Abstract
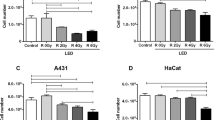
The aim of this study is to investigate the antineoplastic potential of photodynamic therapy (PDT) mediated by an aluminum-phthalocyanine chloride nanoemulsion (AlPc-NE), against an oral squamous cell carcinoma (OSCC) cell line in vitro. Both OSCC (SCC9) and A431 cell lines were studied in vitro. Four study groups were used: Group 1 (phosphate-buffered saline [PBS]), Group 2 (PBS + 28.3 J/cm2 irradiation), Group 3 (AlPc-NE alone), and Group 4 (AlPc-NE + 28.3 J/cm2 irradiation). To test the effect of PDT with AlPc-NE, cell viability, migration, and cell death assays were performed. Moreover, the expressions of Ki-67 and TP53 were evaluated using immunoassays. The results showed that PDT mediated by all AlPc-NE concentrations evaluated (i.e., 0.7, 0.35, and 0.17 nM AlPc) significantly reduced the viability of SCC9 cells. Migration and cell death assays also revealed that PDT with AlPc-NE significantly reduced the rate of migration and increased cell death compared to the control groups. In addition, it was found that PDT with AlPc-NE reduced Ki-67 and mutated TP53 immunoexpression. PDT with AlPc-NE is effective in reducing the viability and migration of SCC9. Moreover, PDT with AlPc-NE nanoemulsions reduces the cell proliferation and expression of mutant TP53.





Similar content being viewed by others
Abbreviations
- PDT:
-
Photodynamic therapy
- AlPc-NE:
-
Aluminum-phthalocyanine chloride nanoemulsion
- OSCC:
-
Oral squamous cell carcinoma
- SCC9:
-
OSCC cell line
- PS:
-
Photosensitizer drug
- ROS:
-
Reactive oxygen species
- PVM/MA:
-
Poly(vinyl methyl ether/maleic anhydride) (polymer nanoparticle)
- LED:
-
Light-emitting diode
- INCT:
-
National Institutes of Science and Technology
- CNPq:
-
National Council for Scientific and Technological Development
- DMEM/Ham’s F-12:
-
Dulbecco’s modified Eagle’s medium/nutrient mixture F-12
- MTT:
-
3-(4,5-Dimethylthiazol-2-Yl)-2,5-diphenyltetrazolium bromide
- AO/EB:
-
Acridine orange/ethidium bromide
References
Bray F et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Global burden of disease cancer collaoration et al (2019) Global regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study. JAMA Oncol 5(12):1749–1768. https://doi.org/10.1001/jamaoncol.2019.2996
Righini CA et al (2008) Risk factors for cancers of the oral cavity, pharynx (cavity excluded) and larynx. Presse Med 37(9):1229–1240
De Paula AM et al (2009) Analysis of 724 cases of primary head and neck squamous cell carcinoma (HNSCC) with a focus on young patients and p53 immunolocalization. Oral Oncol 45(9):777–782
Min S-K et al (2017) Conditional relative survival of oral cavity cancer: Based on Korean Central Cancer Registry. Oral Oncol 72:73–79
Guimaraes TA et al (2016) Metformin increases PDH and suppresses HIF-1alpha under hypoxic conditions and induces cell death in oral squamous cell carcinoma. Oncotarget 7(34):55057–55068
Guimaraes TA et al (2016) Evaluation of the antineoplastic activity of gallic acid in oral squamous cell carcinoma under hypoxic conditions. Anticancer Drugs 27(5):407–416
Shah JP, Gil Z (2009) Current concepts in management of oral cancer – Surgery. Oral Oncol 45(4):394–401
Sobecki M et al (2017) Cell-cycle regulation accounts for variability in Ki-67 expression levels. Cancer Res 77(10):2722–2734
Kanapathipillai M (2018) Treating p53 Mutant Aggregation-Associated Cancer. Cancers (Basel) 10(6):154. https://doi.org/10.3390/cancers10060154
