Abstract
Purpose
To identify damage to the inner retinal layer and microvasculature in the peripapillary area according to the severity of diabetic retinopathy (DR).
Methods
Patients were divided into four groups: control (group 1), type 2 diabetes (T2DM) without DR (group 2), mild to moderate nonproliferative DR (NPDR) (group 3), and severe NPDR (group 4). The peripapillary retinal nerve fiber layer (pRNFL) thickness and peripapillary vessel density (VD) were compared. Linear regression analysis was performed to identify factors associated with the DR severity.
Results
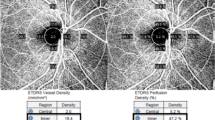
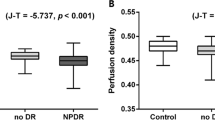
The average pRNFL thicknesses were 96.2 ± 7.1, 94.1 ± 9.6, 92.0 ± 9.9, and 90.3 ± 12.4 μm in groups 1, 2, 3, and 4, respectively (P = 0.003) (post hoc analyses: group 1 vs. group 2, P = 0.529; group 2 vs. group 3, P = 0.627; group 2 vs. group 4, P = 0.172; group 3 vs. group 4, P = 0.823). The VDs of the outer ring were 18.9 ± 0.6, 18.4 ± 0.8, 17.9 ± 1.1, and 17.3 ± 1.6 mm−1 in groups 1, 2, 3 and 4, respectively (P < 0.001) (all pairwise comparisons, P < 0.050). In multivariate analysis, the VD of the outer ring (B = − 0.35, P < 0.001) was significantly associated with the DR severity.
Conclusions
The peripapillary microvasculature reflects retinal damage following DR progression better than the structure of the pRNFL.



Similar content being viewed by others
References
Yau JW, Rogers SL, Kawasaki R et al (2012) Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 35(3):556–564
Simó R, Stitt AW, Gardner TW (2018) Neurodegeneration in diabetic retinopathy: does it really matter? Diabetologia 61:1902–1912
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW (1998) Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest 102(4):783–791. https://doi.org/10.1172/JCI2425
Zeng Y, Cao D, Yu H et al (2019) Early retinal neurovascular impairment in patients with diabetes without clinically detectable retinopathy. Br J Ophthalmol 103(12):1747–1752
Jackson GR, Scott IU, Quillen DA, Walter LE, Gardner TW (2012) Inner retinal visual dysfunction is a sensitive marker of non-proliferative diabetic retinopathy. Br J Ophthalmol 96(5):699–703
Vujosevic S, Muraca A, Gatti V et al (2018) Peripapillary microvascular and neural changes in diabetes mellitus: an OCT-angiography study. Invest Ophthalmol Vis Sci 59(12):5074–5081
Wang X-N, Cai X, Li T-T, Long D, Wu Q (2022) Peripapillary vessel density and retinal nerve fiber layer thickness changes in early diabetes retinopathy. Int J Ophthalmol 15(9):1488
Frizziero L, Parrozzani R, Londei D, Pilotto E, Midena E (2021) Quantification of vascular and neuronal changes in the peripapillary retinal area secondary to diabetic retinopathy. Br J Ophthalmol 105(11):1577–1583
Nesper PL, Roberts PK, Onishi AC et al (2017) Quantifying microvascular abnormalities with increasing severity of diabetic retinopathy using optical coherence tomography angiography. Invest Ophthalmol Vis Sci 58(6):BIO307–BIO315
Sung J-Y, Lee M-W, Lim H-B, Ryu C-K, Yu H-Y, Kim J-Y (2022) The ganglion cell-inner plexiform layer thickness/vessel density of superficial vascular plexus ratio according to the progression of diabetic retinopathy. Invest Ophthalmol Vis Sci 63(6):4–4
Santos T, Warren LH, Santos AR et al (2022) Swept-source OCTA quantification of capillary closure predicts ETDRS severity staging of NPDR. Br J Ophthalmol 106(5):712–718
Maier A-KB, Mandrossa D, Reitemeyer E, Winterhalter S, Rübsam A, Pleyer U (2024) Viral anterior uveitis: differences in retinal vessel area density between the affected and non-affected eye using optical coherence tomography angiography. Ocular Immunol Inflamm 22:1–9
Cornelius A, Pilger D, Riechardt A et al (2022) Macular, papillary and peripapillary perfusion densities measured with optical coherence tomography angiography in primary open angle glaucoma and pseudoexfoliation glaucoma. Graefe's Archive Clin Exp Ophthalmol 260(3):957–965
Lim HB, Shin YI, Lee MW, Park GS, Kim JY (2019) Longitudinal changes in the peripapillary retinal nerve fiber layer thickness of patients with type 2 diabetes. JAMA ophthalmol 137(10):1125–1132
Tang J, Kern TS (2011) Inflammation in diabetic retinopathy. Prog Retin Eye Res 30(5):343–358
Xu Y, Cheng Q, Yang B et al (2015) Increased sCD200 levels in vitreous of patients with proliferative diabetic retinopathy and its correlation with VEGF and proinflammatory cytokines. Invest Ophthalmol Vis Sci 56(11):6565–6572
Cantón A, Martinez-Cáceres EM, Hernández C, Espejo C, García-Arumí J, Simó R (2004) CD4-CD8 and CD28 expression in t cells infiltrating the vitreous fluidin patients with proliferative diabetic retinopathy: A flow cytometric analysis. Arch Ophthalmol 122(5):743–749
Lee M-W, Lee J-W, Lee K-H, Lee Y-H, Kim J-Y (2022) Peripapillary RNFL/vessel density ratio in patients with type2 diabetes without clinical diabetic retinopathy. Sci Rep 12(1):9463
Simo R, Hernández C (2014) Neurodegeneration in the diabetic eye: new insights and therapeutic perspectives. Trends Endocrinol Metab 25(1):23–33
Group UPDS (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). The lancet 352(9131):837–853
Control D, Group CTR (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329(14):977–986
for the Diabetes TWT, Control, Interventions CTEoD, Group CR (2003) Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of diabetes interventions and complications (EDIC) study. JAMA J Am Med Assoc, 290(16): 2159
Author information
Authors and Affiliations
Contributions
Design and conduct of the study (M.W.L., J.T.K.); Collection of data (J.T.K., H.J.S.); Analysis and interpretation of data (M.W.L., K.Y.N.); Writing the article (M.W.L., J.T.K.); Critical revision of the article (M.W.L., K.Y.N.); Final approval of the article (M.W.L., J.T.K., H.J.S., K.Y.N.)
Corresponding author
Ethics declarations
Conflict of interest
No authors have any financial/conflicting interests to disclose.
Human and animal rights
This is study was performed in accordance to the tenets of the Declaration of Helsinki and approved by the Institutional Review Board/Ethics Committee of Konyang University Hospital, Daejeon, Republic of Korea (No. 2023-05-028). This article does not contain any studies with animals performed by any of the authors.
Informed consent
The requirement for informed consent was waived by the Institutional Review Board/Ethics Committee of Konyang University Hospital due to the retrospective nature of the study.
Additional information
Managed by Massimo Federici.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, MW., Kim, JT., Seong, HJ. et al. Peripapillary microvasculature and retinal nerve fiber layer damage according to the severity of diabetic retinopathy. Acta Diabetol (2024). https://doi.org/10.1007/s00592-024-02308-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00592-024-02308-y