Abstract
Purpose
To support shared decision-making, patient-facing resources are needed to complement recently published guidelines on approaches for surveillance mammography in breast cancer survivors aged ≥ 75 or with < 10-year life expectancy. We created a patient guide to facilitate discussions about surveillance mammography in older breast cancer survivors.
Methods
The “Are Mammograms Still Right for Me?” guide estimates future ipsilateral and contralateral breast (in-breast) cancer risks, general health, and the potential benefits/harms of mammography, with prompts for discussion. We conducted in-clinic acceptability testing of the guide by survivors and their clinicians at a National Cancer Institute-designated comprehensive cancer center, including two community practices. Patients and clinicians received the guide ahead of a clinic visit and surveyed patients (pre-/post-visit) and clinicians (post-visit). Acceptability was defined as ≥ 75% of patients and clinicians reporting that the guide (a) should be recommended to others, (b) is clear, (c) is helpful, and (d) contains a suitable amount of information. We also elicited feedback on usability and mammography intentions.
Results
We enrolled 45 patients and their 21 clinicians. Among those responding in post-visit surveys, 33/37 (89%) patients and 15/16 (94%) clinicians would recommend the guide to others; 33/37 (89%) patients and 15/16 (94%) clinicians felt everything/most things were clear. All other pre-specified acceptability criteria were met. Most patients reported strong intentions for mammography (100% pre-visit, 98% post-visit).
Conclusion
Oncology clinicians and older breast cancer survivors found a guide to inform mammography decision-making acceptable and clear. A multisite clinical trial is needed to assess the guide’s impact mammography utilization.
Trial registration: ClinicalTrials.gov-NCT03865654, posted March 7, 2019.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Develo** strategies to optimally support older breast cancer survivors is highly relevant to clinical practice, with more than 1.5 million breast cancer survivors aged ≥ 75 currently living in the U.S [1,2,3,4]. This number is anticipated to grow as the U.S. population ages and breast cancer mortality improves [1,2,3,4], yet guidelines for breast imaging in older breast cancer survivors [5] lack individualization or incorporation of life expectancy. Although screening mammography guidelines (for women without a history of breast cancer) recommend discontinuation of routine testing (i.e., in absence of symptoms or exam findings) when life expectancy is limited [6, 7], survivorship guidelines do not address how to tailor surveillance mammography by life expectancy [8, 9]. Thus, it is not surprising that conversations occur infrequently in clinical practice [9], with high utilization of surveillance mammography even when life expectancy is short [10].
We recently published expert consensus guidelines and talking points focusing on the older breast cancer survivor, with suggestions on how to approach surveillance mammography in the context of one’s life expectancy, age, the anticipated benefits and harms of testing, and patient preferences [9]. The guidelines acknowledge the long time-lag required to achieve minimal benefits in breast cancer-specific mortality from mammography[6, 11] and the more immediate harms of testing (e.g., false positives, overdiagnosis, over-treatment). As those guidelines were designed to support clinicians, they did not include patient-facing educational materials.
Engaging patients in patient-centered decision-making[12, 13] can ease exaggerated perceptions of cancer risk, provide reassurance, promote autonomy, and support clinicians by providing talking points for topics that may be uncomfortable, such as discontinuing a test to which patients/clinicians feel attached. Thus, shared decision-making has emerged as an effective approach for de-implementation of testing [14,15,16], particularly when benefit-to-harm ratios are uncertain or when benefits are outweighed by harms, such as the case of surveillance mammography for those with limited life expectancy. The value of shared decision-making in de-implementation has been demonstrated for screening mammography in primary care settings, facilitating informed decision-making, reduction in over-screening, improved patient knowledge, and satisfaction [16,17,18,19,20,21,22,23,24,25,26,27].
Recognizing the lack of decision support for older breast cancer survivors and their clinicians around surveillance mammograms, we created a patient-facing guide to complement the expert consensus guidelines [9]. Herein, we present results from acceptability testing of the “Are Mammograms Still Right for Me?” guide.
Materials and methods
Guide creation
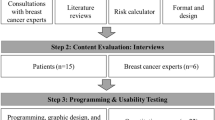
We first obtained broad feedback on the guide’s content from multidisciplinary clinicians and patient advocates serving on our expert consensus panel and from primary care and oncology clinicians participating in five focus groups during guideline development [9]. In collaboration with the Dana-Farber Cancer Institute (DFCI) Health Communications Core, we then created a four-page “Are Mammograms Still Right for Me?” informational pamphlet (also available as a PDF file; shown in its final, revised form in Supplemental File S1), which applied the fundamental theory from the Ottawa Decision Framework[28] and which adapted a decision aid developed for screening mammography [16, 19, 26, 27].
The guide was prepared for an 8th grade reading level (per Flesch-Kincaid literacy criteria) [29], providing information specific for older breast cancer survivors on: (a) why mammography is a decision, (b) estimations of ipsilateral and contralateral breast cancer risk, including risk for recurrences or new primary cancers (i.e., ‘in-breast’ cancer) risks, (c) potential benefits/harms of mammography, (d) how overall health impacts surveillance mammography’s benefits [9], and (e) discussion prompts for ‘what’s important to you?’, encouraging patients to use the guide to deliberate with clinicians. The guide has bulleted text and short sentences to enhance readability, uses pie charts to display the risks for in-breast recurrences and new primary cancer events, and aims to provide a balanced list of the benefits/harms of mammography. The guide’s estimation of in-breast cancer risks is derived from a comprehensive literature review and is based on one’s personal cancer and treatment history [8, 9].
In preparation for in-clinic acceptability testing, we conducted fifteen semi-structured telephone interviews with breast cancer survivors aged ≥ 75. We elicited input on the guide’s content and clarity, while also eliciting intentions (or plans) for mammography and conversations about life expectancy (since one’s life expectancy informs high-quality mammography decision-making). Overall, participants provided affirmative feedback on the guide’s format, length, and content. Because there were no consistent concerns identified, the guide was not modified before in-clinic testing. This research was approved by the DFCI Office of Human Research. Informed consent was obtained from all individual participants included in the study.
In-clinic acceptability testing—approach
We included breast cancer survivors who were aged ≥ 75 (where discontinuation of mammography may be appropriate given median U.S. life expectancy of ~ 10 years at age 75) and who received care at DFCI (Boston, MA) or two community-based satellite practices (Weymouth and Brighton, both in MA). All patients had to have completed active treatment(s) for their breast cancer (endocrine therapy allowed) and were required to read/speak English. We conducted feasibility testing in oncology practices because in prior focus groups of oncology and primary care clinicians, we learned that primary care clinicians defer to oncologists for decision-making around mammography [9].
Trained clinical research coordinators (CRCs) scanned clinic schedules for the upcoming four to six weeks to identify potentially eligible patients. Because we enrolled during the COVID-19 pandemic, we allowed virtual visits. Visit clinicians (physicians, nurse practitioners, physician assistants) were notified about plans to approach each patient and were allowed to opt out (no clinicians opted out). The CRC then contacted the patient to explain the study’s purpose and obtain verbal consent. Once enrolled, patients were mailed or emailed the guide; visit clinicians were notified.
The day before the visit, the CRC reminded the patient and clinician about the visit, encouraged them to use the guide during the visit, and administered the pre-visit survey to the patient. The survey was adapted from those used in mammography screening settings[16] and included questions about mammography intentions [19, 30], decisional conflict [31,32,33], demographics, numeracy [34], and health literacy [35], using validated scales and definitions (eTable 1). In addition, because the patient guide was designed to complement expert consensus approaches to mammography that emphasize considerations of life expectancy, we surveyed patients’ comfort level and preferences regarding life expectancy discussions. After the clinic visit, the CRC administered the post-visit survey within a week (with up to three reminders) and provided a $40 gift card, concluding study participation. The patient post-visit survey included items similar to pre-visit surveys plus acceptability questions about the guide (discussed below). We surveyed all clinicians via email following each visit, asking questions on guide acceptability, intentions (or plans) for mammography for that patient, and comfort with life expectancy discussions. Participating clinicians received a one-time $25 gift card at end-of-study. All patient and clinician surveys were administered via REDCap.
In-clinic acceptability testing—analyses
Our primary endpoint was guide acceptability, defined as ≥ 75% of unique patients and clinicians reporting each of the following responses in post-visit surveys: (a) would recommend use by others, (b) clear or mostly clear in its explanations, (c) helpful, and (d) with a suitable amount of information (right amount, a little more, or a little less information than needed). We tabulated responses to these questions on post-visit patient and clinician surveys; all analyses for the primary endpoint were descriptive.
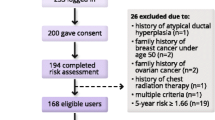
For clinician acceptability, we examined responses to each relevant question by clinician (up to 19 submitted surveys, designated as clinicians A-S in Fig. 1). If a clinician stated that they did not use the guide with one patient but then used it with a second patient and rated it favorably, we counted the response provided for the second patient towards acceptability. Clinicians who never used the guide (e.g., clinician D) were not included in the denominator assessing acceptability.
Pre-visit patient surveys collected demographics, health literacy, numeracy, and preferences for decision-making roles. Post-visit surveys included general guide feedback (e.g., length, clarity). Both surveys inquired about intentions for mammography plus past and current (study visit) experiences and comfort level with life expectancy discussions.
From post-visit clinician surveys, in addition to acceptability, we described clinician responses for each participating patient for questions that were relevant to a particular patient (e.g., “What did you recommend in terms of mammograms for this patient?”, “Did you discuss the pros [and cons] of mammography”). In addition to descriptive analyses and extraction of explanatory comments (when provided), we used t-tests to examine changes in patient decisional conflict around mammography from the pre- and post-survey to inform design of a future trial of the guide. All analyses were conducted using SAS.
Results
Among 88 patients approached between August 18, 2019 and May 11, 2020, 18 declined enrollment, 22 could not be reached, three were ineligible, and 45 women enrolled; no clinicians opted out for their patients. The 45 patient participants (ages 75–92, median = 78 years) had clinic visits with 21 unique oncology clinicians (six nurse practitioners, one physician assistant, 14 physicians); participating clinicians had one to seven patients who enrolled. Of the 45 enrolled patients, 40 completed at least some of the post-visit survey (three could not be reached, one rescheduled her visit twice and never completed the survey, one declined). Among 21 clinicians, 19 completed at least one survey. Patient characteristics are summarized in Table 1; 43 (96%) of patients reported White race, five (11%) reported a high school degree as their highest education, and two (4%) reported feeling extremely good about working with fractions, reflecting high numeracy. One woman reported ever having conversations about life expectancy, although 19 (42%) expressed interest in knowing this information, particularly if life expectancy was < one year. Extreme worry regarding new breast cancers was infrequent.
Acceptability
Acceptability of the guide by patients (Table 2) and clinicians (Fig. 1) was high; overall, clinicians were consistent in post-visit responses across patients. Among patients and clinicians completing post-visit surveys, 33/37 (89%) patients and 15/16 (94%) clinicians stated they would recommend the guide to others; 33/37 (89%) patients and 15/16 (94%) clinicians reported that everything/most things were clear (including one clinician who answered ‘most things were clear’ for one patient and ‘some things were clear’ for another). In addition, 20 patients (54%) and 96% of clinicians reported that the amount of information provided was suitable. Finally, 84% of patients and 80% of clinicians reported the guide was helpful in making decisions about mammography. All of these responses met prespecified criteria for acceptability.
Additional clinician feedback (Table 3).
Overall, clinician feedback on the guide was positive, with 73% stating the length was ‘just right’ and only two clinicians preferred a format other than a printed pamphlet. Several clinicians provided written comments: “…the guide is absolutely wonderful”, and “It would be great to have this…readily available for…discussions with our older patients.” Only one clinician reported that the guide made the visit a ‘lot longer’: “I worry about the ability of busy clinicians to use it during their constrained visits.” Even when clinicians recommended that a participating patient continue mammography, open-ended comments acknowledged (a) the general appropriateness of these discussions, (b) the importance of patient preferences, and (c) when it may not be appropriate to have these discussions.
Clinicians did not use the guide in 22% of visits, with some stating that patients required additional testing (n = 3) or were recommended to continue mammography (n = 4) because of higher-than-average risk for future in-breast cancers, excellent life expectancy, or patient preferences: “My bias was against recommending mammograms…the guide helped us think clearly about pros/cons in a very healthy patient with history of bilateral breast cancers who is more reassured to continue mammograms.” Another clinician stated: “[T]here are many patients who you already know will want to continue due to high anxiety.” Overall, patient preferences were strong for mammography (see below); five clinicians reported that they would have recommended discontinuation of mammography before the visit, but only one clinician reported recommending discontinuation after the visit.
Overall, in 29/41 visits (71%), clinicians reported discussing the pros and cons of mammography and less commonly life expectancy (18/41 visits [44%]). Most clinicians found the guide helpful, and none reported uncomfortable conversations (though one perceived the patient as uncomfortable): “…This tool makes it easier to discuss comorbidity and life expectancy; less awkward”, and “…having a printed tool made the life expectancy discussions…comfortable…It made it less like I had some specific concern that I was bringing up; the discussions are more just a part of standard practice.” For those not discussing life expectancy, the most common reasons provided were “it didn’t come up” or the “patient was healthy, so I didn’t think it was important.”
Mammography intentions and additional patient feedback (Table 2 and eTable 2).
Intentions for mammography were very strong in patient pre- (100%) and post- (98%) visit surveys, with only one patient deciding to discontinue mammography after the visit (her clinician also recommended discontinuation). Patients had low decisional conflict on pre- and post-surveys, with mean total scores of 12.8 and 12.0, respectively (p = 0.79; eTable 2). Overall, in post-clinic surveys, 22 patients (56%) recalled discussing mammography’s benefits, eleven (29%) reported discussing mammography’s cons, and nine (23%) recalled life expectancy discussions (Table 2). Although one patient reported feeling ‘very anxious’, no patients reported increased anxiety after using the guide, and 54% of patients felt the guide prepared them ‘a great deal’ or ‘quite a bit’ to make a better decision on mammography.
Discussion
In acceptability testing, 45 women ages ≥ 75 with history of breast cancer and with strong mammography intentions and their oncology clinicians found an information guide on whether or not to continue mammography acceptable and clear. Overall, 89% of patients and 94% of clinicians stated that they would recommend the guide to others. Patients and clinicians also found the guide reasonable in length and suitable in content. Patients reported no increased anxiety after using the guide, and clinicians were enthusiastic to have the guide available for in-clinic use.
The high mammography utilization observed is consistent with prior observations [10], underscoring the importance of providing support and education to clinicians and patients in this context, likely through longitudinal discussions. Although five clinicians in our study stated they had intended to recommend discontinuing mammography before the visits, only one recommended discontinuation in the post-visit survey, perhaps due to strong patient preferences elicited during visits (though we did not ask why recommendations changed). Another important contributing factor is that 71% of patients in acceptability testing were ages 75–79, an age range where some will have > 10-year life expectancy and will be appropriately recommended to continue mammography [9]. Even when the timing is appropriate for discontinuation, stop** mammograms in survivors may be challenging because of worry related to one’s personal cancer history and the often ingrained, perceived benefits of mammograms, without tools to adequately improve patients’ understanding of the harms of testing.
It is thus not surprising that a one-time intervention with a patient guide may not be sufficient in itself to impact mammography decisions. Instead, this may require repeated discussions over time with engaged clinicians, including primary care clinicians, ideally starting a few years ahead of when discontinuation of mammography will be considered. The patient guide and clinician-facing expert consensus guidelines[9] were developed with this in mind, with the goal to provide reassuring information that promotes informed decision-making, all in the context of one’s life expectancy, preferences, and underlying risk for new cancers. In clinical testing, when the guide was utilized, clinicians reported it was useful in these discussions. However, future testing of a patient guide will include alternative approaches to conveying this information to patients to better accommodate time constraints in clinic.
This high utilization of mammography among older breast cancer survivors in our study also confirms the clinician feedback received during our expert consensus guideline development [9], where focus group clinicians acknowledged the prevailing, habitual continuation of mammograms and difficulties incorporating conversations about life expectancy [9], consistent with the infrequent conversations reported in our study. Interestingly, although nine patients recalled discussing life expectancy, eighteen clinician surveys reported addressing this topic during the visit. Moreover, while 70% of oncology clinicians’ responses indicated they reviewed the downsides of mammography, only 29% of patients perceived this topic was discussed, highlighting differing visit perceptions for clinicians and patients; future study could include observation of discussions to assess for shared decision-making.
To our knowledge, the “Are Mammograms Still Right for Me?” guide is the first resource for older breast cancer survivors to facilitate shared decision-making on mammography. Together with the clinician-facing guidelines and talking points[9], these materials provide much-needed, multifaceted support for patients and their clinicians, including multidisciplinary physicians and advanced practice providers, that informs decisions rather than reflexively and indefinitely promoting mammograms. Based on the constructive feedback we received from acceptability testing, we revised the guide to include a more numerically balanced list of the benefits and harms of mammography, citations for the approximations for in-breast cancer events, and more reassuring text that physical exams and diagnostic evaluations will continue even if routine mammography is discontinued (Supplemental File S1).
We recognize several study limitations. Our study was limited is size and did not include a control population, non-English speaking patients, or a sufficiently diverse sample with regard to race, ethnicity, and socio-demographic factors. In addition, we did not mandate use of the guide or evaluate life expectancy and could not guarantee that the timing for mammography discussions was appropriate. Also, we included only oncology clinicians; we plan to include primary care clinicians in future evaluations and disseminations of the guide. Despite these limitations, clinicians rated the guide favorably and we obtained important initial implementation experience, all providing preliminary results for use of the guide in clinical practice.
Our results for acceptability will inform larger-scale studies that better engage diverse patient populations across various clinical settings and test the implementation of the patient guide through a multi-level, shared decision-making intervention that incorporates longitudinal patient-, clinician-, and practice-facing elements. This guide has the potential to enhance clinical practice by facilitating individualized decision-making for surveillance mammography among older breast cancer survivors but will require further study in larger, diverse practice settings and populations, and in particular, those with more limited life expectancy who are in most need of discussions on discontinuation of testing. The ultimate goal of this work is to further assess impact, standardize practice, and disseminate these novel resources to the growing number of older patients (and their clinicians) who are in urgent need of tailored approaches to survivorship care. Offering multi-layered support to pragmatically individualize mammography with a reassuring and informative approach has the potential to facilitate de-implementation of routine mammography when it is unlikely to provide benefit and may cause harm.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to patient privacy and the limitations stipulated in patient consent.
