A., Evans, D. J., Villullas, S., Van Alphen, L., & Virji, M. (2001). The variable P5 proteins of typeable and non-typeable Haemophilus influenzae target human CEACAM1. Molecular Microbiology, 39(4), 850–862.
Berger, C. N., Billker, O., Meyer, T. F., Servin, A. L., & Kansau, I. (2004). Differential recognition of members of the carcinoembryonic antigen family by Afa/Dr adhesins of diffusely adhering Escherichia coli (Afa/Dr DAEC). Molecular Microbiology, 52(4), 963–983.
Hill, D. J., Edwards, A. M., Rowe, H. A., & Virji, M. (2005). Carcinoembryonic antigen-related cell adhesion molecule (CEACAM)-binding recombinant polypeptide confers protection against infection by respiratory and urogenital pathogens. Molecular Microbiology, 55(5), 1515–1527.
Bookwalter, J. E., Jurcisek, J. A., Gray-Owen, S. D., Fernandez, S., McGillivary, G., & Bakaletz, L. O. (2008). A carcinoembryonic antigen-related cell adhesion molecule 1 homologue plays a pivotal role in nontypeable Haemophilus influenzae colonization of the chinchilla nasopharynx via the outer membrane protein P5-homologous adhesin. Infection and Immunity, 76(1), 48–55.
Sadarangani, M., Pollard, A. J., & Gray-Owen, S. D. (2011). Opa proteins and CEACAMs: pathways of immune engagement for pathogenic Neisseria. FEMS Microbiology Reviews, 35(3), 498–514.
Perez Vidakovics, M. L., & Riesbeck, K. (2009). Virulence mechanisms of Moraxella in the pathogenesis of infection. Current Opinion in Infectious Diseases, 22(3), 279–285.
Boulton, I. C., & Gray-Owen, S. D. (2002). Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes. Nature Immunology, 3(3), 229–236.
Lee, H. S., Ostrowski, M. A., & Gray-Owen, S. D. (2008). CEACAM1 dynamics during neisseria gonorrhoeae suppression of CD4+ T lymphocyte activation. Journal of Immunology, 180(10), 6827–6835.
Slevogt, H., Zabel, S., Opitz, B., Hocke, A., Eitel, J., N’guessan, P. D., Lucka, L., Riesbeck, K., Zimmermann, W., Zweigner, J., Temmesfeld-Wollbrueck, B., Suttorp, N., & Singer, B. B. (2008). CEACAM1 inhibits toll-like receptor 2–triggered antibacterial responses of human pulmonary epithelial cells. Nature Immunology, 9(11), 1270–1278.
Alamgeer, M., Ganju, V., & Watkins, D. N. (2013). Novel therapeutic targets in non-small cell lung cancer. Current Opinion in Pharmacology, 13(3), 394–401.
Al Olayan, A., Al Hussaini, H., & Jazieh, A. R. (2012). The roles of epidermal growth factor receptor (EGFR) inhibitors in the management of lung cancer. Journal of Infection and Public Health, 5(S1), S50–S60.
Ohwada, A., Takahashi, H., Nagaoka, I., & Kira, S. (1994). Biliary glycoprotein mRNA expression is increased in primary lung cancer, especially in squamous cell carcinoma. American Journal of Respiratory Cell and Molecular Biology, 11(2), 214–220.
Laack, E., Nikbakht, H., Peters, A., Kugler, C., Jasiewicz, Y., Edler, L., Brümmer, J., Schumacher, U., & Hossfeld, D. K. (2002). Expression of CEACAM1 in adenocarcinoma of the lung: a factor of independent prognostic significance. Journal of Clinical Oncology, 20(21), 4279–4284.
Sienel, W., Dango, S., Woelfle, U., Morresi-Hauf, A., Wagener, C., Brümmer, J., Mutschler, W., Passlick, B., & Pantel, K. (2003). Elevated expression of carcinoembryonic antigen-related cell adhesion molecule 1 promotes progression of non-small cell lung cancer. Clinical Cancer Research, 9(6), 2260–2266.
Thöm, I., Schult-Kronefeld, O., Burkholder, I., Schuch, G., Andritzky, B., Kastendieck, H., Edler, L., Wagener, C., Bokemeyer, C., Schumacher, U., & Laack, E. (2009). Expression of CEACAM-1 in pulmonary adenocarcinomas and their metastases. Anticancer Research, 29(1), 249–254.
Dango, S., Sienel, W., Schreiber, M., Stremmel, C., Kirschbaum, A., Pantel, K., & Passlick, B. (2008). Elevated expression of carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM-1) is associated with increased angiogenic potential in non-small-cell lung cancer. Lung Cancer, 60(3), 426–433.
Wang, L., Lin, S. H., Wu, W. G., Kemp, B. L., Walsh, G. L., Hong, W. K., & Mao, L. (2000). C-CAM1, a candidate tumor suppressor gene, is abnormally expressed in primary lung cancers. Clinical Cancer Research, 6(8), 2988–2993.
Zhou, M. Q., Du, Y., Liu, Y. W., Wang, Y. Z., He, Y. Q., Yang, C. X., Wang, W. J., & Gao, F. (2013). Clinical and experimental studies regarding the expression and diagnostic value of carcinoembryonic antigen-related cell adhesion molecule 1 in non-small-cell lung cancer. BMC Cancer, 13, 359.
Sethi, S., Yin, J., Pamela, K., & Anderson, P. K. (2014). Lung flute improves symptoms and health status in COPD with chronic bronchitis: a 26 week randomized controlled trial. Clinical and Translational Medicine, 3, 29.
