Abstract
Key message
Two large-effect QTLs and 14 candidate genes conferring resistance to Marssonina leaf spot disease were identified in an F1Populus deltoides pedigree, which provided valuable information for cloning the particular underlying genes in future.
Abstract
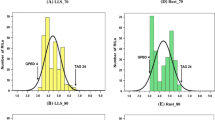
Marssonina leaf spot disease (MLSD), which is caused by Marssonina brunnea, is a devastating threat to poplar plantations. To map quantitative trait loci (QTLs) underlying resistance to MLSD, an F1P. deltoides pedigree has been established and genetic maps were constructed for the map** parents according to the two-way pseudo-testcross map** strategy. The female map contained 913 markers spanning a total genetic distance of 3132 cM, with linkage groups (LGs) corresponding to the 19 haploid chromosomes in poplar, whereas the paternal map contained 252 markers distributed on 22 LGs and covered a genetic length of 1809 cM. The established maps were further aligned to the poplar consensus genetic map based on the integrated SSR markers. The resistance to MLSD was recorded as a complex binary trait based on the black spot symptom on leaves. Analyses of QTLs revealed two large-effect QTLs in LGs VI and XVI, namely qMLSD-VI-1 and qMLSD-XVI-2, which explained 50.3% and 34.5% of the total phenotypic variance, respectively. A significant interaction between these two QTLs was detected based on a two-way ANOVA. In this map** pedigree, the female parent contributed all of the QTL alleles conferring resistance to M. brunnea. Genome sequences in the target regions were obtained by aligning the QTL intervals to the poplar genome sequence with the mapped SSR markers. The importance and utility of the 14 candidate genes associated with disease resistance identified in the QTL intervals should be more thoroughly characterized in future studies.

Similar content being viewed by others
References
Alheit KV, Reif JC, Maurer HP, Hahn V, Weissmann EA, Miedaner T, Würschum T (2011) Detection of segregation distortion loci in triticale (× Triticosecale Wittmack) based on a high-density DArT marker consensus genetic linkage map. BMC Genom 12:380. https://doi.org/10.1186/1471-2164-12-380
Anderson JP, Badruzsaufari E, Schenk PM, Manners JM, Desmond OJ, Ehlert C, Maclean DJ, Ebert PR, Kazan K (2004) Antagonistic interaction between abscisic acid and jasmonate-ethylene signaling pathways modulates defense gene expression and disease resistance in Arabidopsis. Plant Cell 16:3460–3479. https://doi.org/10.1105/tpc.104.025833
Cervera MT, Storme V, Ivens B, Gusmao J, Liu BH, Hostyn V, Van Slycken J, Van Montagu M, Boerjan W (2001) Dense genetic linkage maps of three Populus species (Populus deltoides, P. nigra and P. trichocarpa) based on AFLP and microsatellite markers. Genetics 158:787–809
Charlesworth D, Charlesworth B (1987) Inbreeding depression and its evolutionary consequences. Ann Rev Ecol Syst 18:237–268. https://doi.org/10.1146/annurev.es.18.110187.001321
de Torres-Zabala M, Truman W, Bennett MH, Lafforgue G, Mansfield JW, Egea PR, Bögre L, Grant M (2007) Pseudomonas syringae pv. tomato hijacks the Arabidopsis abscisic acid signaling pathway to cause disease. Embo J 26:1434–1443. https://doi.org/10.1038/sj.emboj.7601575
Doerge RW, Churchill GA (1996) Permutation tests for multiple loci affecting a quantitative character. Genetics 142:285–294
Dowkiw A, Bastien C (2007) Presence of defeated qualitative resistance genes frequently has major impact on quantitative resistance to Melampsora larici-populina leaf rust in P. × interamericana hybrid poplars. Tree Genet Genomes 3:261–274. https://doi.org/10.1007/s11295-006-0062-0
Elberse IAM, Vanhala TK, Turin JHB, Stam P, van Damme JMM, van Tienderen PH (2004) Quantitative trait loci affecting growth-related traits in wild barley (Hordeum spontaneum) grown under different levels of nutrient supply. Heredity 93:22–33. https://doi.org/10.1038/sj.hdy.6800467
Fishman L, Kelly AJ, Morgan E, Willis JH (2001) A genetic map in the Mimulus guttatus species complex reveals transmission ratio distortion due to heterospecific interactions. Genetics 159:1701–1716
Gaudet M, Jorge V, Paolucci I, Beritognolo I, Scarascia Mugnozza G, Sabatti M (2008) Genetic linkage maps of Populus nigra L. including AFLPs, SSRs, SNPs, and sex trait. Tree Genet Genomes 4:25–36. https://doi.org/10.1007/s11295-007-0085-1
Grattapaglia D, Sederoff R (1994) Genetic linkage maps of Eucalyptus grandis and Eucalyptus urophylla using a pseudo-testcross: map** strategy and RAPD markers. Genetics 137:1121–1137