References
Bluethmann SM, Mariotto AB, Rowland JH (2016) Anticipating the “Silver Tsunami”: prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiol Biomarkers Prev 25(7):1029–1036. https://doi.org/10.1158/1055-9965.EPI-16-0133
Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, Alfano CM, Jemal A, Kramer JL (2019) Siegel RL (2019) cancer treatment and survivorship statistics. CA Cancer J Clin 69(5):363–385. https://doi.org/10.3322/caac.21565
Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Female Breast Cancer. https://seer.cancer.gov/statfacts/html/breast.html (accessed 27 July 2021).
Smith BD, Smith GL, Hurria A, Hortobagyi GN, Buchholz TA (2009) Future of cancer incidence in the United States: burdens upon an aging, changing nation. J Clin Oncol 27(17):2758–2765. https://doi.org/10.1200/JCO.2008.20.8983
Runowicz CD, Leach CR, Henry NL, Henry KS, Mackey HT, Cowens-Alvarado RL, Cannady RS, Pratt-Chapman ML, Edge SB, Jacobs LA, Hurria A, Marks LB, LaMonte SJ, Warner E, Lyman GH, Ganz PA (2016) American cancer society/American society of clinical oncology breast cancer survivorship care guideline. J Clin Oncol 34(6):611–635. https://doi.org/10.1200/JCO.2015.64.3809
Walter LC, Schonberg MA (2014) Screening mammography in older women: a review. JAMA 311(13):1336–1347. https://doi.org/10.1001/jama.2014.2834
United States Preventative Services Task Force Final Recommendation Statement, Jan 11, 2016 https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/breast-cancer-screening.
Freedman RA, Keating NL, Partridge AH, Muss HB, Hurria A, Winer EP (2017) Surveillance mammography in older patients with breast cancer-can we ever stop? A review. JAMA Oncol 3(3):402–409. https://doi.org/10.1001/jamaoncol.2016.3931
Freedman RA, Minami CA, Winer EP, Morrow M, Smith AK, Walter LC, Sedrak MS, Gagnon H, Perilla-Glen A, Wildiers H, Wildes TM, Lichtman SM, Loh KP, Brain EGC, Ganschow PS, Hunt KK, Mayer DK, Ruddy KJ, Jagsi R, Lin NU, Canin B, LeStage BK, Revette AC, Schonberg MA, Keating NL (2021) Individualizing surveillance mammography for older patients after treatment for early-stage breast cancer: multidisciplinary expert panel and international society of geriatric oncology consensus statement. JAMA Oncol 7(4):609–615. https://doi.org/10.1001/jamaoncol.2020.7582
Freedman RA, Keating NL, Pace LE, Lii J, McCarthy EP, Schonberg MA (2017) Use of surveillance mammography among older breast cancer survivors by life expectancy. J Clin Oncol 35(27):3123–3130. https://doi.org/10.1200/JCO.2016.72.1209
Lee SJ, Boscardin WJ, Stijacic-Cenzer I, Conell-Price J, O’Brien S, Walter LC (2013) Time lag to benefit after screening for breast and colorectal cancer: meta-analysis of survival data from the United States, Sweden, United Kingdom, and Denmark. BMJ 346:e8441. https://doi.org/10.1136/bmj.e8441
Reuben DB, Tinetti ME (2012) Goal-oriented patient care–an alternative health outcomes paradigm. N Engl J Med 366(9):777–779. https://doi.org/10.1056/NEJMp1113631
Colla CH (2014) Swimming against the current–what might work to reduce low-value care? N Engl J Med 371(14):1280–1283. https://doi.org/10.1056/NEJMp1404503
Sypes EE, de Grood C, Whalen-Browne L, Clement FM, Parsons Leigh J, Niven DJ, Stelfox HT (2020) Engaging patients in de-implementation interventions to reduce low-value clinical care: a systematic review and meta-analysis. BMC Med 18(1):116. https://doi.org/10.1186/s12916-020-01567-0
