Abstract
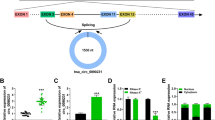
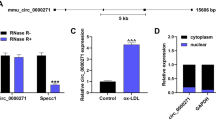
This study was designed to investigate the role of a disintegrin and metalloproteinase domain-like protein decysin 1 (ADAMDEC-1) in atherosclerosis (AS). The Gene Expression Omnibus (GEO) database was utilized to identify differentially expressed genes (DEGs) between carotid atheroma plaque and carotid tissue adjacent atheroma plaque obtained from AS patients. Gene functional enrichment analysis was conducted on DEGs using Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG). QRT-PCR was employed to quantify mRNAs expression. AS animal model was established using ApoE-/- mice; serum triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C) levels were detected. Aortic sinus atherosclerotic lesions were observed using H&E staining and Oil Red O staining. ADAMDEC-1 was silenced using small interfering RNAs (siRNAs) in human vascular smooth muscle cells (HVSMCs). Cell proliferation, migration, and cell cycle progression were detected by cell count kit-8 (CCK8), 5-ethynyl-2′-deoxyuridine (EDU), wound scratch healing assay, transwell assay, and flow cytometry, respectively. Western blot was used to evaluate various protein expression levels. Our results showed that ADAMDEC-1 was highly expressed in the serum of AS patients, consistent with the in silico results. The elevated TG, LDL-C, and HDL-C levels along with H&E and Oil Red O staining confirmed the successful establishment of the AS mouse model. ADAMDEC-1 expression was also elevated in AS mice. ADAMDEC-1 knockdown in HVSMCs suppressed cell proliferation, inhibited the expression of proliferating cell nuclear antigen (PCNA), and reduced the levels of matrix metalloproteinases (MMP2 and MMP9) proteins. Protein-protein interaction (PPI) analysis indicated that ADAMDEC-1 was associated with CXCL9, CCR5, TNF-α, TNFR1, and NF-κB-p50. The expression levels of CXCL9, CCR5, TNF-α, TNFR1, and NF-κB-p50 increased, while ADAMDEC-1 knockdown attenuated the expression of these proteins. Our study findings substantiate that ADAMDEC-1 may represent a novel target for AS.







Similar content being viewed by others
Data availability
The dataset used and/or analyzed in this study is available from the corresponding author on reasonable request.
References
Alfandari D, Taneyhill LA (2018) Cut loose and run: the complex role of ADAM proteases during neural crest cell development. Genesis (New York, NY: 2000) 56:e23095. https://doi.org/10.1002/dvg.23095
Chen L, Zhou Z, Hu C et al (2022) Platelet membrane-coated nanocarriers targeting plaques to deliver anti-CD47 antibody for atherosclerotic therapy. Research (Washington, DC) 2022:9845459. https://doi.org/10.34133/2022/9845459
Chen R, ** G, McIntyre TM (2017) The soluble protease ADAMDEC1 released from activated platelets hydrolyzes platelet membrane pro-epidermal growth factor (EGF) to active high-molecular-weight EGF. J Biol Chem 292:10112–10122. https://doi.org/10.1074/jbc.M116.771642
Chen Z, **e X, Jiang N et al (2021) CCR5 signaling promotes lipopolysaccharide-induced macrophage recruitment and alveolar developmental arrest. Cell Death Dis 12:184. https://doi.org/10.1038/s41419-021-03464-7
Couchie D, Vaisman B, Abderrazak A et al (2017) Human plasma thioredoxin-80 increases with age and in ApoE(-/-) mice induces inflammation, angiogenesis, and atherosclerosis. Circulation 136:464–475. https://doi.org/10.1161/circulationaha.117.027612
