Abstract
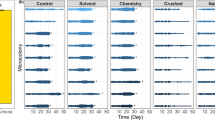
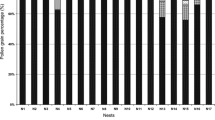
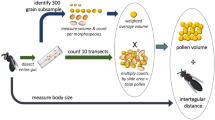
Many bee species are dietary specialists and restrict their pollen foraging to a subset of the available flowers. However, the reasons for specialization—and the reasons certain plant taxa support numerous specialists—are often unclear. Many bees specialize on the plant family Asteraceae, despite evidence its pollen is a poor food for non-specialists. Here, we studied six mason bee (Osmia) species, including three Asteraceae specialists, to test whether observed pollen-usage patterns reflect larval nutritional requirements, to investigate what aspects of Asteraceae pollen make it unsuitable for non-specialists, and to understand how Asteraceae specialists tolerate their seemingly low-quality diet. We reared larval bees on host and nonhost pollen and found that Asteraceae specialists could develop on nonhost provisions, but that other bees could not survive on Asteraceae provisions. These effects did not seem related to nutritional deficiencies, since Asteraceae provisions were not amino acid deficient, and we found no consistent differences in digestive efficiency among pollen types. However, Asteraceae specialists completed more foraging flights per larva, generally collected relatively larger provisions, and produced more frass (waste) than the other species, suggesting quantitative compensation for low food quality. Toxins, deficiencies in unmeasured nutrients, or aspects of pollen grain structure might explain poor survival of non-specialists on Asteraceae provisions. Our results suggest that floral host selection by specialist bees is not related to optimizing larval nutrition. We recommend further investigation of host-selection behaviour in adult bees and of pollen digestion in larvae to better understand the evolution of bee–flower associations.





Similar content being viewed by others
Data availability
Data related to this manuscript are available here: https://doi.org/10.17605/OSF.IO/R4EQ3.
References
Adler LS, Fowler AE, Malfi RL, Anderson PR, Cop**er LM, Deneen PM, Lopez S, Irwin RE, Farrell IW, Stevenson PC (2020) Assessing chemical mechanisms underlying the effects of sunflower pollen on a gut pathogen in bumble bees. J Chem Ecol 46:649–658. https://doi.org/10.1007/s10886-020-01168-4
Alexander MP (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 44:117–122. https://doi.org/10.3109/10520296909063335
Alexander MP (1980) A versatile stain for pollen fungi, yeast and bacteria. Stain Technol 55:13–18. https://doi.org/10.3109/10520298009067890
Arnold SEJ, Idrovo MEP, Arias LJL, Belmain SR, Stevenson PC (2014) Herbivore defence compounds occur in pollen and reduce bumblebee colony fitness. J Chem Ecol 40:878–881. https://doi.org/10.1007/s10886-014-0467-4
Auclair JL, Jamieson CA (1948) A qualitative analysis of amino acids in pollen collected by bees. Science 108:357–358. https://doi.org/10.1126/science.108.2805.357
Ballabeni P, Wlodarczyk M, Rahier M (2001) Does enemy-free space for eggs contribute to a leaf beetle’s oviposition preference for a nutritionally inferior host plant? Funct Ecol 15:318–324. https://doi.org/10.1046/j.1365-2435.2001.00529.x
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bernays EA (2001) Neural limitations in phytophagous insects: implications for diet breadth and evolution of host affiliation. Annu Rev Entomol 46:703–727. https://doi.org/10.1146/annurev.ento.46.1.703
