Abstract
Purpose
To evaluate the efficacy of aprepitant and its effect on the quality of life when added to standard antiemetic therapy as salvage therapy.
Patients and methods
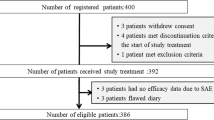
This is an open-label, noncomparative prospective phase II clinical trial. A total of 224 patients receiving initial moderately emetogenic chemotherapy (MEC) were enrolled. Patients received standard antiemetic treatment (5-hydroxytryptamine (5-HT3) antagonists + dexamethasone) in the first cycle. Patients failing to achieve a complete response (CR) during cycle 1 entered cycle 2 with the addition of aprepitant as salvage treatment.
Results
Of the 224 patients eligible for cycle 1, 159 (71.0%, 95% CI 65.0–77.0) had a CR, and 65 patients with a noncomplete response (NCR) proceeded to cycle 2 to receive aprepitant-based salvage therapy. Then, 49 (75.4%, 95% CI 64.6–84.1) of these patients achieved a CR. The Functional Living Index-Emesis (FLIE) questionnaire showed that NCR patients in cycle 1 had an improved quality of life after receiving aprepitant-based salvage therapy in cycle 2 (cycle 1 and 2: 80.3 vs 114.0, P < 0.001). Failing to achieve a CR had a significant impact on the quality of life in both cycle 1 (FLIE score of NCR and CR patients: 80.3 vs 119.7, P < 0.001) and cycle 2 (FLIE score of NCR and CR patients: 88.3 vs 122.4, P < 0.001).
Conclusion
The addition of aprepitant as salvage therapy for patients with gastrointestinal cancer is effective and has a positive effect on quality of life.






Similar content being viewed by others
References
Glaus A, Knip** C, Morant R et al (2004) Chemotherapy-induced nausea and vomiting in routine practice: a European perspective. Support Care Cancer 12(10):708–715
Schnell FM (2003) Chemotherapy-induced nausea and vomiting: the importance of acute antiemetic control. Oncologist 8(2):187–198
National Comprehensive Cancer Networks (2019) NCCN Clinical practice guidelines in oncology: colon cancer (Version 2.2019). https://www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed 15 May 2019
National Comprehensive Cancer Networks (2019) NCCN Clinical Practice Guidelines in oncology: rectal Cancer (Version 2.2019). https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf. Accessed 15 May 2019
National Comprehensive Cancer Networks (2019) NCCN Clinical Practice Guidelines in Oncology: Gastric Cancer (Version 2.2019).https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Accessed 3 June 2019
National Comprehensive Cancer Networks (2020) NCCN Clinical Practice Guidelines in Oncology: Antiemesis(Version1.2020).https://www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf. Accessed 19 Feb 2020
Hesketh PJ, Kris MG, Basch E et al (2017) Antiemetics: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol 35(28):3240–3261
Einhorn LH, Rapoport B, Navari RM et al (2017) 2016 updated MASCC/ESMO consensus recommendations: prevention of nausea and vomiting following multiple-day chemotherapy, high-dose chemotherapy, and breakthrough nausea and vomiting. Support Care Cancer 25(1):303–308
Navari RM (2015) Treatment of breakthrough and refractory chemotherapy-induced nausea and vomiting. Biomed Res Int, pp 1–6.