Biel MA (2007) Photodynamic therapy treatment of early oral and laryngeal cancers. Photochem Photobiol 83(5):1063–1068
Chen HM et al (2005) Successful treatment of an extensive verrucous carcinoma with topical 5-aminolevulinic acid-mediated photodynamic therapy. J Oral Pathol Med 34(4):253–256
Castilho-Fernandes A et al (2017) Photodynamic process induced by chloro-aluminum phthalocyanine nanoemulsion in glioblastoma. Photodiagn Photodyn Ther 19:221–228
Agostinis P et al (2011) Photodynamic therapy of cancer: an update. CA Cancer J Clin 61(4):250–281
Huang Z et al (2008) Photodynamic therapy for treatment of solid tumors–potential and technical challenges. Technol Cancer Res Treat 7(4):309–320
Py-Daniel KR et al (2016) Highly efficient photodynamic therapy colloidal system based on chloroaluminum phthalocyanine/pluronic micelles. Eur J Pharm Biopharm 103:23–31
Nunes SM, Sguilla FS, Tedesco AC (2004) Photophysical studies of zinc phthalocyanine and chloroaluminum phthalocyanine incorporated into liposomes in the presence of additives. Braz J Med Biol Res 37(2):273–284
Moll UM et al (2005) Transcription-independent pro-apoptotic functions of p53. Curr Opin Cell Biol 17(6):631–636
Longo JPF et al (2016) Prevention of distant lung metastasis after photodynamic therapy application in a breast cancer tumor model. J Biomed Nanotechnol 12(4):689–699
Muehlmann LA et al (2015) Aluminium-phthalocyanine chloride nanoemulsions for anticancer photodynamic therapy: development and in vitro activity against monolayers and spheroids of human mammary adenocarcinoma MCF-7 cells. J Nanobiotechnology 13:36
Morgado LF et al (2017) Photodynamic Therapy treatment of onychomycosis with Aluminium-Phthalocyanine Chloride nanoemulsions: a proof of concept clinical trial. J Photochem Photobiol B 173:266–270
Chan SW et al (2008) A role for TAZ in migration, invasion, and tumorigenesis of breast cancer cells. Cancer Res 68(8):2592–2598
Kasibhatla S et al (2006) Acridine Orange/Ethidium Bromide (AO/EB) staining to detect apoptosis. Cold Spring Harb Protoc 2006(3): pdb.prot4493
Santos EMS et al (2018) Gallic acid modulates phenotypic behavior and gene expression in oral squamous cell carcinoma cells by interfering with leptin pathway. Pathol Res Pract 214(1):30–37
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Fraga CA et al (2012) A high HIF-1alpha expression genotype is associated with poor prognosis of upper aerodigestive tract carcinoma patients. Oral Oncol 48(2):130–135
Fonseca-Silva T et al (2012) Analysis of p16(CDKN2A) methylation and HPV-16 infection in oral mucosal dysplasia. Pathobiology 79(2):94–100
Pereira CS et al (2012) Impact of the epithelial dysplasia grading and Ki67 proliferation index in the adjacent non-malignant mucosa on recurrence and survival in head and neck squamous cell carcinoma. Pathol Res Pract 208(11):651–656
de Oliveira MV et al (2013) Prognostic value of microvessel density and p53 expression on the locoregional metastasis and survival of the patients with head and neck squamous cell carcinoma. Appl Immunohistochem Mol Morphol 21(5):444–451
Barclay CW, Foster EC, Taylor CL (2018) Restorative aspects of oral cancer reconstruction. Br Dent J 225(9):848–854
Muehlmann LA et al (2014) Aluminum-phthalocyanine chloride associated to poly(methyl vinyl ether-co-maleic anhydride) nanoparticles as a new third-generation photosensitizer for anticancer photodynamic therapy. Int J Nanomedicine 9:1199–1213