Han Z, Li C, Huang M (1997) Further studies on the two specialized forms of Marssonina brunnea (ELL.& EV.) Magn. J Nan**g For Univ 21:40–44
Han Z, Li C, Huang M (1998) Comparative studies of isolates of Marssonina brunnea in China. Sci Silvae Sin 34:59–65. https://doi.org/10.3321/j.issn:1001-7488.1998.03.009
Han Z, Yin T, Li C, Huang M, Wu R (2000) Host effect on genetic variation of Marssonina brunnea pathogenic to poplars. Theor Appl Genet 100:614–620. https://doi.org/10.1007/s001220050081
Hartl DL (1974) Genetic dissection of segregation distortion. I. Suicide combinations of SD genes. Genetics 76:477–486
He W, Yang W (1991) Host range and distribution of three Marssonina species pathogenic to poplars in part region of China. Sci Silvae Sin 27:560–564
Jain SK, Singh P (2000) Economic analysis of industrial agroforestry: poplar (Populus deltoides) in Uttar Pradesh (India). Agrofor Syst 49:255–273. https://doi.org/10.1023/A:1006388128069
Kartesz JT, Meacham CA (1999) Synthesis of the North American flora. North Carolina Botanical Garden, University of North Carolina at Chapel Hill
Kuang H, Richardson T, Carson S, Wilcox P, Bongarten B (1999) Genetic analysis of inbreeding depression in plus tree 850.55 of Pinus radiata D. Don. I. Genetic map with distorted markers. Theor Appl Genet 98:697–703. https://doi.org/10.1007/s001220051123
Lander ES, Green P, Abrahamson J, Barlow A, Daly MJ, Lincoln SE, Newburg L (1987) MAPMAKER: an interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1:174–181. https://doi.org/10.1016/0888-7543(87)90010-3
Lange K, Boehnke M (1982) How many polymorphic genes will it take to span the human genome? Am J Hum Genet 34:842–845
Li C (1984) Two specialized forms of Marssonina populi (Lib.) Magn. J Nan**g For Univ 8:10–17
Li X, Quigg RJ, Zhou J, Xu S, Masinde G, Mohan S, Baylink DJ (2006a) A critical evaluation of the effect of population size and phenotypic measurement on QTL detection and localization using a large F2 murine map** population. Genet Mol Biol 29:166–173. https://doi.org/10.1590/S1415-47572006000100030
Li C, Zhou A, Sang T (2006b) Rice domestication by reducing shattering. Science 311:1936–1939. https://doi.org/10.1126/science.1123604
Li S, Jia J, Wei X et al (2007) A intervarietal genetic map and QTL analysis for yield traits in wheat. Mol Breed 20:167–178. https://doi.org/10.1007/s11032-007-9080-3
Liu J, Yin T, Ye N, Chen Y, Yin T, Liu M, Hassani D (2013) Transcriptome analysis of the differentially expressed genes in the male and female shrub willows (Salix suchowensis). PloS One 8:e60181. https://doi.org/10.1371/journal.pone.0060181
Luo L, Zhang Y, Xu S (2005) A quantitative genetics model for viability selection. Heredity 94:347–355. https://doi.org/10.1038/sj.hdy.6800615
Luo X, Xu N, Huang J, Gao F, Zou H, Boudsocq M, Coaker G, Liu J (2017) A lectin receptor-like kinase mediates pattern-triggered salicylic acid signaling. Plant Physiol 174:2501–2514. https://doi.org/10.1104/pp.17.00404
Merlot S, Gosti F, Guerrier D, Vavasseur A, Giraudat J (2001) The ABI1 and ABI2 protein phosphatases 2C act in a negative feedback regulatory loop of the abscisic acid signalling pathway. Plant J 25:295–303. https://doi.org/10.1046/j.1365-313x.2001.00965.x
Niderman T, Genetet I, Bruyère T, Gees R, Stintzi A, Legrand M, Fritig B, Mösinger E (1995) Pathogenesis-related PR-1 proteins are antifungal: isolation and characterization of three 14-kilodalton proteins of tomato and of a basic PR-1 of tobacco with inhibitory activity against Phytophthora infestans. Plant Physiol 108:17–27. https://doi.org/10.1104/pp.108.1.17
Oh IS, Park AR, Bae MS et al (2005) Secretome analysis reveals an Arabidopsis lipase involved in defense against Alternaria brassicicola. Plant Cell 17:2832–2847. https://doi.org/10.1105/tpc.105.034819
Orr HA, Turelli M (2001) The evolution of postzygotic isolation: accumulating Dobzhansky-Muller incompatibilities. Evolution 55:1085–1094. https://doi.org/10.1111/j.0014-3820.2001.tb00628.x
Remington DL, Whetten RW, Liu BH, O’Malley DM (1999) Construction of an AFLP genetic map with nearly complete genome coverage in Pinus taeda. Theor Appl Genet 98:1279–1292. https://doi.org/10.1007/s001220051194
Sano Y (1990) The genic nature of gamete eliminator in rice. Genetics 125:183–191
Spiers AG (1988) Comparative studies of type and herbarium specimens of Marssonina species pathogenic to poplars. Eur J For Pathol 18:140–156. https://doi.org/10.1111/j.1439-0329.1988.tb00912.x
