Abstract
Purpose
The use of the Oncotype DX recurrence score (RS) to predict chemotherapy benefit in patients with hormone receptor-positive/HER2 negative (HR+/HER2-) breast cancer has recently expanded to include postmenopausal patients with N1 disease. RS availability is limited in resource-poor settings, however, prompting the development of statistical models that predict RS using clinicopathologic features. We sought to assess the performance of our supervised machine learning model in a cohort of patients > 50 years of age with N1 disease.
Methods
We identified patients > 50 years of age with pT1-2N1 HR+/HER2- breast cancer and applied the statistical model previously developed in a node-negative cohort, which uses age, pathologic tumor size, histology, progesterone receptor expression, lymphovascular invasion, and tumor grade to predict RS. We measured the model’s ability to predict RS risk category (low: RS ≤ 25; high: RS > 25).
Results
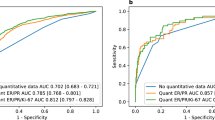
Our cohort included 401 patients, 60.6% of whom had macrometastases, with a median of 1 positive node. The majority of patients had a low-risk observed RS (85.8%). For predicting RS category, the model had specificity of 97.3%, sensitivity of 31.8%, a negative predictive value of 87.9%, and a positive predictive value of 70.0%.
Conclusion
Our model, developed in a cohort of node-negative patients, was highly specific for identifying cN1 patients > 50 years of age with a low RS who could safely avoid chemotherapy. The use of this model for identifying patients in whom genomic testing is unnecessary would help decrease the cost burden in resource-poor settings as reliance on RS for adjuvant treatment recommendations increases.

Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the study are available from the corresponding author on reasonable request.
References
Paik S, Shak S, Tang G et al (2004) A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med 351:2817–2826. https://doi.org/10.1056/NEJMoa041588
Paik S, Tang G, Shak S et al (2006) Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol 24:3726–3734. https://doi.org/10.1200/JCO.2005.04.7985
Sparano JA, Gray RJ, Makower DF et al (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379:111–121. https://doi.org/10.1056/NEJMoa1804710
Lund MJ, Mosunjac M, Davis KM et al (2012) 21-Gene recurrence scores: racial differences in testing, scores, treatment, and outcome. Cancer 118:788–796. https://doi.org/10.1002/CNCR.26180
Guth AA, Fineberg S, Fei K et al (2013) Utilization of oncotype DX in an inner city population: race or place? Int J Breast Cancer 2013:1–3. https://doi.org/10.1155/2013/653805
Cress RD, Chen YS, Morris CR et al (2016) Underutilization of gene expression profiling for early-stage breast cancer in California. Cancer Causes Control 27:721–727. https://doi.org/10.1007/S10552-016-0743-4/TABLES/3
Press DJ, Ibraheem A, Dolan ME et al (2018) Racial disparities in omission of oncotype DX but no racial disparities in chemotherapy receipt following completed oncotype DX test results. Breast Cancer Res Treat 168:207–220. https://doi.org/10.1007/s10549-017-4587-8
Kozick Z, Hashmi A, Dove J et al (2018) Disparities in compliance with the oncotype DX breast cancer test in the United States: a national cancer data base assessment. Am J Surg 215:686–692. https://doi.org/10.1016/J.AMJSURG.2017.05.008
Roberts MC, Kurian AW, Petkov VI (2019) Uptake of the 21-gene assay among women with node-positive, hormone receptor-positive breast cancer. J Natl Compr Canc Netw 17:662–668. https://doi.org/10.6004/JNCCN.2018.7266
Choi JDW, Hughes TMD, Marx G et al (2022) The utility of the oncotype DX test for breast cancer patients in an Australian multidisciplinary setting. Breast J 2022:1–7. https://doi.org/10.1155/2022/1199245
Pawloski KR, Gonen M, Wen HY et al (2022) Supervised machine learning model to predict oncotype DX risk category in patients over age 50. Breast Cancer Res Treat 191:423–430. https://doi.org/10.1007/S10549-021-06443-W
Kalinsky K, Barlow WE, Gralow JR et al (2021) 21-gene assay to inform chemotherapy benefit in node-positive breast cancer. N Engl J Med 385:2336–2347. https://doi.org/10.1056/NEJMOA2108873/SUPPL_FILE/NEJMOA2108873_DATA-SHARING.PDF
Ha R, Chang P, Mutasa S et al (2019) Convolutional neural network using a breast MRI tumor dataset can predict oncotype Dx recurrence score. J Magn Reson Imaging 49:518–524. https://doi.org/10.1002/JMRI.26244
Flanagan MB, Dabbs DJ, Brufsky AM et al (2008) Histopathologic variables predict oncotype DXTM recurrence score. Mod Pathol 21:1255–1261. https://doi.org/10.1038/modpathol.2008.54