Bernays EA, Funk DJ (1999) Specialists make faster decisions than generalists: experiments with aphids. Proc R Soc B 266:151–156
Bernays E, Graham M (1988) On the evolution of host specificity in phytophagous arthropods. Ecology 69:886–892. https://doi.org/10.2307/1941237
Bohart GE, Youssef NN (1976) The biology and behavior of Evylaeus galpinsiae Cockerell (Hymenoptera: Halictidae). Wasmann J Biol 34:185–234
Cane JH (2016) Adult pollen diet essential for egg maturation by a solitary Osmia bee. J Insect Physiol 95:105–109. https://doi.org/10.1016/j.**sphys.2016.09.011
Cane JH, Sipes SD (2006) Characterizing floral specialization by bees: analytical methods and a revised lexicon for oligolecty. In: Waser NM, Ollerton J (eds) Plant-pollinator interactions: from specialization to generalization. University of Chicago Press, Chicago, pp 99–122
Cane JH, Griswold T, Parker FD (2007) Substrates and materials used for nesting by North American Osmia bees (Hymenoptera: Apiformes: Megachilidae). Ann Entomol Soc Am 100:350–358
Cane JH, Dobson HEM, Boyer B (2017) Timing and size of daily pollen meals eaten by adult females of a solitary bee (Nomia melanderi) (Apiformes: Halictidae). Apidologie 48:17–30. https://doi.org/10.1007/s13592-016-0444-8
Cornell HV, Hawkins BA (2003) Herbivore responses to plant secondary compounds: a test of phytochemical coevolution theory. Am Nat 161:507–522. https://doi.org/10.1086/368346
Couture JJ, Mason CJ, Habeck CW, Lindroth RL (2016) Behavioral and morphological responses of an insect herbivore to low nutrient quality are inhibited by plant chemical defenses. Arthropod Plant Interact 10:341–349. https://doi.org/10.1007/s11829-016-9439-7
Cripps C, Rust RW (1989a) Pollen foraging in a community of Osmia bees (Hymenoptera: Megachilidae). Environ Entomol 18:582–589. https://doi.org/10.1093/ee/18.4.582
Cripps C, Rust RW (1989b) Pollen preferences of seven Osmia species (Hymenoptera: Megachilidae). Environ Entomol 18:133–138. https://doi.org/10.1093/ee/18.1.133
Darwin C (1859) On the origin of the species by means of natural selection. J. Murray, London
De Groot AP (1953) Protein and amino acid requirements of the honey bee (Apis mellifica L.). Physiol Comp Oecol 3:197–285
Denno RF, Larsson S, Olmstead KL (1990) Role of enemy-free space and plant quality in host-plant selection by willow beetles. Ecology 71:124–137. https://doi.org/10.2307/1940253
Detzel A, Wink M (1993) Attraction, deterrence or intoxication of bees (Apis mellifera) by plant allelochemicals. Chemoecology 4:8–18. https://doi.org/10.1007/BF01245891
Dharampal PS, Carlson C, Currie CR, Steffan SA (2019) Pollen-borne microbes shape bee fitness. Proc R Soc B 286:20182894. https://doi.org/10.1098/rspb.2018.2894
Dobson HEM, Peng Y-S (1997) Digestion of pollen components by larvae of the flower-specialist bee Chelostoma florisomne (Hymenoptera: Megachilidae). J Insect Physiol 43:89–100. https://doi.org/10.1016/s0022-1910(96)00024-8
Eby P (1998) An analysis of diet specialization in frugivorous Pteropus poliocephalus (Megachiroptera) in Australian subtropical rainforest. Aust J Ecol 23:443–456. https://doi.org/10.1111/j.1442-9993.1998.tb00752.x
Engel P, Martinson VG, Moran NA (2012) Functional diversity within the simple gut microbiota of the honey bee. Proc Natl Acad Sci 109:11002–11007. https://doi.org/10.1073/pnas.1202970109
