Abstract
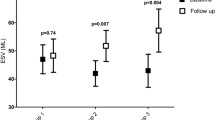
Sparse information is available on the role of cardiac viability imaging in elderly patients. We aimed at evaluating the prognostic value of FDG-PET/CT in elderly patients with stable coronary artery disease (CAD) and reduced left ventricular ejection fraction (rLVEF) before revascularisation. Elderly patients (> 65 years old, mean 74 ± 7 years old) with CAD and rLVEF were followed after cardiac FDG-PET/CT and stratified according to presence/absence of viable myocardium and subsequent revascularisation. Fatal events of any cause as well as hospitalisations related to acute cardiac conditions were reported as clinical end-points. Predictors of fatal events in patients with viable myocardium (> 1 myocardium segment/20) were analysed. A total of 89 patients were followed (64 viable myocardia; 37 and 27 patients with and without subsequent revascularisation, respectively). The change in LVEF during follow-up (2.1 ± 1.6 years) was 3.8 ± 6.6% (P = 0.013) and − 0.75 ± 2.6% (P = 0.170) in patients with and without revascularisation, respectively. Log-rank (P = 0.037) and multivariate analysis (Wald: 6.305, P = 0.012) showed viable myocardium to be significantly associated with fatal events if not revascularised. Elderly patients with viable myocardium might potentially benefit from revascularisation procedures as improved left ventricular ejection fraction and survival were observed in our retrospective study as compared to patients in whom a revascularisation procedure was denied. Viable myocardium as detected by cardiac FDG PET/CT was associated with better clinical outcomes in elderly patients when revascularised.



Similar content being viewed by others
References
Moran AE, Forouzanfar MH, Roth GA, Mensah GA, Ezzati M, Murray CJ et al (2014) Temporal trends in ischemic heart disease mortality in 21 world regions, 1980 to 2010: the Global Burden of Disease 2010 study. Circulation 129(14):1483–1492
Borlaug BA, Redfield MM, Melenovsky V, Kane GC, Karon BL, Jacobsen SJ et al (2013) Longitudinal changes in left ventricular stiffness: a community-based study. Circ Heart Fail 6(5):944–952
Olivetti G, Melissari M, Capasso JM, Anversa P (1991) Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circ Res 68(6):1560–1568
Selby DE, Palmer BM, LeWinter MM, Meyer M (2011) Tachycardia-induced diastolic dysfunction and resting tone in myocardium from patients with a normal ejection fraction. J Am Coll Cardiol 58(2):147–154
van Heerebeek L, Franssen CP, Hamdani N, Verheugt FW, Somsen GA, Paulus WJ (2012) Molecular and cellular basis for diastolic dysfunction. Curr Heart Fail Rep 9(4):293–302
Sardar MR, Badri M, Prince CT, Seltzer J, Kowey PR (2014) Underrepresentation of women, elderly patients, and racial minorities in the randomized trials used for cardiovascular guidelines. JAMA Intern Med 174(11):1868–1870
Shanmugam VB, Harper R, Meredith I, Malaiapan Y, Psaltis PJ (2015) An overview of PCI in the very elderly. J Geriatr Cardiol 12(2):174–184
Moscucci M, Fox KA, Cannon CP, Klein W, Lopez-Sendon J, Montalescot G et al (2003) Predictors of major bleeding in acute coronary syndromes: the Global Registry of Acute Coronary Events (GRACE). Eur Heart J 24(20):1815–1823
Feldman DN, Gade CL, Slotwiner AJ, Parikh M, Bergman G, Wong SC et al (2006) Comparison of outcomes of percutaneous coronary interventions in patients of three age groups (< 60, 60 to 80, and> 80 years) (from the New York State Angioplasty Registry). Am J Cardiol 98(10):1334–1339.
Orlandini A, Castellana N, Pascual A, Botto F, Cecilia Bahit M, Chacon C et al (2015) Myocardial viability for decision-making concerning revascularization in patients with left ventricular dysfunction and coronary artery disease: a meta-analysis of non-randomized and randomized studies. Int J Cardiol 182:494–499
Mc Ardle B, Shukla T, Nichol G, deKemp RA, Bernick J, Guo A et al ( 2016) Long-term follow-up of outcomes with F-18-fluorodeoxyglucose positron emission tomography imaging-assisted management of patients with severe left ventricular dysfunction secondary to coronary disease. Circ Cardiovas Imaging 9(9):e004331
Ratib O, Phelps ME, Huang SC, Henze E, Selin CE, Schelbert HR (1982) Positron tomography with deoxyglucose for estimating local myocardial glucose metabolism. J Nucl Med 23(7):577–586
Dilsizian V, Bacharach SL, Beanlands RS, Bergmann SR, Delbeke D, Dorbala S et al (2016) ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol 23(5):1187–1226
Nkoulou R, Pazhenkottil AP, Buechel RR, Husmann L, Valenta I, Herzog BA et al (2011) Impact of CT attenuation correction on the viability pattern assessed by 99mTc-tetrofosmin SPECT/ 18F-FDG PET. Int J Cardiovasc Imaging 27(6):913–921
Di Carli MF, Davidson M, Little R, Khanna S, Mody FV, Brunken RC et al (1994) Value of metabolic imaging with positron emission tomography for evaluating prognosis in patients with coronary artery disease and left ventricular dysfunction. Am J Cardiol 73(8):527–533
Thygesen K, Alpert JS, White HD et al (2007) Universal definition of myocardial infarction. J Am Coll Cardiol 50(22):2173–2195
Tillisch J, Brunken R, Marshall R, Schwaiger M, Mandelkern M, Phelps M et al (1986) Reversibility of cardiac wall-motion abnormalities predicted by positron tomography. New Engl J Med 314(14):884–888
Braunwald E, Rutherford JD (1986) Reversible ischemic left ventricular dysfunction: evidence for the “hibernating myocardium”. J Am Coll Cardiol 8(6):1467–1470
Rahimtoola SH (1982) Coronary bypass surgery for chronic angina–1981. A perspective. Circulation 65(2):225–241
Allman KC, Shaw LJ, Hachamovitch R, Udelson JE (2002) Myocardial viability testing and impact of revascularization on prognosis in patients with coronary artery disease and left ventricular dysfunction: a meta-analysis. J Am Coll Cardiol 39(7):1151–1158
Inaba Y, Chen JA, Bergmann SR (2010) Quantity of viable myocardium required to improve survival with revascularization in patients with ischemic cardiomyopathy: a meta-analysis. J Nucl Cardiol 17(4):646–654
Pavlou M, Ambler G, Seaman SR, Guttmann O, Elliott P, King M et al (2015) How to develop a more accurate risk prediction model when there are few events. BMJ 351:h3868
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Namdar, M., Rager, O., Priamo, J. et al. Prognostic value of revascularising viable myocardium in elderly patients with stable coronary artery disease and left ventricular dysfunction: a PET/CT study. Int J Cardiovasc Imaging 34, 1673–1678 (2018). https://doi.org/10.1007/s10554-018-1380-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-018-1380-7