Auerbach J, Kim M, Fineberg S (2010) Can features evaluated in the routine pathologic assessment of lymph node-negative estrogen receptor-positive stage I or II invasive breast cancer be used to predict the oncotype DX recurrence score? Arch Pathol Lab Med 134:1697–1701. https://doi.org/10.5858/2009-0439-OAR.1
Allison KH, Kandalaft PL, Sitlani CM et al (2012) Routine pathologic parameters can predict Oncotype DXTM recurrence scores in subsets of ER positive patients: who does not always need testing? Breast Cancer Res Treat 131:413–424. https://doi.org/10.1007/S10549-011-1416-3/TABLES/5
Mattes MD, Mann JM, Ashamalla H, Tejwani A (2013) Routine histopathologic characteristics can predict oncotype DXTM recurrence score in subsets of breast cancer patients. Cancer Investig 31:604–606. https://doi.org/10.3109/07357907.2013.849725
Gage MM, Rosman M, Mylander WC et al (2015) A validated model for identifying patients unlikely to benefit from the 21-gene recurrence score assay. Clin Breast Cancer 15:467–472. https://doi.org/10.1016/J.CLBC.2015.04.006
Turner BM, Skinner KA, Tang P et al (2015) Use of modified Magee equations and histologic criteria to predict the oncotype DX recurrence score. Mod Pathol 28:921–931. https://doi.org/10.1038/MODPATHOL.2015.50
Eaton AA, Pesce CE, Murphy JO et al (2017) Estimating the oncotype DX score: validation of an inexpensive estimation tool. Breast Cancer Res Treat 161:435–441. https://doi.org/10.1007/S10549-016-4069-4/TABLES/3
Orucevic A, Bell JL, King M et al (2019) Nomogram update based on TAILORx clinical trial results—oncotype DX breast cancer recurrence score can be predicted using clinicopathologic data. Breast 46:116–125. https://doi.org/10.1016/J.BREAST.2019.05.006
Lee SB, Kim J, Sohn G et al (2019) A nomogram for predicting the oncotype DX recurrence score in women with T1–3N0-1miM0 hormone receptor-positive, human epidermal growth factor 2 (HER2)-negative breast cancer. Cancer Res Treat 51:1073. https://doi.org/10.4143/CRT.2018.357
Yoo SH, Kim TY, Kim M et al (2020) Development of a nomogram to predict the recurrence score of 21-gene prediction assay in hormone receptor-positive early breast cancer. Clin Breast Cancer 20:98-107.e1. https://doi.org/10.1016/J.CLBC.2019.07.010
Batra A, Nixon NA, Roldan-Urgoiti G et al (2021) Develo** a clinical-pathologic model to predict genomic risk of recurrence in patients with hormone receptor positive, human epidermal growth factor receptor-2 negative, node negative breast cancer. Cancer Treat Res Commun 28:100401. https://doi.org/10.1016/J.CTARC.2021.100401
Marazzi F, Barone R, Masiello V et al (2020) Oncotype DX predictive nomogram for recurrence score output: the novel system ADAPTED01 based on quantitative immunochemistry analysis. Clin Breast Cancer 20:e600–e611. https://doi.org/10.1016/J.CLBC.2020.04.012
Acs G, Esposito NN, Kiluk J et al (2011) A mitotically active, cellular tumor stroma and/or inflammatory cells associated with tumor cells may contribute to intermediate or high oncotype DX recurrence scores in low-grade invasive breast carcinomas. Modern Pathol 25:556–566. https://doi.org/10.1038/modpathol.2011.194
Kalinsky KM, Barlow WE, Gralow JR et al (2022) Abstract GS2-07: Updated results from a phase 3 randomized clinical trial in participants (pts) with 1-3 positive lymph nodes (LN), hormone receptor-positive (HR+) and HER2-negative (HER2-) breast cancer (BC) with recurrence score (RS) ≤ 25 randomized to endocrine therapy (ET) +/- chemotherapy (CT): SWOG S1007 (RxPONDER). Can Res 82:GS2-07. https://doi.org/10.1158/1538-7445.SABCS21-GS2-07
Funding
This work was supported in part by NIH/NCI Cancer Center Support Grant No. P30CA008748 to Memorial Sloan Kettering Cancer Center. Editorial support in preparing this manuscript was provided by Hannah Rice, BA, ELS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Monica Morrow has received honoraria from Exact Sciences and Roche. All other authors have no conflict of interest disclosures to report.
Ethical approval
This study was approved by the Memorial Sloan Kettering Cancer Center Institutional Review Board.
Informed consent
Informed consent for this study was waived by the Memorial Sloan Kettering Cancer Center Institutional Review Board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Williams, A.D., Pawloski, K.R., Wen, H.Y. et al. Use of a supervised machine learning model to predict Oncotype DX risk category in node-positive patients older than 50 years of age. Breast Cancer Res Treat 196, 565–570 (2022). https://doi.org/10.1007/s10549-022-06763-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06763-5