Feder JL (1995) The effects of parasitoids on sympatric host races of Rhagoletis pomonella (Diptera: Tephritidae). Ecology 76:801–813. https://doi.org/10.2307/1939346
Forister ML, Dyer LA, Singer MS, Stireman JO 3rd, Lill JT (2012) Revisiting the evolution of ecological specialization, with emphasis on insect–plant interactions. Ecology 93:981–991. https://doi.org/10.1890/11-0650.1
Forrest JRK, Chisholm SPM (2017) Direct benefits and indirect costs of warm temperatures for high-elevation populations of a solitary bee. Ecology 98:359–369. https://doi.org/10.1002/ecy.1655
Franzén M, Larsson M (2007) Pollen harvesting and reproductive rates in specialized solitary bees. Ann Zool Fennici 44:405–414
Futuyma DJ, Moreno G (1988) The evolution of ecological specialization. Annu Rev Ecol Syst 19:207–233. https://doi.org/10.1146/annurev.es.19.110188.001231
Gathmann A, Tscharntke T (2002) Foraging ranges of solitary bees. J Anim Ecol 71:757–764. https://doi.org/10.1046/j.1365-2656.2002.00641.x
Giacomini JJ, Leslie J, Tarpy DR, Palmer-Young EC, Irwin RE, Adler LS (2018) Medicinal value of sunflower pollen against bee pathogens. Sci Rep 8:14394. https://doi.org/10.1038/s41598-018-32681-y
Guirguis GN, Brindley WA (1974) Insecticide susceptibility and response to selected pollens of larval alfalfa leafcutting bees, Megachile pacifica (Panzer) (Hymenoptera: Megachilidae). Environ Entomol 3:691–694. https://doi.org/10.1093/ee/3.4.691
Haider M, Dorn S, Müller A (2014) Better safe than sorry? A Fabaceae species exhibits unfavourable pollen properties for develo** bee larvae despite its hidden anthers. Arthropod Plant Interact 8:221–231. https://doi.org/10.1007/s11829-014-9299-y
Hardy NB, Kaczvinsky C, Bird G, Normark BB (2020) What we don’t know about diet-breadth evolution in herbivorous insects. Annu Rev Ecol Evol Syst 51:103–122. https://doi.org/10.1146/annurev-ecolsys-011720-023322
Heinrikson RL, Meredith SC (1984) Amino acid analysis by reverse-phase high-performance liquid chromatography: precolumn derivatization with phenylisothiocyanate. Anal Biochem 136:65–74. https://doi.org/10.1016/0003-2697(84)90307-5
Herbert EW, Bickley WE, Shimanuki H (1970) The brood-rearing capability of caged honey bees fed dandelion and mixed pollen diets. J Econ Entomol 63:215–218. https://doi.org/10.1093/jee/63.1.215
Hervé M (2020) RVAideMemoire: testing and plotting procedures for biostatistics. R package version 0.9-75. https://CRAN.R-project.org/package=RVAideMemoire. Accessed 13 Apr 2020
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. https://doi.org/10.1002/bimj.200810425
Human H, Nicolson SW, Strauss K, Pirk CWW, Dietemann V (2007) Influence of pollen quality on ovarian development in honeybee workers (Apis mellifera scutellata). J Insect Physiol 53:649–655. https://doi.org/10.1016/j.**sphys.2007.04.002
Hurd PD Jr, LaBerge WE, Linsley EG (1980) Principal sunflower bees of North America with emphasis on the Southwestern United States (Hymenoptera, Apoidea). Smithson Contrib Zool 310:1–158. https://doi.org/10.5479/si.00810282.310
Jaenike J (1978) On optimal oviposition behavior in phytophagous insects. Theor Popul Biol 14:350–356. https://doi.org/10.1016/0040-5809(78)90012-6
Jaenike J (1990) Host specialization in phytophagous insects. Annu Rev Ecol Syst 21:243–273. https://doi.org/10.1146/annurev.es.21.110190.001331
