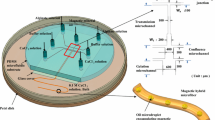
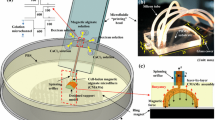
Abstract
The storage of living organisms in alginate gel fibers of various shapes is critical for various applications, and has, therefore, attracted research attention in the field of regenerative medicine. This paper presents a report on the on-demand formation of heterogeneous alginate gel fibers using a microfluidic device with narrowly spaced, two-dimensional array of microapertures. The device was fabricated by bonding stainless-steel substrates that formed an array of 48 microapertures individually connected to microchannels. This configuration allowed for the independent control of fluid injected from the microapertures at a narrow spacing of 50 μm, and the formation of gel fibers of various shapes and dimensions with a single device. Using this stainless device, a single-component type gel was formed by setting one aperture for Na-alginate injection at the center of the aperture array; the surrounding apertures were set for CaCl2 injection; the fiber diameter ranged from 8 to 28 μm. In addition, two-component, staggered-grid, hollow, and asymmetric gel fibers were formed by designing apertures for fluid injection. In the staggered-grid type formation, the cross-sectional area of the fiber was controlled between 6.0 and 12.0 × 104 μm2 by changing the CaCl2 injection flow rate. This adaptability to the formation of various two-dimensional heterogeneous gel fibers with a single device can promote the development of three-dimensional cultures and regenerative medicine based on the gel fiber technique.








Similar content being viewed by others
References
Bidarra SJ, Barrias CC (2019) 3D culture of mesenchymal stem cells in alginate hydrogels. Methods Mol Biol 2002:165–180. https://doi.org/10.1007/7651_2018_185
Bsoul A, Beyer S, Ahmadi A, et al (2013) Molded biocompatible and disposable PDMS/SU-8 inkjet dispenser, vol 1. In: 17th international conference on miniaturized systems for chemistry and life sciences, MicroTAS 2013, p 636–638
Cheng Y, Zheng F, Lu J et al (2014) Bioinspired multicompartmental microfibers from microfluidics. Adv Mater 26:5184–5190. https://doi.org/10.1002/adma.201400798
Colosi C, Costantini M, Latini R et al (2014) Rapid prototy** of chitosan-coated alginate scaffolds through the use of a 3D fiber deposition technique. J Mater Chem B 2:6779–6791. https://doi.org/10.1039/c4tb00732h
Ghorbanian S, Qasaimeh MA, Akbari M et al (2014) Microfluidic direct writer with integrated declogging mechanism for fabricating cell-laden hydrogel constructs. Biomed Microdevices 16:387–395. https://doi.org/10.1007/s10544-014-9842-8
Kang E, Jeong GS, Choi YY et al (2011) Digitally tunable physicochemical coding of material composition and topography in continuous microfibres. Nat Mater 10:877–883. https://doi.org/10.1038/nmat3108
Kang E, Choi YY, Chae SK et al (2012) Microfluidic spinning of flat alginate fibers with grooves for cell-aligning scaffolds. Adv Mater 24:4271–4277. https://doi.org/10.1002/adma.201201232
Kobayashi A, Yamakoshi K, Yajima Y et al (2013) Preparation of stripe-patterned heterogeneous hydrogel sheets using microfluidic devices for high-density coculture of hepatocytes and fibroblasts. J Biosci Bioeng 116:761–767. https://doi.org/10.1016/j.jbiosc.2013.05.034
Lee KY, Mooney DJ (2012) Alginate: properties and biomedical applications. Prog Polym Sci 37:106–126. https://doi.org/10.1016/j.progpolymsci.2011.06.003
Lee KH, Asthana A, Shin SJ et al (2009a) Enzyme loaded alginate hollow fiber for reactor. In: Proceedings of conference. Micro TAS 2009—the 13th international conference on miniaturized systems for chemistry and life sciences, p 645–657