Crouser ED, Culver DA, Knox KS et al (2009) Gene expression profiling identifies MMP-12 and ADAMDEC1 as potential pathogenic mediators of pulmonary sarcoidosis. Am J Respir Crit Care Med 179:929–938. https://doi.org/10.1164/rccm.200803-490OC
Deng X, Zhang X, Tang B et al (2018) Design, synthesis, and evaluation of dihydrobenzo[cd]indole-6-sulfonamide as TNF-α inhibitors. Front Chem 6:98. https://doi.org/10.3389/fchem.2018.00098
Gareus R, Kotsaki E, Xanthoulea S et al (2008) Endothelial cell-specific NF-kappaB inhibition protects mice from atherosclerosis. Cell Metab 8:372–383. https://doi.org/10.1016/j.cmet.2008.08.016
Geng YJ (2001) Biologic effect and molecular regulation of vascular apoptosis in atherosclerosis. Curr Atheroscler Rep 3:234–242. https://doi.org/10.1007/s11883-001-0066-z
Ha SE, Jorgensen BG, Wei L et al (2022) Metalloendopeptidase ADAM-like decysin 1 (ADAMDEC1) in colonic subepithelial PDGFRα(+) cells is a new marker for inflammatory bowel disease. Int J Mol Sci 23. https://doi.org/10.3390/ijms23095007
Hendricks WPD, Briones N, Halperin RF et al (2019) PD-1-associated gene expression signature of neoadjuvant trastuzumab-treated tumors correlates with patient survival in HER2-positive breast cancer. Cancers 11. https://doi.org/10.3390/cancers11101566
Ji N, Wang Y, Gong X et al (2021) CircMTO1 inhibits ox-LDL-stimulated vascular smooth muscle cell proliferation and migration via regulating the miR-182-5p/RASA1 axis. Mol Med (Cambridge, Mass) 27:73. https://doi.org/10.1186/s10020-021-00330-2
Jimenez-Pascual A, Hale JS, Kordowski A et al (2019) ADAMDEC1 maintains a growth factor signaling loop in cancer stem cells. Cancer Discov 9:1574–1589. https://doi.org/10.1158/2159-8290.cd-18-1308
Kuniyoshi N, Imai H, Kiso Y et al (2021) Biological potentials for a family of disintegrin and metalloproteinase (ADAMDEC)-1 in mouse normal pregnancy. J Vet Med Sci 83:512–521. https://doi.org/10.1292/jvms.20-0570
Li W, Gonzalez KM, Chung J et al (2022) Surface-modified nanotherapeutics targeting atherosclerosis. Biomater Sci 10:5459–5471. https://doi.org/10.1039/d2bm00660j
Liu D, Wang X, Zhang M et al (2020) WISP1 alleviates lipid deposition in macrophages via the PPARγ/CD36 pathway in the plaque formation of atherosclerosis. J Cell Mol Med 24:11729–11741. https://doi.org/10.1111/jcmm.15783
Lund J, Troeberg L, Kjeldal H et al (2015) Evidence for restricted reactivity of ADAMDEC1 with protein substrates and endogenous inhibitors. J Biol Chem 290:6620–6629. https://doi.org/10.1074/jbc.M114.601724
Moldovan GL, Pfander B, Jentsch S (2007) PCNA, the maestro of the replication fork. Cell 129:665–679. https://doi.org/10.1016/j.cell.2007.05.003
Nakashima Y, Plump AS, Raines EW et al (1994) ApoE-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arterioscler Thromb 14:133–140. https://doi.org/10.1161/01.atv.14.1.133
Nasiri-Ansari Ν, Dimitriadis GK, Agrogiannis G et al (2018) Canagliflozin attenuates the progression of atherosclerosis and inflammation process in APOE knockout mice. Cardiovasc Diabetol 17:106. https://doi.org/10.1186/s12933-018-0749-1
Oh BY, Cho J, Hong HK et al (2017) Exome and transcriptome sequencing identifies loss of PDLIM2 in metastatic colorectal cancers. Cancer Manag Res 9:581–589. https://doi.org/10.2147/cmar.s149002
Papaspyridonos M, Smith A, Burnand KG et al (2006) Novel candidate genes in unstable areas of human atherosclerotic plaques. Arterioscler Thromb Vasc Biol 26:1837–1844. https://doi.org/10.1161/01.atv.0000229695.68416.76