Kendall LK, Rader R, Gagic V, Cariveau DP, Albrecht M, Baldock KCR, Freitas BM, Hall M, Holzschuh A, Molina FP, Morten JM, Pereira JS, Portman ZM, Roberts SPM, Rodriguez J, Russo L, Sutter L, Vereecken NJ, Bartomeus I (2019) Pollinator size and its consequences: robust estimates of body size in pollinating insects. Ecol Evol 9:1702–1714. https://doi.org/10.1002/ece3.4835
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest package: tests in linear mixed effects models. J Stat Softw 82:1–26. https://doi.org/10.18637/jss.v082.i13
Lee FJ, Rusch DB, Stewart FJ, Mattila HR, Newton ILG (2015) Saccharide breakdown and fermentation by the honey bee gut microbiome. Environ Microbiol 17:796–815. https://doi.org/10.1111/1462-2920.12526
Levin MD, Haydak MH (1957) Comparative value of different pollens in the nutrition of Osmia lignaria. Bee World 38:221–226. https://doi.org/10.1080/0005772X.1957.11095007
Mayhew PJ (2001) Herbivore host choice and optimal bad motherhood. Trends Ecol Evol 16:165–167. https://doi.org/10.1016/S0169-5347(00)02099-1
McAulay MK, Forrest JRK (2019) How do sunflower pollen mixtures affect survival of queenless microcolonies of bumblebees (Bombus impatiens)? Arthropod Plant Interact 13:517–529. https://doi.org/10.1007/s11829-018-9664-3
Michener CD (2000) The bees of the world. Johns Hopkins University Press, Baltimore
Minckley RL, Roulston TH (2006) Incidental mutualisms and pollen specialization among bees. In: Waser NM, Ollerton J (eds) Plant-pollinator interactions: from specialization to generalization. University of Chicago Press, Chicago, pp 69–98
Minckley RL, Wcislo WT, Yanega D, Buchmann SL (1994) Behavior and phenology of a specialist bee (Dieunomia) and sunflower (Helianthus) pollen availability. Ecology 75:1406–1419. https://doi.org/10.2307/1937464
Müller A (1996) Host-plant specialization in western palearctic anthidiine bees (Hymenoptera: Apoidea: Megachilidae). Ecol Monogr 66:235–257. https://doi.org/10.2307/2963476
Müller A, Kuhlmann M (2008) Pollen hosts of western palaearctic bees of the genus Colletes (Hymenoptera: Colletidae): the Asteraceae paradox. Biol J Linn Soc 95:719–733. https://doi.org/10.1111/j.1095-8312.2008.01113.x
Müller A, Diener S, Schnyder S, Stutz K, Sedivy C, Dorn S (2006) Quantitative pollen requirements of solitary bees: implications for bee conservation and the evolution of bee-flower relationships. Biol Conserv 130:604–615. https://doi.org/10.1016/j.biocon.2006.01.023
Nation JL (2008) Insect physiology and biochemistry, 2nd edn. CRC Press, Boca Raton
Neff JL (2008) Components of nest provisioning behavior in solitary bees (Hymenoptera: Apoidea). Apidologie 39:30–45. https://doi.org/10.1051/apido:2007055
Nicolson SW, Human H (2013) Chemical composition of the “low quality” pollen of sunflower (Helianthus annuus, Asteraceae). Apidologie 44:144–152. https://doi.org/10.1007/s13592-012-0166-5
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2019) Vegan: community ecology package. R package version 2.5-6. https://CRAN.R-project.org/package=vegan. Accessed 13 Apr 2020
Peng Y-S, Nasr ME, Marston JM, Fang Y (1985) The digestion of dandelion pollen by adult worker honeybees. Physiol Entomol 10:75–82. https://doi.org/10.1111/j.1365-3032.1985.tb00021.x
Praz CJ, Müller A, Dorn S (2008a) Specialized bees fail to develop on non-host pollen: do plants chemically protect their pollen? Ecology 89:795–804. https://doi.org/10.1890/07-0751.1
Praz CJ, Müller A, Dorn S (2008b) Host recognition in a pollen-specialist bee: evidence for a genetic basis. Apidologie 39:547–557. https://doi.org/10.1051/apido:2008034
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 9 Oct 2019
Rayner CJ, Langridge DF (1985) Amino acids in bee-collected pollens from Australian indigenous and exotic plants. Aust J Exp Agric 25:722–726. https://doi.org/10.1071/EA9850722
Regali A, Rasmont P (1995) Nouvelles méthodes de test pour l’évaluation du régime alimentaire chez des colonies orphelines de Bombus terrestris (L) (Hymenoptera, Apidae). Apidologie 26:273–281. https://doi.org/10.1051/apido:19950401 (in French)
Rivest S, Forrest JRK (2020) Defence compounds in pollen: why do they occur and how do they affect the ecology and evolution of bees? New Phytol 225:1053–1064. https://doi.org/10.1111/nph.16230
Robertson C (1925) Heterotropic bees. Ecology 6:412–436. https://doi.org/10.2307/1929107
Roubik DW (1982) Obligate necrophagy in a social bee. Science 217:1059–1060
Roulston TH, Cane JH (2000) Pollen nutritional content and digestibility for animals. Plant Syst Evol 222:187–209. https://doi.org/10.1007/BF00984102
Roulston TH, Cane JH, Buchmann SL (2000) What governs protein content of pollen: pollinator preferences, pollen-pistil interactions, or phylogeny? Ecol Monogr 70:617–643. https://doi.org/10.1890/0012-9615(2000)070[0617:WGPCOP]2.0.CO;2
Ruedenauer FA, Raubenheimer D, Kessner-Beierlein D, Grund-Mueller N, Noack L, Spaethe J, Leonhardt SD (2020) Best be(e) on low fat: linking nutrient perception, regulation and fitness. Ecol Lett 23:545–554. https://doi.org/10.1111/ele.13454
Rust RW (1974) The systematics and biology of the genus Osmia, subgenera Osmia, Chalcosmia, and Cephalosmia (Hymenoptera: Megachilidae). Wasmann J Biol 32:1–93
Rust RW, Clement SL (1972) The biology of Osmia glauca and Osmia nemoris (Hymenoptera: Megachilidae). J Kansas Entomol Soc 45:523–528
Scheirs J, De Bruyn L, Verhagen R (2000) Optimization of adult performance determines host choice in a grass miner. Proc R Soc B 267:2065–2069. https://doi.org/10.1098/rspb.2000.1250
Sedivy C, Praz CJ, Müller A, Widmer A, Dorn S (2008) Patterns of host-plant choice in bees of the genus Chelostoma: the constraint hypothesis of host-range evolution in bees. Evolution 62:2487–2507. https://doi.org/10.1111/j.1558-5646.2008.00465.x
Sedivy C, Müller A, Dorn S (2011) Closely related pollen generalist bees differ in their ability to develop on the same pollen diet: evidence for physiological adaptations to digest pollen. Funct Ecol 25:718–725. https://doi.org/10.1111/j.1365-2435.2010.01828.x
Sedivy C, Piskorski R, Müller A, Dorn S (2012) Too low to kill: concentration of the secondary metabolite ranunculin in buttercup pollen does not affect bee larval survival. J Chem Ecol 38:996–1002. https://doi.org/10.1007/s10886-012-0153-3
Simon MP, Toft CA (1991) Diet specialization in small vertebrates: mite-eating in frogs. Oikos 61:263–278. https://doi.org/10.2307/3545344
Sipes SD, Tepedino VJ (2005) Pollen-host specificity and evolutionary patterns of host switching in a clade of specialist bees (Apoidea: Diadasia). Biol J Linn Soc 86:487–505. https://doi.org/10.1111/j.1095-8312.2005.00544.x
Spear DM, Silverman S, Forrest JRK (2016) Asteraceae pollen provisions protect Osmia mason bees (Hymenoptera: Megachilidae) from brood parasitism. Am Nat 187:797–803. https://doi.org/10.1086/686241
Steffan SA, Dharampal PS, Danforth BN, Gaines-Day HR, Takizawa Y, Chikaraishi Y (2019) Omnivory in bees: elevated trophic positions among all major bee families. Am Nat 194:414–421. https://doi.org/10.1086/704281
Stensrud MJ, Hernán MA (2020) Why test for proportional hazards? JAMA 323:1401–1402. https://doi.org/10.1001/jama.2020.1267
Strickler K (1979) Specialization and foraging efficiency of solitary bees. Ecology 60:998–1009. https://doi.org/10.2307/1936868
Suárez-Cervera M, Marquez J, Bosch J, Seoane-Camba J (1994) An ultrastructural study of pollen grains consumed by larvae of Osmia bees (Hymenoptera, Megachilidae). Grana 33:191–204. https://doi.org/10.1080/00173139409429000
Taniguchi S (1956) Biological studies on the Japanese bees (III): request in flower-visiting of infrasocial bees. Sci Rep Hyogo Univ Agric Ser Agric Biol 2:37–51. https://doi.org/10.24546/81005994
Tasei J-N, Aupinel P (2008) Nutritive value of 15 single pollens and pollen mixes tested on larvae produced by bumblebee workers (Bombus terrestris, Hymenoptera: Apidae). Apidologie 39:397–409. https://doi.org/10.1051/apido:2008017
Therneau TM (2019) Survival: survival analysis. R package version 2.44-1.1. https://CRAN.R-project.org/package=survival. Accessed 9 Oct 2019
Thorp RW (1969) Systematics and ecology of bees of the subgenus Diandrena (Hymenoptera: Andrenidae). Univ Calif Publ Entomol 52:1–146
Torchio PF (1989) In-nest biologies and development of immature stages of three Osmia species (Hymenoptera: Megachilidae). Ann Entomol Soc Am 82:599–615. https://doi.org/10.1093/aesa/82.5.599
Trunz V (2017) Phylogenetics as a tool for evolutionary studies: evolution of the leafcutter bees and chemical ecology of bee-flower relationships. PhD dissertaton, Faculté des sciences, Université de Neuchâtel, Neuchâtel, Switzerland
Turner V (1984) Banksia pollen as a source of protein in the diet of two Australian marsupials Cercartetus nanus and Tarsipes rostratus. Oikos 43:53–61. https://doi.org/10.2307/3544245
Vanderplanck M, Moerman R, Rasmont P, Lognay G, Wathelet B, Wattiez R, Michez D (2014) How does pollen chemistry impact development and feeding behavior of polylectic bees? PLoS ONE 9:e86209. https://doi.org/10.1371/journal.pone.0086209
Vanderplanck M, Vereecken NJ, Grumiau L, Esposito F, Lognay G, Wattiez R, Michez D (2017) The importance of pollen chemistry in evolutionary host shifts of bees. Sci Rep 7:43058. https://doi.org/10.1038/srep43058
Vanderplanck M, Decleves S, Roger N, Decroo C, Caulier G, Glauser G, Gerbaux P, Lognay G, Richel A, Escaravage N, Michez D (2018) Is non-host pollen suitable for generalist bumblebees? Insect Sci 25:259–272. https://doi.org/10.1111/1744-7917.12410
Vaudo AD, Tooker JF, Grozinger CM, Patch HM (2015) Bee nutrition and floral resource restoration. Curr Opin Insect Sci 10:133–141. https://doi.org/10.1016/j.cois.2015.05.008
Vaudo AD, Patch HM, Mortensen DA, Tooker JF, Grozinger CM (2016) Macronutrient ratios in pollen shape bumbe bee (Bomus impatiens) foraging strategies and floral preferences. Proc Natl Acad Sci 113:E4035-E4042. https://doi.org/10.1073/pnas.1606101113
Vaudo AD, Tooker JF, Patch HM, Biddinger DJ, Coccia M, Crone MK, Fiely M, Francis JS, Hines HM, Hodges M, Jackson SW, Michez D, Mu J, Russo L, Safari M, Treanore ED, Vanderplanck M, Yip E, Leonard AS, Grozinger CM (2020) Pollen protein: lipid macronutrient ratios may guide broad patterns of bee species floral preferences. Insects 11:132. https://doi.org/10.3390/insects11020132
