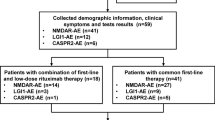
Abstract
Background
For patients with neuronal surface antibody-associated autoimmune encephalitis (NSAE) whose clinical symptoms gradually improve, the recommended course of immunotherapy in China is about 6 months. We aim to explore the relationship between persistent antibody positivity when immunotherapy is discontinued at 6 months and subsequent relapse.
Methods
Prospective inclusion of NSAE patients with clinical remission after 6-month immunotherapy. Their antibody titers and other clinical data were collected at onset and 6 months later. Based on the antibody test results at 6 months, patients were divided into an antibody-persistent group and an antibody-negative conversion group, and then the rate of relapse between the two groups were compared.
Results
The study included 28 NSAE patients who were antibody-positive at diagnosis. After 6-month immunotherapy, there were 16 (57.1%) cases with persistent antibodies and 12 (42.9%) cases with antibody-negative conversion. In the acute phase of onset, seizures were more common in patients with persistent antibodies (87.5% vs. 50.0%, p = 0.044). During a mean follow-up period of 22 months, patients with persistent antibodies were more likely to experience relapse than those with antibody-negative conversion (37.5% vs. 0.0%, p = 0.024). There were no significant differences in antibody types, CSF findings, results of MRI and EEG, tumor combination, immunotherapy, and long-term outcome between the two groups (p > 0.05).
Conclusions
For patients with persistent antibodies when immunotherapy is discontinued at 6 months, persistent antibody positivity was associated with a higher relapse rate.


Similar content being viewed by others
Data Availability
Anonymized data, not published in the article, can be made available upon reasonable request from the corresponding author.
References
Graus F, Titulaer MJ, Balu R et al (2016) A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol 15(4):391–404. https://doi.org/10.1016/S1474-4422(15)00401-9
Chinese Society of Neuroinfectious Diseases and Cerebrospinal Fluid Cytology (2022) Expert consensus on diagnosis and treatment of autoimmune encephalitis in China. Chin J Neurol 55(9):931–949. https://doi.org/10.3760/cma.j.cn113694-20220219-00118
Lai QL, Cai MT, Zheng Y et al (2022) Evaluation of csf albumin quotient in neuronal surface antibody-associated autoimmune encephalitis. Fluids Barriers Cns 19(1):93. https://doi.org/10.1186/s12987-022-00392-2
Titulaer MJ, Mccracken L, Gabilondo I et al (2013) Treatment and prognostic factors for long-term outcome in patients with anti-nmda receptor encephalitis: an observational cohort study. Lancet Neurol 12(2):157–165. https://doi.org/10.1016/S1474-4422(12)70310-1
Rodriguez A, Klein CJ, Sechi E et al (2022) Lgi1 antibody encephalitis: acute treatment comparisons and outcome. J Neurol Neurosurg Psychiatry 93(3):309–315. https://doi.org/10.1136/jnnp-2021-327302
Varley JA, Strippel C, Handel A, Irani SR (2023) Autoimmune encephalitis: recent clinical and biological advances. J Neurol. https://doi.org/10.1007/s00415-023-11685-3
Zhong R, Chen Q, Zhang X, Zhang H, Lin W (2022) Relapses of anti-nmdar, anti-gababr and anti-lgi1 encephalitis: a retrospective cohort study. Front Immunol 13:918396. https://doi.org/10.3389/fimmu.2022.918396
Gresa-Arribas N, Titulaer MJ, Torrents A et al (2014) Antibody titres at diagnosis and during follow-up of anti-nmda receptor encephalitis: a retrospective study. Lancet Neurol 13(2):167–177. https://doi.org/10.1016/S1474-4422(13)70282-5
Ariño H, Armangué T, Petit-Pedrol M et al (2016) Anti-lgi1-associated cognitive impairment: presentation and long-term outcome. Neurology 87(8):759–765. https://doi.org/10.1212/WNL.0000000000003009
Ciano-Petersen NL, Robert M, Muñiz-Castrillo Set al (2023) Prognostic value of persistent csf antibodies at 12 months in anti-nmdar encephalitis. Neurol Neuroimmunol Neuroinflamm 10(4). https://doi.org/10.1212/NXI.0000000000200108
Gong X, Chen C, Liu X et al (2021) Long-term functional outcomes and relapse of anti-nmda receptor encephalitis. Neurology - Neuroimmunol Neuroinflammation 8(2):e958. https://doi.org/10.1212/NXI.0000000000000958
Yu Y, Wu Y, Cao X et al (2021) The clinical features and prognosis of anti-nmdar encephalitis depends on blood brain barrier integrity. Mult Scler Relat Disord 47:102604. https://doi.org/10.1016/j.msard.2020.102604
Nosadini M, Granata T, Matricardi S et al (2019) Relapse risk factors in anti-n-methyl-d-aspartate receptor encephalitis. Dev Med Child Neurol 61(9):1101–1107. https://doi.org/10.1111/dmcn.14267
Wu CY, Wu JD, Chen CC (2021) The association of ovarian teratoma and anti-n-methyl-d-aspartate receptor encephalitis: an updated integrative review. Int J Mol Sci 22(20). https://doi.org/10.3390/ijms222010911
Dalmau J, Gleichman AJ, Hughes EG et al (2008) Anti-nmda-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7(12):1091–1098. https://doi.org/10.1016/S1474-4422(08)70224-2