Patel AP, Natarajan P (2019) Completing the genetic spectrum influencing coronary artery disease: from germline to somatic variation. Cardiovasc Res 115:830–843. https://doi.org/10.1093/cvr/cvz032
Pateras I, Giaginis C, Tsigris C et al (2014) NF-κB signaling at the crossroads of inflammation and atherogenesis: searching for new therapeutic links. Expert Opin Ther Targets 18:1089–1101. https://doi.org/10.1517/14728222.2014.938051
Tedgui A, Mallat Z (2006) Cytokines in atherosclerosis: pathogenic and regulatory pathways. Physiol Rev 86:515–581. https://doi.org/10.1152/physrev.00024.2005
Tombor LS, John D, Glaser SF et al (2021) Single cell sequencing reveals endothelial plasticity with transient mesenchymal activation after myocardial infarction. Nat Commun 12:681. https://doi.org/10.1038/s41467-021-20905-1
Xue X, Li F, Xu M et al (2023) Gastrodin ameliorates atherosclerosis by inhibiting foam cells formation and inflammation through down-regulating NF-κB pathway. Nutr Metab 20:9. https://doi.org/10.1186/s12986-022-00722-z
Yako Y, Hayashi T, Takeuchi Y et al (2018) ADAM-like decysin-1 (ADAMDEC1) is a positive regulator of epithelial defense against cancer (EDAC) that promotes apical extrusion of RasV12-transformed cells. Sci Rep 8:9639. https://doi.org/10.1038/s41598-018-27469-z
Yang CY, Chanalaris A, Troeberg L (2017) ADAMTS and ADAM metalloproteinases in osteoarthritis - looking beyond the ‘usual suspects’. Osteoarthr Cartil 25:1000–1009. https://doi.org/10.1016/j.joca.2017.02.791
Zadelaar S, Kleemann R, Verschuren L et al (2007) Mouse models for atherosclerosis and pharmaceutical modifiers. Arterioscler Thromb Vasc Biol 27:1706–1721. https://doi.org/10.1161/atvbaha.107.142570
Zernecke A, Weber C (2010) Chemokines in the vascular inflammatory response of atherosclerosis. Cardiovasc Res 86:192–201. https://doi.org/10.1093/cvr/cvp391
Zhao J, Wang J, Liu J et al (2022) Effect and mechanisms of kaempferol against endometriosis based on network pharmacology and in vitro experiments. BMC Complementary Med Ther 22:254. https://doi.org/10.1186/s12906-022-03729-4
Zhou H, Jiang F, Leng Y (2021) Propofol ameliorates ox-LDL-induced endothelial damage through enhancing autophagy via PI3K/Akt/m-TOR pathway: a novel therapeutic strategy in atherosclerosis. Front Mol Biosci 8:695336. https://doi.org/10.3389/fmolb.2021.695336
Zhu W, Shi L, Gong Y et al (2022) Upregulation of ADAMDEC1 correlates with tumor progression and predicts poor prognosis in non-small cell lung cancer (NSCLC) via the PI3K/AKT pathway. Thoracic cancer 13:1027–1039. https://doi.org/10.1111/1759-7714.14354
Funding
This study was supported by the General Project of the Health Commission of Anhui Province (AHWJ2022b020).
Ethics declarations
Ethics approval and consent to participate
An ethical approval was obtained from the Ethics Committee of the Second Affiliated Hospital of Anhui Medical University for this study (YX2021-039). Informed consent was obtained from the patients involved. All animal experiments were conducted according to the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996). All animal experiments in the present study were approved by the Ethic Committee of the Second Affiliated Hospital of Anhui Medical College.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 371 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Gao, F., Cheng, C. et al. Knockdown of ADAMDEC1 ameliorates ox-LDL-induced endothelial cell injury and atherosclerosis progression. Funct Integr Genomics 24, 1 (2024). https://doi.org/10.1007/s10142-023-01278-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10142-023-01278-8