Voulgari-Kokota A, Ankenbrand MJ, Grimmer G, Steffan-Dewenter I, Keller A (2019a) Linking pollen foraging of megachilid bees to their nest bacterial microbiota. Ecol Evol 9:10788–10800. https://doi.org/10.1002/ece3.5599
Voulgari-Kokota A, McFrederick QS, Steffan-Dewenter I, Keller A (2019b) Drivers, diversity, and functions of the solitary-bee microbiota. Trends Microbiol 27:1034–1044. https://doi.org/10.1016/j.tim.2019.07.011
Wang X-Y, Tang J, Wu T, Wu D, Huang S-Q (2019) Bumblebee rejection of toxic pollen facilitates pollen transfer. Curr Biol 29:1401–1406. https://doi.org/10.1016/j.cub.2019.03.023
Wcislo WT, Cane JH (1996) Floral resource utilization by solitary bees (Hymenoptera:Apoidea) and exploitation of their stored foods by natural enemies. Annu Rev Entomol 41:257–286. https://doi.org/10.1146/annurev.en.41.010196.001353
Weiner CN, Hilpert A, Werner M, Linsenmair KE, Blüthgen N (2010) Pollen amino acids and flower specialisation in solitary bees. Apidologie 41:476–487. https://doi.org/10.1051/apido/2009083
White JA, Hart RJ, Fry JC (1986) An evaluation of the Waters Pico-Tag system for the amino-acid analysis of food materials. J Autom Chem 8:170–177. https://doi.org/10.1155/S1463924686000330
Wille H, Wille M, Kilchenmann V, Imdorf A, Bühlmann G (1985) Pollenernte und massenwechsel von drei Apis mellifera-völkern auf demselben bienenstand in zwei aufeinanderfolgenden jahren. Rev Suisse Zool 92:897–914 (in German)
Williams NM (2003) Use of novel pollen species by specialist and generalist solitary bees (Hymenoptera: Megachilidae). Oecologia 134:228–237. https://doi.org/10.1007/s00442-002-1104-4
Acknowledgements
We thank Shang-Yao Lin, Abbie Hudson, Charlotte Cahill, and Christopher Schuller for their help with data collection, and two anonymous reviewers for comments that helped improve the manuscript. Natalia Libreros helped prepare and analyse bee frass samples. The Rocky Mountain Biological Laboratory provided research facilities, including access to field sites, growth chambers (funded by National Science Foundation grant DBI 1315705), housing, and funding, through a RMBL Graduate Fellowship to MKM and a RMBL Fellowship in memory of Dr. Navjot Sodhi and his contribution to Conservation Biology to JF. We received additional funding from the Natural Sciences and Engineering Research Council of Canada (MKM, JF), the Ontario Council on Graduate Studies (MKM), the National Science Foundation (SZK, through NSF grant DBI 1262713 to RMBL), and the University of Ottawa (MKM).
Author information
Authors and Affiliations
Contributions
MKM, JRKF and SZK conceived, designed and conducted the study. MKM and SZK analyzed the data with guidance from JRKF. MKM prepared the manuscript. JRKF and SZK provided editorial advice.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Roland A. Brandl.
Our study goes beyond past studies of pollen specialization in bees by examining mechanisms underlying bee survival on different pollens and by studying foraging behaviour of the bees in the field.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McAulay, M.K., Killingsworth, S.Z. & Forrest, J.R.K. Understanding pollen specialization in mason bees: a case study of six species. Oecologia 195, 559–574 (2021). https://doi.org/10.1007/s00442-020-04786-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04786-7