Fang, X., Li, S., Gao, L., Zhao, N., Wang, X., & Bai, C. (2012). A short-term educational program improved physicians’ adherence to guidelines for COPD and asthma in Shanghai. Clinical and Translational Medicine, 1, 13.
Klaile, E., Klassert, T. E., Scheffrahn, I., Müller, M. M., Heinrich, A., Heyl, K. A., Dienemann, H., Grünewald, C., Bals, R., Singer, B. B., & Slevogt, H. (2013). Carcinoembryonic antigen (CEA)-related cell adhesion molecules are co-expressed in the human lung and their expression can be modulated in bronchial epithelial cells by non-typable Haemophilus influenzae, Moraxella catarrhalis, TLR3, and type I and II interferons. Respiratory Research, 14, 85.
Wu, X., Sun, X., Chen, C., Bai, C., & Wang, X. D. (2014). Dynamic gene expressions of peripheral blood mononuclear cells in patients with acute exacerbation of chronic obstructive pulmonary disease: a preliminary study. Critical Care, 18(6), 508.
Wu, X., Chen, L., & Wang, X. D. (2014). Network biomarkers, interaction networks and dynamical network biomarkers in respiratory diseases. Clinical and Translational Medicine, 3, 16.
Sohal, S., Mahmood, M., & Walters, E. (2014). Clinical significance of epithelial mesenchymal transition (EMT) in chronic obstructive pulmonary disease (COPD): potential target for prevention of airway fibrosis and lung cancer. Clinical and Translational Medicine, 3, 33.
Wu, X., Yuan, B., López, E., Bai, C., & Wang, X. (2014). Gene polymorphisms and chronic obstructive pulmonary disease. Journal of Cellular and Molecular Medicine, 18(1), 15–26.
Reichert, J. M. (2012). Marketed therapeutic antibodies compendium. MAbs, 4(3), 413–415.
Markel, G., Wolf, D., Hanna, J., Gazit, R., Goldman-Wohl, D., Lavy, Y., Yagel, S., & Mandelboim, O. (2002). Pivotal role of CEACAM1 protein in the inhibition of activated decidual lymphocyte functions. Journal of Clinical Investigation, 110(7), 943–953.
Markel, G., Lieberman, N., Katz, G., Arnon, T. I., Lotem, M., Drize, O., Blumberg, R. S., Bar-Haim, E., Mader, R., Eisenbach, L., & Mandelboim, O. (2002). CD66a interactions between human melanoma and NK cells: a novel class I MHC-independent inhibitory mechanism of cytotoxicity. Journal of Immunology, 168(6), 2803–2810.
Markel, G., Seidman, R., Stern, N., Cohen-Sinai, T., Izhaki, O., Katz, G., Besser, M., Treves, A. J., Blumberg, R. S., Loewenthal, R., Mandelboim, O., Orenstein, A., & Schachter, J. (2006). Inhibition of human tumor-infiltrating lymphocyte effector functions by the homophilic carcinoembryonic cell adhesion molecule 1 interactions. Journal of Immunology, 177(9), 6062–6071.
Markel, G., Seidman, R., Cohen, Y., Besser, M. J., Sinai, T. C., Treves, A. J., Orenstein, A., Berger, R., & Schachter, J. (2009). Dynamic expression of protective CEACAM1 on melanoma cells during specific immune attack. Immunology, 126(2), 186–200.
Sapoznik, S., Hammer, O., Ortenberg, R., Besser, M. J., Ben-Moshe, T., Schachter, J., & Markel, G. (2012). Novel anti-melanoma immunotherapies: disarming tumor escape mechanisms. Journal of Immunology Research, 2012, 818214.
Ortenberg, R., Sapir, Y., Raz, L., Hershkovitz, L., Ben Arav, A., Sapoznik, S., Barshack, I., Avivi, C., Berkun, Y., Besser, M. J., Ben-Moshe, T., Schachter, J., & Markel, G. (2012). Novel immunotherapy for malignant melanoma with a monoclonal antibody that blocks CEACAM1 homophilic interactions. Molecular Cancer Therapeutics, 11(6), 1300–1310.
Liu, J., Di, G., Wu, C. T., Hu, X., & Duan, H. (2013). Development and evaluation of a novel anti-colorectal cancer monoclonal antibody, WL5. Biochemical and Biophysical Research Communications, 432(2), 370–377.
Holliger, P., & Hudson, P. J. (2005). Engineered antibody fragments and the rise of single domains. Nature Biotechnology, 23(9), 1126–1136.
Moricoli, D., Laguardia, M. E., Carbonella, D. C., Balducci, M. C., Dominici, S., Fiori, V., Serafini, G., Flego, M., Cianfriglia, M., & Magnani, M. (2014). Isolation of a new human scFv antibody recognizing a cell surface binding site to CEACAM1. Large yield production, purification and characterization in E. coli expression system. Protein Expression and Purification, 93, 38–45.
Lauro, S., Onesti, C. E., Righini, R., & Marchetti, P. (2014). The use of bevacizumab in non-small cell lung cancer: an update. Anticancer Research, 34(4), 1537–1545.
Acknowledgments
The work was supported by Zhongshan Distinguished Professor Grant (XDW), The National Nature Science Foundation of China (91230204, 81270099, 81320108001, 81270131, 81300010), The Shanghai Committee of Science and Technology (12JC1402200, 12431900207, 11410708600, 14431905100), Operation funding of Shanghai Institute of Clinical Bioinformatics, and Ministry of Education, Academic Special Science and Research Foundation for PhD Education (20130071110043).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ling, Y., Wang, J., Wang, L. et al. Roles of CEACAM1 in cell communication and signaling of lung cancer and other diseases. Cancer Metastasis Rev 34, 347–357 (2015). https://doi.org/10.1007/s10555-015-9569-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-015-9569-x