Lee KH, Shin SJ, Park Y, Lee SH (2009b) Synthesis of cell-laden alginate hollow fibers using microfluidic chips and microvascularized tissue-engineering applications. Small 5:1264–1268. https://doi.org/10.1002/smll.200801667
Lin H, Li Q, Wang O et al (2018) Automated expansion of primary human T cells in scalable and cell-friendly hydrogel microtubes for adoptive immunotherapy. Adv Healthc Mater 7:e1701297. https://doi.org/10.1002/adhm.201701297,29749707
Liu Q, Li Q, Xu S et al (2018) Preparation and properties of 3D printed alginate–chitosan polyion complex hydrogels for tissue engineering. Polymers 10:664. https://doi.org/10.3390/polym10060664
Lu B, Li M, Fang Y et al (2020) Rapid fabrication of cell-laden microfibers for construction of aligned biomimetic tissue. Front Bioeng Biotechnol 8:610249. https://doi.org/10.3389/fbioe.2020.610249
Mulhall HJ, Hughes MP, Kazmi B et al (2013) Epithelial cancer cells exhibit different electrical properties when cultured in 2D and 3-D environments. Biochim Biophys Acta 1830:5136–5141. https://doi.org/10.1016/j.bbagen.2013.07.008
Nakazawa K (2013) Effects of culture conditions on a micropatterned co-culture of rat hepatocytes with 3T3 cells. J Bioproces Biotech. https://doi.org/10.4172/2155-9821.S3-002
Nusterer M, Rauch J, Viljoen H (2022) Theoretical and experimental investigation of alginate microtube extrusion for cell culture applications. Biochem Eng J. https://doi.org/10.1016/j.bej.2021.108236
Onoe H, Okitsu T, Itou A et al (2013) Metre-long cell-laden microfibres exhibit tissue morphologies and functions. Nat Mater 12:584–590. https://doi.org/10.1038/nmat3606
Pendyala G, Bithi SS, Vanapalli SA, Fernandes GE (2019) Continuous and high throughput production of alginate fibers using co-flow in a millifluidic T-junction. J Appl Polym Sci 136:47120. https://doi.org/10.1002/app.47120
Shima A, Itou A, Takeuchi S (2020) Cell fibers promote proliferation of co-cultured cells on a dish. Sci Rep 10:288. https://doi.org/10.1038/s41598-019-57213-0
Shin SJ, Park JY, Lee JY et al (2007) “On the fly” continuous generation of alginate fibers using a microfluidic device. Langmuir 23:9104–9108. https://doi.org/10.1021/la700818q
Takahashi K, Kamiya S, Takao H et al (2021) Stainless microfluidic probe with 2D-array microapertures. AIP Adv. https://doi.org/10.1063/5.0014119
Weigelt B, Lo AT, Park CC et al (2010) HER2 signaling pathway activation and response of breast cancer cells to HER2-targeting agents is dependent strongly on the 3D microenvironment. Breast Cancer Res Treat 122:35–43. https://doi.org/10.1007/s10549-009-0502-2
Wu Z, Su X, Xu Y et al (2016) Bioprinting three-dimensional cell-laden tissue constructs with controllable degradation. Sci Rep 6:24474. https://doi.org/10.1038/srep24474
Yu Y, Wei W, Wang Y et al (2016) Simple spinning of heterogeneous hollow microfibers on chip. Adv Mater 28:6649–6655. https://doi.org/10.1002/adma.201601504
Zhang YS, Pi Q, van Genderen AM (2017) Microfluidic bioprinting for engineering vascularized tissues and organoids. J vis Exp Jove 126:e55957. https://doi.org/10.3791/55957
Zhang X, Weng L, Liu Q et al (2019) Facile fabrication and characterization on alginate microfibres with grooved structure via microfluidic spinning. R Soc Open Sci 6:181928. https://doi.org/10.1098/rsos.181928
Acknowledgements
This work was supported by JST-PRESTO (grant number JPMJPR14FB), JST-FOREST (grant number JPMJFR212D) and JSPS KAKENHI (16H06077, 20K21900). The work was partly conducted at the Kagawa University Nano-Processing Facility, supported by the “Nanotechnology Platform Program” of the MEXT.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Takahashi, K., Takao, H., Shimokawa, F. et al. On-demand formation of heterogeneous gel fibers using two-dimensional micronozzle array. Microfluid Nanofluid 26, 15 (2022). https://doi.org/10.1007/s10404-022-02525-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-022-02525-3