Berlin NL, Skolarus TA, Kerr EA, Dossett LA (2020) Too much surgery: overcoming barriers to deimplementation of low-value surgery. Ann Surg 271(6):1020–1022. https://doi.org/10.1097/SLA.0000000000003792
Schonberg MA, Kistler CE, Pinheiro A, Jacobson AR, Aliberti GM, Karamourtopoulos M, Hayes M, Neville BA, Lewis CL, Wee CC, Fagerlin A, Nekhlyudov L, Marcantonio ER, Hamel MB, Davis RB (2020) Effect of a mammography screening decision aid for women 75 years and older: a cluster randomized clinical trial. JAMA Intern Med. https://doi.org/10.1001/jamainternmed.2020.0440
Schonberg MA, Karamourtopoulos M, Jacobson AR, Aliberti GM, Pinheiro A, Smith AK, Davis RB, Schuttner LC, Hamel MB (2020) A strategy to prepare primary care clinicians for discussing stop** cancer screening with adults older than 75 years. Innov Aging 4(4):igaa027. https://doi.org/10.1093/geroni/igaa027
Schonberg MA, Jacobson AR, Karamourtopoulos M, Aliberti GM, Pinheiro A, Smith AK, Schuttner LC, Park ER, Hamel MB (2020) Scripts and strategies for discussing stop** cancer screening with adults > 75 years: a qualitative study. J Gen Intern Med 35(7):2076–2083. https://doi.org/10.1007/s11606-020-05735-z
Schonberg MA, Hamel MB, Davis RB, Griggs MC, Wee CC, Fagerlin A, Marcantonio ER (2014) Development and evaluation of a decision aid on mammography screening for women 75 years and older. JAMA Intern Med 174(3):417–424. https://doi.org/10.1001/jamainternmed.2013.13639
Schonberg MA (2016) Decision-making regarding mammography screening for older women. J Am Geriatr Soc. https://doi.org/10.1111/jgs.14503
Schonberg MA, Silliman RA, Marcantonio ER (2009) Weighing the benefits and burdens of mammography screening among women age 80 years or older. J Clin Oncol 27(11):1774–1780. https://doi.org/10.1200/JCO.2008.19.9877
Schonberg MA, Breslau ES, McCarthy EP (2013) Targeting of mammography screening according to life expectancy in women aged 75 and older. J Am Geriatr Soc 61(3):388–395. https://doi.org/10.1111/jgs.12123
Schonberg MA, Walter LC (2013) Talking about stop** cancer screening-not so easy. JAMA Intern Med 173(7):532–533. https://doi.org/10.1001/jamainternmed.2013.3233
Schonberg MA, Breslau ES (2015) Mammography screening for women aged 70 and older: at a crossroads. J Am Geriatr Soc 63(1):170–172. https://doi.org/10.1111/jgs.13189
Schonberg MA, McCarthy EP, York M, Davis RB, Marcantonio ER (2007) Factors influencing elderly women’s mammography screening decisions: implications for counseling. BMC Geriatr 7:26. https://doi.org/10.1186/1471-2318-7-26
Schonberg MA, Kistler CE, Nekhlyudov L, Fagerlin A, Davis RB, Wee CC, Marcantonio ER, Lewis CL, Stanley WA, Crutchfield TM, Hamel MB (2014) Evaluation of a mammography screening decision aid for women aged 75 and older: protocol for a cluster-randomized controlled trial. J Clin Trials 4:191. https://doi.org/10.4172/2167-0870.1000191
Schonberg MA, Ramanan RA, McCarthy EP, Marcantonio ER (2006) Decision making and counseling around mammography screening for women aged 80 or older. J Gen Intern Med 21(9):979–985. https://doi.org/10.1111/j.1525-1497.2006.00487.x
The Ottawa Hospital. Patient Decision Aids. https://decisionaid.ohri.ca/models.html.
Kincaid JP, Fishburne RP, Rogers RL, Chissom BS (1975) Derivation of new readability formulas (automated readability index, fog count and flesch reading ease formula) for Navy enlisted personnel. Research Branch Report 8–75, Millington, TN: Naval Technical Training, U.S. Naval Air Station, Memphis, TN.
