Abstract
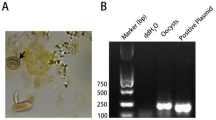
Toxoplasma gondii infection is a worldwide parasitic zoonosis with a high-health risk for humans. The key epidemiological role played by felids is related to oocyst shedding. The present study compared two amplification protocols for the molecular diagnosis of Toxoplasma infection in owned cats. A total of 78 owned cats referred to an Italian university-teaching hospital and exposed to various T. gondii-associated risk factors were sampled for blood and faeces. Faecal specimens were processed by flotation and tested using 2 copro-PCRs targeting the widely used B1 gene and the 529-bp repetitive element (RE). The sera were tested by the indirect immunofluorescent antibody test (IFAT) for the detection of immunoglobulins against T. gondii. Sixteen faeces (20.52%) tested positive for T. gondii DNA; 12 samples were positive only at B1-PCR, two at 529-bp RE-PCR and two at both genetic targets (overall agreement = 82.11%). The amplicons obtained were sequenced, and the Basic Local Alignment Search Tool analysis showed a high homology with the T. gondii strains available in reference databases. Two stool samples were microscopically positive for T. gondii-like oocysts and also tested positive by both B1 and 529-bp RE-PCRs. Thirty-three (42.3%) sera tested positive for antibodies; of which, seven were found to have T. gondii DNA-positive results using the B1 genetic target (overall agreement = 57.77%). The amplification sets targeting B1 and 529-bp RE showed substantially different yields. Further research is needed to better understand the significance and the sensitivities of using these multi-copy-targeted molecular methods from cat faeces before being used for routine diagnosis.
Similar content being viewed by others
References
Barutzki D, Schaper R (2003) Endoparasites in dogs and cats in Germany 1999–2002. Parasitol Res 90(Suppl 3):S148–S150
Belaz S, Gangneux JP, Dupretz P, Guiguen C, Gangneux FR (2015) A 10-year retrospective comparison of two target sequences, REP-529 and B1, for Toxoplasma gondii detection by quantitative PCR. J Clin Microbiol 53(4):1294–1300
Berger-Schoch AE, Herrmann DC, Schares G, Muller N, Bernet D, Gottstein B, Frey CF (2011) Prevalence and genotypes of Toxoplasma gondii in feline faeces (oocysts) and meat from sheep, cattle and pigs in Switzerland. Vet Parasitol 177(3–4):290–297
Burg JL, Grover CM, Pouletty P, Boothroyd JC (1989) Direct and sensitive detection of a pathogenic protozoan, Toxoplasma gondii, by polymerase chain reaction. J Clin Microbiol 27(8):1787–1792
Cenci-Goga BT, Rossitto PV, Sechi P, McCrindle CM, Cullor JS (2011) Toxoplasma in animals, food, and humans: an old parasite of new concern. Foodborne Pathog Dis 8:751–762
Chemoh W, Sawangjaroen N, Nissapatorn V, Sarmwittayawon N (2016) Molecular investigation on the occurrence of Toxoplasma gondii oocysts in cat faeces using TOX-element and ITS-1 region targets. Vet J 215:118–122
Cook AJ, