Abstract
Ultrasound-assisted magnetic nanofluid-based liquid-phase microextraction was coupled to GC-MS in a method for simultaneous determination of the pyrethroid insecticides permethrin, deltamethrin, and cypermethrin. A highly efficient extraction solvent called “magnetic nanofluid (MNF)” is introduced for preconcentration of pyrethroids. The MNF consists of magnetic multiwalled carbon nanotubes (MMWCNTs) and deep eutectic solvent. Following microextraction, the MNF was detached by an external magnet from the medium without the need for centrifugation. 2-Pyridinecarboxamide, choline chloride, and anhydrous ferric chloride were used for the synthesis of the deep eutectic solvent. The effects of pH value, volume of MNF, sonication time, sample volume, and ionic strength, type and amount of back extraction solvent were investigated. Under optimum conditions of MNF volume of 50 μL, time of 5 min, 100 μL acetone as back extraction solvent, NaCl concentration of 1 mol.L−1 and sample volume of 30 mL, the detection limits are 2.8, 2.7 and 2.0 ng·mL−1 for permethrin, deltamethrin and cypermethrin, respectively. The linear response ranges are between 0.01 and 250 ng·mL−1, and relative standard deviations (for n = 7) are 3.5, 3.2 and 2.8%. The method was successfully applied to the determination of trace levels of permethrin, deltamethrin and cypermethrin in (spiked) milk samples.

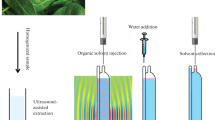
Schematic representation of ultrasound assisted magnetic nanofluid-based liquid phase microextraction separation/preconcentration of permethrin, deltametrin, and cypermethrin residues in milk using GC-MS.






Similar content being viewed by others
References
Liu X, Liu C, Qian H, Qu Y, Zhang S, Lu R, Gao H, Zhou W (2019) Ultrasound-assisted dispersive liquid-liquid microextraction based on a hydrophobic deep eutectic solvent for the preconcentration of pyrethroid insecticides prior to determination by high-performance liquid chromatography. Microchem J 146:614–621. https://doi.org/10.1016/j.microc.2019.01.048
Shirani M, Haddadi H, Rezaee M, Semnani A, Habibollahi S (2016) Solid-phase extraction combined with dispersive liquid–liquid microextraction for the simultaneous determination of Deltamethrin and permethrin in honey by gas chromatography–mass spectrometry. Food Anal Methods 9(9):2613–2620. https://doi.org/10.1007/s12161-016-0455-0
Liu F, Yang X, Wu X, ** X, Gao H, Zhang S, Zhou W, Lu R (2018) A dispersive magnetic solid phase microextraction based on ionic liquid-coated and cyclodextrin-functionalized magnetic core dendrimer nanocomposites for the determination of pyrethroids in juice samples. Food Chem 268:485–491. https://doi.org/10.1016/j.foodchem.2018.06.105
Zhou P, Chen K, Gao M, Qu J, Zhang Z, Dahlgren RA, Li Y, Liu W, Huang H, Wang X (2018) Magnetic effervescent tablets containing ionic liquids as a non-conventional extraction and dispersive agent for determination of pyrethroids in milk. Food Chem 268:468–475. https://doi.org/10.1016/j.foodchem.2018.06.099
Manav ÖG, Dinç-Zor Ş, Alpdoğan G (2019) Optimization of a modified QuEChERS method by means of experimental design for multiresidue determination of pesticides in milk and dairy products by GC–MS. Microchem J 144:124–129. https://doi.org/10.1016/j.microc.2018.08.056
Sharif Z, Man YBC, Hamid NSA, Keat CC (2006) Determination of organochlorine and pyrethroid pesticides in fruit and vegetables using solid phase extraction clean-up cartridges. J Chromatogr A 1127(1):254–261. https://doi.org/10.1016/j.chroma.2006.06.007
Zhou Q, Gao Y, Bai H, **e G (2010) Preconcentration sensitive determination of pyrethroid insecticides in environmental water samples with solid phase extraction with SiO2 microspheres cartridge prior to high performance liquid chromatography. J Chromatogr A 1217(31):5021–5025. https://doi.org/10.1016/j.chroma.2010.05.060
Pastor-Belda M, Navarro-Jiménez T, Garrido I, Viñas P, Campillo N, Fenoll J, Hernández-Córdoba M (2018) Magnetic solid-phase extraction or dispersive liquid–liquid microextraction for pyrethroid determination in environmental samples. J Sep Sci 41(12):2565–2575. https://doi.org/10.1002/jssc.201800109
Campillo N, López-García I, Hernández-Córdoba M, Viñas P (2018) Food and beverage applications of liquid-phase microextraction. TrAC Trend Anal Chem 109:116–123. https://doi.org/10.1016/j.trac.2018.10.004
Feng J, Wang X, Tian Y, Luo C, Sun M (2018) Melamine–formaldehyde aerogel coating for in-tube solid-phase microextraction. J Chromatogr A 1577:8–14. https://doi.org/10.1016/j.chroma.2018.09.047
Barrionuevo WR, Lanças FM (2002) Comparison of liquid–liquid extraction (LLE), solid-phase extraction (SPE), and solid-phase microextraction (SPME) for Pyrethroid pesticides analysis from Enriched River water. Bull Environ Contam Toxicol 69(1):123–128. https://doi.org/10.1007/s00128-002-0018-5
dos Anjos JP, de Andrade JB (2014) Determination of nineteen pesticides residues (organophosphates, organochlorine, pyrethroids, carbamate, thiocarbamate and strobilurin) in coconut water by SDME/GC–MS. Microchem J 112:119–126. https://doi.org/10.1016/j.microc.2013.10.001