Aapro MS, Schmoll HJ, Jahn F et al (2013) Review of the efficacy of aprepitant for the prevention of chemotherapy-induced nausea and vomiting in a range of tumor types. Cancer Treat Rev 39(1):113–117
Lindley CM, Hirsch JD, O’Neill CV et al (1992) Quality of life consequences of chemotherapy-induced emesis. Qual Life Res 1(5):331–340
Yeo W, Mo FKF, Suen JJS et al (2009) A randomized study of aprepitant, ondansetron and dexamethasone for chemotherapy-induced nausea and vomiting in Chinese breast cancer patients receiving moderately emetogenic chemotherapy. Breast Cancer Res Treat 113(3):529–535
Martin AR, Pearson JD, Cai B et al (2003) Assessing the impact of chemotherapy-induced nausea and vomiting on patients’ daily lives: a modified version of the Functional Living Index-Emesis (FLIE) with 5-day recall. Support Care Cancer 11(8):522–527
Rapoport BL, Jordan K, Boice JA et al (2010) Aprepitant for the prevention of chemotherapy-induced nausea and vomiting associated with a broad range of moderately emetogenic chemotherapies and tumor types: a randomized, double-blind study. Support Care Cancer 18(4):423–431
Hesketh PJ, Wright O, Rosati G et al (2012) Single-dose intravenous casopitant in combination with ondansetron and dexamethasone for the prevention of oxaliplatin-induced nausea and vomiting: a multicenter, randomized, double-blind, active-controlled, two arm, parallel group study. Support Care Cancer 20(7):1471–1478
Warr DG, Hesketh PJ, Gralla RJ et al (2005) Efficacy and tolerability of aprepitant for the prevention of chemotherapy-induced nausea and vomiting in patients with breast cancer after moderately emetogenic chemotherapy. J Clin Oncol 23(12):2822–2830
Navari RM, Nagy CK, Gray SE (2013) The use of olanzapine versus metoclopramide for the treatment of breakthrough chemotherapy-induced nausea and vomiting in patients receiving highly emetogenic chemotherapy. Support Care Cancer. 21(6):1655–1663
Yanai T, Iwasa S, Hashimoto H et al (2018) A double-blind randomized phase II dose-finding study of olanzapine 10 mg or 5 mg for the prophylaxis of emesis induced by highly emetogenic cisplatin-based chemotherapy. Int J Clin Oncol 23(2):382–388
Tan L, Liu J, Liu X et al (2009) Clinical research of olanzapine for prevention of chemotherapy-induced nausea and vomiting. J Exp Clin Cancer Res 28(1):131
Llombart-Cussac A, Ramos M, Dalmau E et al (2016) Incidence of chemotherapy-induced nausea and vomiting associated with docetaxel and cyclophosphamide in early breast cancer patients and aprepitant efficacy as salvage therapy. Results from the Spanish Breast Cancer Group/2009–02 study. Eur J Cancer. 58:122–129
Inoue T, Kimura M, Uchida J et al (2017) Aprepitant for the treatment of breakthrough chemotherapy-induced nausea and vomiting in patients receiving moderately emetogenic chemotherapy. Int J Clin Oncol 22(3):600–604
Wu CE, Liaw CC (2012) Using aprepitant as secondary antiemetic prophylaxis for cancer patients with cisplatin-induced emesis. Support Care Cancer 20(10):2357–2361
Hesketh PJ, Younger J, Sanz-Altamira P et al (2009) Aprepitant as salvage antiemetic therapy in breast cancer patients receiving doxorubicin and cyclophosphamide. Support Care Cancer 17(8):1065–1070
Nishimura J, Satoh T, Fukunaga M et al (2015) Combination antiemetic therapy with aprepitant/fosaprepitant in patients with colorectal cancer receiving oxaliplatin-based chemotherapy (SENRI trial): a multicentre, randomised, controlled phase 3 trial. Eur J Cancer 51(10):1274–1282
Navari RM, Schwartzberg LS (2018) Evolving role of neurokinin 1-receptor antagonists for chemotherapy-induced nausea and vomiting. OncoTargets Therapy 11:6459–6478
Yahata H, Kobayashi H, Sonoda K et al (2016) Efficacy of aprepitant for the prevention of chemotherapy-induced nausea and vomiting with a moderately emetogenic chemotherapy regimen: a multicenter, placebo-controlled, double-blind, randomized study in patients with gynecologic cancer receiving paclitaxel and carboplatin. Int J Clin Oncol 21(3):491–497
Martin AR, Carides AD, Pearson JD et al (2003) Functional relevance of antiemetic control: experience using the FLIE questionnaire in a randomized study of the NK-1 antagonist aprepitant. Eur J Cancer 39(10):1395–1401
du Bois A, Meerpohl HG, Vach W et al (1992) Course, patterns, and risk-factors for chemotherapy-induced emesis in cisplatin pretreated patients: a study with ondansetron. Eur J Cancer 28(2–3):450–457
Sekine I, Segawa Y, Kubota K et al (2013) Risk factors of chemotherapy induced nausea and vomiting: index for personalized antiemetic prophylaxis. Cancer Sci 104(6):711–717
Acknowledgements
This study was supported by the Chinese Society of Clinical Oncology. We thank Miss Yan Zhou for assistance in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
The authors declare that they have no conflicts of interest associated with this work. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Gong, Q., Tian, J., Jiang, Y. et al. The efficacy of aprepitant in salvage treatment of chemotherapy-induced nausea and vomiting associated with moderately emetogenic chemotherapy for patients with gastrointestinal cancer. Int J Clin Oncol 26, 1091–1098 (2021). https://doi.org/10.1007/s10147-021-01898-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-021-01898-9