Hughes EG, Peng X, Gleichman AJ et al (2010) Cellular and synaptic mechanisms of anti-nmda receptor encephalitis. J Neurosci 30(17):5866–5875. https://doi.org/10.1523/JNEUROSCI.0167-10.2010
Lancaster E, Dalmau J (2012) Neuronal autoantigens–pathogenesis, associated disorders and antibody testing. Nat Rev Neurol 8(7):380–390. https://doi.org/10.1038/nrneurol.2012.99
Gabilondo I, Saiz A, Galán L et al (2011) Analysis of relapses in anti-nmdar encephalitis. Neurology 77(10):996–999. https://doi.org/10.1212/WNL.0b013e31822cfc6b
Dai Y, Zhang J, Ren H et al (2019) Surgical outcomes in patients with anti-n-methyl d-aspartate receptor encephalitis with ovarian teratoma. Am J Obstet Gynecol 221(5):481–485. https://doi.org/10.1016/j.ajog.2019.05.026
Leypoldt F, Höftberger R, Titulaer MJ et al (2015) Investigations on cxcl13 in anti-n-methyl-d-aspartate receptor encephalitis: a potential biomarker of treatment response. Jama Neurol 72(2):180–186. https://doi.org/10.1001/jamaneurol.2014.2956
Ciano-Petersen NL, Cabezudo-García P, Muñiz-Castrillo S, Honnorat J, Serrano-Castro PJ, Oliver-Martos B (2021) Current status of biomarkers in anti-n-methyl-d-aspartate receptor encephalitis. Int J Mol Sci 22(23). https://doi.org/10.3390/ijms222313127
Zeng C, Chen L, Chen B et al (2018) Th17 cells were recruited and accumulated in the cerebrospinal fluid and correlated with the poor prognosis of anti-nmdar encephalitis. Acta Biochim Biophys Sin (Shanghai) 50(12):1266–1273. https://doi.org/10.1093/abbs/gmy137
Wendel EM, Thonke HS, Bertolini A et al (2022) Temporal dynamics of mog antibodies in children with acquired demyelinating syndrome. Neurology - Neuroimmunol Neuroinflammation 9(6):e200035. https://doi.org/10.1212/NXI.0000000000200035
Waters P, Fadda G, Woodhall M et al (2020) Serial anti-myelin oligodendrocyte glycoprotein antibody analyses and outcomes in children with demyelinating syndromes. Jama Neurol 77(1):82–93. https://doi.org/10.1001/jamaneurol.2019.2940
López-Chiriboga AS, Majed M, Fryer J et al (2018) Association of mog-igg serostatus with relapse after acute disseminated encephalomyelitis and proposed diagnostic criteria for mog-igg-associated disorders. Jama Neurol 75(11):1355–1363. https://doi.org/10.1001/jamaneurol.2018.1814
Oliveira LM, Apóstolos-Pereira SL, Pitombeira MS, Bruel TP, Callegaro D, Sato DK (2019) Persistent mog-igg positivity is a predictor of recurrence in mog-igg-associated optic neuritis, encephalitis and myelitis. Mult Scler 25(14):1907–1914. https://doi.org/10.1177/1352458518811597
Alexopoulos H, Kosmidis ML, Dalmau J, Dalakas MC (2011) Paraneoplastic anti-nmdar encephalitis: long term follow-up reveals persistent serum antibodies. J Neurol 258(8):1568–1570. https://doi.org/10.1007/s00415-011-5982-4
Hansen HC, Klingbeil C, Dalmau J, Li W, Weissbrich B, Wandinger KP (2013) Persistent intrathecal antibody synthesis 15 years after recovering from anti-n-methyl-d-aspartate receptor encephalitis. Jama Neurol 70(1):117–119. https://doi.org/10.1001/jamaneurol.2013.585
de Bruijn M, van Sonderen A, van Coevorden-Hameete MH et al (2019) Evaluation of seizure treatment in anti-lgi1, anti-nmdar, and anti-gaba(b)r encephalitis. Neurology 92(19):e2185–e2196. https://doi.org/10.1212/WNL.0000000000007475
Liu X, Guo K, Lin J et al (2022) Long-term seizure outcomes in patients with autoimmune encephalitis: a prospective observational registry study update. Epilepsia 63(7):1812–1821. https://doi.org/10.1111/epi.17245
Zhong R, Zhang X, Chen Q, Li M, Guo X, Lin W (2022) Acute symptomatic seizures and risk of epilepsy in autoimmune encephalitis: a retrospective cohort study. Front Immunol 13:813174. https://doi.org/10.3389/fimmu.2022.813174
Shen CH, Fang GL, Yang F et al (2020) Seizures and risk of epilepsy in anti-nmdar, anti-lgi1, and anti-gaba(b) r encephalitis. Ann Clin Transl Neurol 7(8):1392–1399. https://doi.org/10.1002/acn3.51137
Rada A, Birnbacher R, Gobbi C et al (2021) Seizures associated with antibodies against cell surface antigens are acute symptomatic and not indicative of epilepsy: insights from long-term data. J Neurol 268(3):1059–1069. https://doi.org/10.1007/s00415-020-10250-6
Author information
Authors and Affiliations
Contributions
FZ designed the study and drafted the manuscript. YG, ML, HS, HZ, and YY collected and analyzed the data. JW designed the study and revised the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethical approval
The study was performed according to the ethical principles of the Declaration of Helsinki and was approved by the Ethics Committee of the First Hospital of Shanxi Medical University. Written informed consent was obtained from all patients or their legal representatives.
Conflict of interest
The authors declare that there are no conflicts of interest in connection with this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, F., Guo, Y., Liu, M. et al. Predictive value of persistent antibodies at 6 months for relapse in neuronal surface antibody-associated autoimmune encephalitis. Neurol Sci 45, 1599–1607 (2024). https://doi.org/10.1007/s10072-023-07151-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-07151-7