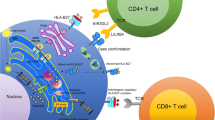
Abstract
Susceptibility to ankylosing spondylitis is highly genetic, with a heritability greater than 90 %. Presence of the HLA-B27 MHC class I allele remains the greatest genetic risk factor identified to date. Beyond its nominal role in antigen presentation, HLA-B27 displays interesting and possibly unique biochemical characteristics which may contribute to disease pathogenesis. During its biosynthesis in the endoplasmic reticulum (ER), HLA-B27 folds very slowly and misfolds, inducing ER stress. Herein, we describe a major outcome of ER stress, the unfolded protein response (UPR), as well as consequences of the UPR for inflammation and autophagy. The ability of the UPR to augment inflammatory cytokine production is particularly intriguing given the centrality of cytokines in spondyloarthritis. Evidence for the relevance of an HLA-B27-related UPR to spondyloarthritis pathogenesis in animal models and human subjects will be reviewed. As greater pharmacologic capacity to modulate ER stress becomes available, improved understanding of the role of the UPR in spondyloarthritis may yield new therapeutic targets.


Similar content being viewed by others
References
Brown MA, Kennedy LG, MacGregor AJ, Darke C, Duncan E et al (1997) Susceptibility to ankylosing spondylitis in twins: the role of genes, HLA, and the environment. Arthritis Rheum 40:1823–1828
Reveille JD (2014) An update on the contribution of the MHC to AS susceptibility. Clin Rheumatol 33:749–757
Reveille JD (2012) Genetics of spondyloarthritis—beyond the MHC. Nat Rev Rheumatol 8:296–304
Robinson PC, Brown MA (2014) Genetics of ankylosing spondylitis. Mol Immunol 57:2–11
Hammer RE, Maika SD, Richardson JA, Tang JP, Taurog JD (1990) Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human beta 2m: an animal model of HLA-B27-associated human disorders. Cell 63:1099–1112
Bowness P (2015) Hla-B27. Annu Rev Immunol 33:29–48
Fiorillo MT, Meadows L, D’Amato M, Shabanowitz J, Hunt DF et al (1997) Susceptibility to ankylosing spondylitis correlates with the C-terminal residue of peptides presented by various HLA-B27 subtypes. Eur J Immunol 27:368–373
Benjamin R, Parham P (1990) Guilt by association: HLA-B27 and ankylosing spondylitis. Immunol Today 11:137–142
Hulsmeyer M, Fiorillo MT, Bettosini F, Sorrentino R, Saenger W et al (2004) Dual, HLA-B27 subtype-dependent conformation of a self-peptide. J Exp Med 199:271–281
Narzi D, Becker CM, Fiorillo MT, Uchanska-Ziegler B, Ziegler A et al (2012) Dynamical characterization of two differentially disease associated MHC class I proteins in complex with viral and self-peptides. J Mol Biol 415:429–442
Ruckert C, Fiorillo MT, Loll B, Moretti R, Biesiadka J et al (2006) Conformational dimorphism of self-peptides and molecular mimicry in a disease-associated HLA-B27 subtype. J Biol Chem 281:2306–2316
May E, Dorris ML, Satumtira N, Iqbal I, Rehman MI et al (2003) CD8 alpha beta T cells are not essential to the pathogenesis of arthritis or colitis in HLA-B27 transgenic rats. J Immunol 170:1099–1105
Antoniou AN, Ford S, Taurog JD, Butcher GW, Powis SJ (2004) Formation of HLA-B27 homodimers and their relationship to assembly kinetics. J Biol Chem 279:8895–8902
Dangoria NS, DeLay ML, Kingsbury DJ, Mear JP, Uchanska-Ziegler B et al (2002) HLA-B27 misfolding is associated with aberrant intermolecular disulfide bond formation (dimerization) in the endoplasmic reticulum. J Biol Chem 277:23459–23468
Galocha B, de Castro JA (2008) Folding of HLA-B27 subtypes is determined by the global effect of polymorphic residues and shows incomplete correspondence to ankylosing spondylitis. Arthritis Rheum 58:401–412
Taurog JD (2010) The role of HLA-B27 in spondyloarthritis. J Rheumatol 37:2606–2616
Tran TM, Satumtira N, Dorris ML, May E, Wang A et al (2004) HLA-B27 in transgenic rats forms disulfide-linked heavy chain oligomers and multimers that bind to the chaperone BiP. J Immunol 172:5110–5119
Mear JP, Schreiber KL, Munz C, Zhu X, Stevanovic S et al (1999) Misfolding of HLA-B27 as a result of its B pocket suggests a novel mechanism for its role in susceptibility to spondyloarthropathies. J Immunol 163:6665–6670
Jeanty C, Sourisce A, Noteuil A, Jah N, Wielgosik A et al (2014) HLA-B27 Subtype oligomerization and intracellular accumulation patterns correlate with predisposition to spondyloarthritis. Arthritis Rheumatol 66:2113–2123
Schroder M, Kaufman RJ (2005) The mammalian unfolded protein response. Annu Rev Biochem 74:739–789
Ron D, Walter P (2007) Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8:519–529
Bettigole SE, Glimcher LH (2015) Endoplasmic reticulum stress in immunity. Annu Rev Immunol 33:107–138
Oakes SA, Papa FR (2015) The role of endoplasmic reticulum stress in human pathology. Annu Rev Pathol 10:173–194
Malhotra JD, Kaufman RJ (2007) The endoplasmic reticulum and the unfolded protein response. Semin Cell Dev Biol 18:716–731
Urano F, Wang X, Bertolotti A, Zhang Y, Chung P et al (2000) Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287:664–666
Tam AB, Mercado EL, Hoffmann A, Niwa M (2012) ER stress activates NF-kappaB by integrating functions of basal IKK activity, IRE1 and PERK. PLoS One 7:e45078
Deng J, Lu PD, Zhang Y, Scheuner D, Kaufman RJ et al (2004) Translational repression mediates activation of nuclear factor kappa B by phosphorylated translation initiation factor 2. Mol Cell Biol 24:10161–10168
Martinon F, Chen X, Lee AH, Glimcher LH (2010) TLR activation of the transcription factor XBP1 regulates innate immune responses in macrophages. Nat Immunol 11:411–418
Zeng L, Liu YP, Sha H, Chen H, Qi L et al (2010) XBP-1 couples endoplasmic reticulum stress to augmented IFN-beta induction via a cis-acting enhancer in macrophages. J Immunol 185:2324–2330
Goodall JC, Wu C, Zhang Y, McNeill L, Ellis L et al (2010) Endoplasmic reticulum stress-induced transcription factor, CHOP, is crucial for dendritic cell IL-23 expression. Proc Natl Acad Sci U S A 107:17698–17703
Smith JA, Turner MJ, DeLay ML, Klenk EI, Sowders DP et al (2008) Endoplasmic reticulum stress and the unfolded protein response are linked to synergistic IFN-beta induction via X-box binding protein 1. Eur J Immunol 38:1194–1203
DeLay ML, Turner MJ, Klenk EI, Smith JA, Sowders DP et al (2009) HLA-B27 misfolding and the unfolded protein response augment interleukin-23 production and are associated with Th17 activation in transgenic rats. Arthritis Rheum 60:2633–2643
Yeremenko N, Paramarta JE, Baeten D (2014) The interleukin-23/interleukin-17 immune axis as a promising new target in the treatment of spondyloarthritis. Curr Opin Rheumatol 26:361–370
Sherlock JP, Joyce-Shaikh B, Turner SP, Chao CC, Sathe M et al (2012) IL-23 induces spondyloarthropathy by acting on ROR-gammat + CD3 + CD4-CD8- entheseal resident T cells. Nat Med 18:1069–1076
Guiliano DB, Fussell H, Lenart I, Tsao E, Nesbeth D et al (2014) Endoplasmic reticulum degradation-enhancing alpha-mannosidase-like protein 1 targets misfolded HLA-B27 dimers for endoplasmic reticulum-associated degradation. Arthritis Rheumatol 66:2976–2988