Castano AP, Demidova TN, Hamblin MR (2004) Mechanisms in photodynamic therapy: part one—-photosensitizers, photochemistry and cellular localization. Photodiagn Photodyn Ther 1(4):279–293
Debele TA, Peng S, Tsai H-C (2015) Drug carrier for photodynamic cancer therapy. Int J Mol Sci 16(9):22094–22136
Justus CR et al (2014) In vitro cell migration and invasion assays. J Vis Exp: JoVE 88:51046
Domingos PLB et al (2017) Hypoxia reduces the E-cadherin expression and increases OSCC cell migration regardless of the E-cadherin methylation profile. Pathol Res Pract 213(5):496–501
Sobecki M et al (2016) The cell proliferation antigen Ki-67 organises heterochromatin. eLife 5:e13722–e13722
Sobecki M et al (2016) The cell proliferation antigen Ki-67 organises heterochromatin. eLife 5:e13722
Scholzen T, Gerdes J (2000) The Ki-67 protein: from the known and the unknown. J Cell Physiol 182(3):311–322
**g Y et al (2018) Higher Ki67 expression in fibroblast like cells at invasive front indicates better clinical outcomes in oral squamous cell carcinoma patients. Biosci Rep 38(6)
Li C-F et al (2014) AMACR amplification and overexpression in primary imatinib-naïve gastrointestinal stromal tumors: a driver of cell proliferation indicating adverse prognosis. Oncotarget 5(22):11588–11603
Mohamed AA et al (2018) Assessment of expression of Ki-67 in benign and malignant prostatic lesions among sudanese patients. Open Access Maced J Med Sci 6(10):1809–1812
Farias LC et al (2010) Effect of age on the association between p16CDKN2A methylation and DNMT3B polymorphism in head and neck carcinoma and patient survival. Int J Oncol 37(1):167–176
Zhu J et al (2015) Prevalent p53 mutants co-opt chromatin pathways to drive cancer growth. Nature 525(7568):206–211
Ozaki T, Nakagawara A (2011) Role of p53 in cell death and human cancers. Cancers 3(1):994–1013
Pinheiro UB et al (2015) Fuzzy clustering demonstrates that codon 72 SNP rs1042522 of TP53 gene associated with HNSCC but not with prognoses. Tumour Biol 36(12):9259–9265
Souza LR et al (2011) Immunohistochemical analysis of p53, APE1, hMSH2 and ERCC1 proteins in actinic cheilitis and lip squamous cell carcinoma. Histopathology 58(3):352–360
Pereira T et al (2018) MicroRNA profiling reveals dysregulated microRNAs and their target gene regulatory networks in cemento-ossifying fibroma. J Oral Pathol Med 47(1):78–85
Hientz K et al (2017) The role of p53 in cancer drug resistance and targeted chemotherapy. Oncotarget 8(5):8921–8946
Chan JY-H et al (2014) Characterization of a new mouse p53 variant: loss-of-function and gain-of-function. J Biomed Sci 21(1):40–40
Funding
This study was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and the Fundação de Amparo a Pesquisa do Estado de Minas Gerais (FAPEMIG). Dr. Guimarães, Dr. Dr. Santos, and Dr. de Paula are research fellows of the CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All performed procedures involving human participants were conducted following the ethical standards of the institutional and national research committees, the 1964 Helsinki declaration, and its later amendments, or comparable ethical standards. Ethical approval for this study (No. 52760016.9.0000.5146) was obtained from the Institutional Review Board.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cangussu, L.M.B., de Souza, L.R., de Souza, M.G. et al. Photodynamic therapy mediated by nanoparticles Aluminum Chloro Phthalocyanine in oral squamous carcinoma cells. Lasers Med Sci 37, 2509–2516 (2022). https://doi.org/10.1007/s10103-022-03517-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-022-03517-z