Spiers AG (1998) Melampsora and Marssonina pathogens of poplars and willows in New Zealand. Eur J For Path 28:233–240. https://doi.org/10.1111/j.1439-0329.1998.tb01178.x
Spiers AG, Hopcroft DH (1983) Ultrastructure of conidial and microconidial ontogeny of Marssonina species pathogenic to poplars. Can J Bot 61:3529–3532. https://doi.org/10.1139/b83-398
Springer NM (2010) Isolation of plant DNA for PCR and genoty** using organic extraction and CTAB. CSH Protoc. https://doi.org/10.1101/pdb.prot5515
Stettler R, Bradshaw T, Heilman P, Hinckley T (1996) Biology of Populus and its implications for management and conservation. NRC Research Press, Ottawa
Tuskan GA, Gunter LE, Yang ZK, Yin T, Sewell MM, DiFazio SP (2004) Characterization of microsatellites revealed by genomic sequencing of Populus trichocarpa. Can J For Res 34:85–93. https://doi.org/10.1139/X03-283
Tuskan GA, DiFazio S, Jansson S et al (2006) The genome of black cottonwood, Populus trichocarpa (Torr.&Gray). Science 313:1596–1604. https://doi.org/10.1126/science.1128691
Vales MI, Schön CC, Capettini F et al (2005) Effect of population size on the estimation of QTL: a test using resistance to barley stripe rust. Theor Appl Genet 111:1260–1270. https://doi.org/10.1007/s00122-005-0043-y
Van Ooijen JW (2004) MapQTL 5, software for the map** of quantitative trait loci in experimental population. Kyazma BV, Wageningen
Vanden Broeck A, Villar M, Van Bockstaele E, Van Slycken J (2005) Natural hybridization between cultivated poplars and their wild relatives: evidence and consequences for native poplar populations. Ann For Sci 62:601–613. https://doi.org/10.1051/forest:2005072
Vogl C, Xu S (2000) Multipoint map** of viability and segregation distorting loci using molecular markers. Genetics 155:1439–1447
Voorrips RE (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93:77–78. https://doi.org/10.1093/jhered/93.1.77
Wan Z, Li Y, Liu M, Chen Y, Yin T (2015) Natural infectious behavior of the urediniospores of Melampsora larici-populina on poplar leaves. J For Res 26:225–231. https://doi.org/10.1007/s11676-015-0021-4
Wang X, Lu S (2012) Effect of marker density on QTL map** in a backcross design. J Henan Inst Sci Technol 40:32–36. https://doi.org/10.3969/j.issn.1008-7516.2012.01.008
Wang Y, Bouwmeester K, Beseh P, Shan W, Govers F (2014) Phenotypic analyses of Arabidopsis T-DNA insertion lines and expression profiling reveal that multiple L-type lectin receptor kinases are involved in plant immunity. Mol Plant-Microbe Interact 27:1390–1402. https://doi.org/10.1094/MPMI-06-14-0191-R
Wu H, Tian Y, Wan Q et al (2018) Genetics and evolution of MIXTA genes regulating cotton lint fiber development. New Phytol 217:883–895. https://doi.org/10.1111/nph.14844
Yin T, DiFazio SP, Gunter LE, Riemenschneider D, Tuskan GA (2004) Large-scale heterospecific segregation distortion in Populus revealed by a dense genetic map. Theor Appl Genet 109:451–463. https://doi.org/10.1007/s00122-004-1653-5
Yin T, DiFazio SP, Gunter LE et al (2008) Genome structure and emerging evidence of an incipient sex chromosome in Populus. Genome Res 18:422–430. https://doi.org/10.1101/gr.7076308
Yuan K, Zhang B, Zhang Y, Cheng Q, Wang M, Huang M (2008) Identification of differentially expressed proteins in poplar leaves induced by Marssonina brunnea f. sp. Multigermtubi. J Genet Genom 35:49–60. https://doi.org/10.1016/S1673-8527(08)60007-7
Zhou W, Tang Z, Hou J, Hu N, Yin T (2015) Genetic map construction and detection of genetic loci underlying segregation distortion in an intraspecific cross of Populus deltoides. PloS One 10:e0126077. https://doi.org/10.1371/journal.pone.0126077
Zhu S, Cao Y, Jiang C et al (2012) Sequencing the genome of Marssonina brunnea reveals fungus-poplar co-evolution. BMC Genom 13:382. https://doi.org/10.1186/1471-2164-13-382
Acknowledgements
The study was supported by the National Key Research and Development Plan of China (2016YFD0600101), and the National Natural Science Foundation of China (31500533 and 31570662). It was also supported by the PAPD (Priority Academic Program Development) program at Nan**g Forestry University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Francisco M. Cánovas.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, H., Wan, Z., Liu, M. et al. Map** quantitative trait loci conferring resistance to Marssonina leaf spot disease in Populus deltoides. Trees 33, 697–706 (2019). https://doi.org/10.1007/s00468-018-01809-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-018-01809-y