Lapaquette P, Guzzo J, Bretillon L, Bringer MA (2015) Cellular and molecular connections between autophagy and inflammation. Mediators Inflamm 2015:398483
Deegan S, Saveljeva S, Gorman AM, Samali A (2013) Stress-induced self-cannibalism: on the regulation of autophagy by endoplasmic reticulum stress. Cell Mol Life Sci 70:2425–2441
Turner MJ, Sowders DP, DeLay ML, Mohapatra R, Bai S et al (2005) HLA-B27 misfolding in transgenic rats is associated with activation of the unfolded protein response. J Immunol 175:2438–2448
Tran TM, Dorris ML, Satumtira N, Richardson JA, Hammer RE et al (2006) Additional human beta2-microglobulin curbs HLA-B27 misfolding and promotes arthritis and spondylitis without colitis in male HLA-B27-transgenic rats. Arthritis Rheum 54:1317–1327
Turner MJ, Delay ML, Bai S, Klenk E, Colbert RA (2007) HLA-B27 up-regulation causes accumulation of misfolded heavy chains and correlates with the magnitude of the unfolded protein response in transgenic rats: Implications for the pathogenesis of spondylarthritis-like disease. Arthritis Rheum 56:215–223
Benham H, Rehaume LM, Hasnain SZ, Velasco J, Baillet AC et al (2014) Interleukin-23 mediates the intestinal response to microbial beta-1,3-glucan and the development of spondyloarthritis pathology in SKG mice. Arthritis Rheumatol 66:1755–1767
Gu J, Rihl M, Marker-Hermann E, Baeten D, Kuipers JG et al (2002) Clues to pathogenesis of spondyloarthropathy derived from synovial fluid mononuclear cell gene expression profiles. J Rheumatol 29:2159–2164
Dong W, Zhang Y, Yan M, Liu H, Chen Z et al (2008) Upregulation of 78-kDa glucose-regulated protein in macrophages in peripheral joints of active ankylosing spondylitis. Scand J Rheumatol 37:427–434
Feng Y, Ding J, Fan CM, Zhu P (2012) Interferon-gamma contributes to HLA-B27-associated unfolded protein response in spondyloarthropathies. J Rheumatol 39:574–582
Neerinckx B, Carter S, Lories RJ (2014) No evidence for a critical role of the unfolded protein response in synovium and blood of patients with ankylosing spondylitis. Ann Rheum Dis 73:629–630
Zeng L, Lindstrom MJ, Smith JA (2011) Ankylosing spondylitis macrophage production of higher levels of interleukin-23 in response to lipopolysaccharide without induction of a significant unfolded protein response. Arthritis Rheum 63:3807–3817
Ciccia F, Accardo-Palumbo A, Rizzo A, Guggino G, Raimondo S et al (2014) Evidence that autophagy, but not the unfolded protein response, regulates the expression of IL-23 in the gut of patients with ankylosing spondylitis and subclinical gut inflammation. Ann Rheum Dis 73:1566–1574
Engin F, Yermalovich A, Nguyen T, Hummasti S, Fu W et al (2013) Restoration of the unfolded protein response in pancreatic beta cells protects mice against type 1 diabetes. Sci Transl Med 5:211ra156
Layh-Schmitt G, Yang EY, Kwon G, Colbert RA (2013) HLA-B27 alters the response to tumor necrosis factor alpha and promotes osteoclastogenesis in bone marrow monocytes from HLA-B27-transgenic rats. Arthritis Rheum 65:2123–2131
Rivas A, Vidal RL, Hetz C (2015) Targeting the unfolded protein response for disease intervention. Expert Opin Ther Targets 19:1203–1218
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Funding
This study has been funded by the Rheumatology Research Foundation, NIH/NHLBI P01 HL070831.
Rights and permissions
About this article
Cite this article
Smith, J.A. The role of the unfolded protein response in axial spondyloarthritis. Clin Rheumatol 35, 1425–1431 (2016). https://doi.org/10.1007/s10067-015-3117-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-015-3117-5