Hu L, Wang H, Qian H, Liu C, Lu R, Zhang S, Zhou W, Gao H, Xu D (2016) Centrifuge-less dispersive liquid-liquid microextraction base on the solidification of switchable solvent for rapid on-site extraction of four pyrethroid insecticides in water samples. J Chromatogr A 1472:1–9. https://doi.org/10.1016/j.chroma.2016.10.013
Torbati M, Farajzadeh MA, Torbati M, Nabil AAA, Mohebbi A, Afshar Mogaddam MR (2018) Development of salt and pH–induced solidified floating organic droplets homogeneous liquid–liquid microextraction for extraction of ten pyrethroid insecticides in fresh fruits and fruit juices followed by gas chromatography-mass spectrometry. Talanta 176:565–572. https://doi.org/10.1016/j.talanta.2017.08.074
Haddadi H, Shirani M, Semnani A, Rezaee M, Mashayekhi HA, Hosseinian A (2014) Simultaneous determination of Deltamethrin and permethrin in water samples using homogeneous liquid–liquid microextraction via flotation assistance and GC-FID. Chromatographia 77(9):715–721. https://doi.org/10.1007/s10337-014-2666-0
Shirani M, Akbari A, Hassani M, Goli A, Habibollahi S, Akbarian P (2018) Homogeneous liquid-liquid microextraction via flotation assistance coupled with gas chromatography-mass spectrometry for determination of myclobutanil in cucumber, tomato, grape, and strawberry using genetic algorithm. Int J Environ Anal Chem 98(3):271–285. https://doi.org/10.1080/03067319.2018.1454439
Kanberoglu GS, Yilmaz E, Soylak M (2019) Develo** a new and simple ultrasound-assisted emulsification liquid phase microextraction method built upon deep eutectic solvents for patent blue V in syrup and water samples. Microchem J 145:813–818. https://doi.org/10.1016/j.microc.2018.11.053
Florindo C, Lima F, Ribeiro BD, Marrucho IM (2019) Deep eutectic solvents: overcoming 21st century challenges. Curr Opin Green Sust Chem 18:31–36. https://doi.org/10.1016/j.cogsc.2018.12.003
Xu K, Wang Y, Wei X, Chen J, Xu P, Zhou Y (2018) Preparation of magnetic molecularly imprinted polymers based on a deep eutectic solvent as the functional monomer for specific recognition of lysozyme. Microchim Acta 185(2):146. https://doi.org/10.1007/s00604-018-2707-8
Shirani M, Habibollahi S, Akbari A (2019) Centrifuge-less deep eutectic solvent based magnetic nanofluid-linked air-agitated liquid–liquid microextraction coupled with electrothermal atomic absorption spectrometry for simultaneous determination of cadmium, lead, copper, and arsenic in food samples and non-alcoholic beverages. Food Chem 281:304–311. https://doi.org/10.1016/j.foodchem.2018.12.110
Shirani M, Semnani A, Habibollahi S, Haddadi H (2015) Ultrasound-assisted, ionic liquid-linked, dual-magnetic multiwall carbon nanotube microextraction combined with electrothermal atomic absorption spectrometry for simultaneous determination of cadmium and arsenic in food samples. JAAS 30(5):1057–1063. https://doi.org/10.1039/C4JA00481G
Shamsipur M, Yazdanfar N, Ghambarian M (2016) Combination of solid-phase extraction with dispersive liquid–liquid microextraction followed by GC–MS for determination of pesticide residues from water, milk, honey and fruit juice. Food Chem 204:289–297. https://doi.org/10.1016/j.foodchem.2016.02.090
Samadi S, Sereshti H, Assadi Y (2012) Ultra-preconcentration and determination of thirteen organophosphorus pesticides in water samples using solid-phase extraction followed by dispersive liquid–liquid microextraction and gas chromatography with flame photometric detection. J Chromatogr A 1219:61–65. https://doi.org/10.1016/j.chroma.2011.11.019
Hernandes T, Dores EFGC, Ribeiro ML, Rossignoli PA, Malm O (2014) Simple method to determine residual cypermethrin and deltamethrin in bovine milk. J Braz Chem Soc 25:1656–1661
Gao Y, Sun P (2018) Determination of five pyrethroid pesticides residue in liquid milk by gas chromatography using multi-walled carbon nanotubes as dispersion solid phase extraction sorbent. Acta Chromatogr 30(2):141–146
San Román I, Alonso ML, Bartolomé L, Alonso RM (2012) Hollow fibre-based liquid-phase microextraction technique combined with gas chromatography–mass spectrometry for the determination of pyrethroid insecticides in water samples. Talanta 100:246–253. https://doi.org/10.1016/j.talanta.2012.04.039
Boonchiangma S, Ngeontae W, Srijaranai S (2012) Determination of six pyrethroid insecticides in fruit juice samples using dispersive liquid–liquid microextraction combined with high performance liquid chromatography. Talanta 88:209–215. https://doi.org/10.1016/j.talanta.2011.10.033
Acknowledgements
The authors gratefully appreciate University of Jiroft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 181 kb)
Rights and permissions
About this article
Cite this article
Shirani, M., Akbari-adergani, B., Jazi, M.B. et al. Green ultrasound assisted magnetic nanofluid-based liquid phase microextraction coupled with gas chromatography-mass spectrometry for determination of permethrin, deltamethrin, and cypermethrin residues. Microchim Acta 186, 674 (2019). https://doi.org/10.1007/s00604-019-3763-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3763-4