Diet, nutrition, physical activity and cancer: a global perspective (2018).
O’Connor AM. User manual- decisional conflict scale. https://decisionaid.ohri.ca/docs/develop/Tools/DCS_English.pdf.
O’Connor AM (1995) Validation of a decisional conflict scale. Med Decis Making 15(1):25–30. https://doi.org/10.1177/0272989X9501500105
O’Connor AM (1993) User manual: decisional conflict scale (16 item statement format) Ottawa Hospital Research Institute. https://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf (accessed 10 Sept 2021).
Fagerlin A, Zikmund-Fisher BJ, Ubel PA, Jankovic A, Derry HA, Smith DM (2007) Measuring numeracy without a math test: development of the Subjective Numeracy Scale. Med Decis Making 27(5):672–680. https://doi.org/10.1177/0272989X07304449
Chew LD, Griffin JM, Partin MR, Noorbaloochi S, Grill JP, Snyder A, Bradley KA, Nugent SM, Baines AD, Vanryn M (2008) Validation of screening questions for limited health literacy in a large VA outpatient population. J Gen Intern Med 23(5):561–566. https://doi.org/10.1007/s11606-008-0520-5
Degner LF, Kristjanson LJ, Bowman D, Sloan JA, Carriere KC, O’Neil J, Bilodeau B, Watson P, Mueller B (1997) Information needs and decisional preferences in women with breast cancer. JAMA 277(18):1485–1492
Health Information National Trends Survey (HINTS) 2003, 2005, and 2008. http://hints.cancer.gov/default.aspx
Andersen MR, Smith R, Meischke H, Bowen D, Urban N (2003) Breast cancer worry and mammography use by women with and without a family history in a population-based sample. Cancer Epidemiol Biomarkers Prev 12(4):314–320
Graham ID, O’Connor AM. User manual. https://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_PrepDM.pdf
Acknowledgements
We thank all the patients, clinicians, and expert consensus guideline panelists who participated in this work and who provided feedback on the patient guide. We also thank the Health Communications Core at Dana-Farber Cancer Institute who designed and collaborated on the development of the guide. We thank Kate Bifolck and Valerie Hope Goldstein for their assistance with manuscript preparation and submission.
Funding
National Cancer Institute (R21CA227615-01A1 to RAF), Susan G. Komen (CCRCR18552788 to RAF), American Cancer Society (MRSG-14–240-01-CPPB to RAF), National Institute on Aging (K24AG071906 to MAS).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Rachel Freedman, Anna Revette, Nancy Keating, and Mara Schonberg. The first draft of the manuscript was written by Rachel Freedman and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Nancy U. Lin reports institutional research funding from Genentech, Merck, Pfizer, Seattle Genetics, AstraZeneca, Zion Pharmaceuticals, and Olema Pharmaceuticals; consultant/advisory board work for Pfizer, Puma, Seattle Genetics, Daiichi Sankyo, AstraZeneca, Prelude Therapeutics, Denali Therapeutics, Olema Pharmaceuticals, Aleta BioPharma, Affinia Therapeutics, Voyager Therapeutics, and Artera, Inc.; royalties from UpToDate; and stock or other ownership interests in Artera Inc. (a startup with no current value, but options only valued at < 5% and < $50,000 will be provided at a later date). The remaining authors declare that they have no conflicts of interest.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki and was approved by the DFCI Office of Human Research. Informed consent was obtained from all individual participants included in the study.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Nancy L. Keating and Mara A. Schonberg Co-senior authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Freedman, R.A., Revette, A.C., Gagnon, H. et al. Acceptability of a companion patient guide to support expert consensus guidelines on surveillance mammography in older breast cancer survivors. Breast Cancer Res Treat 195, 141–152 (2022). https://doi.org/10.1007/s10549-022-06676-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06676-3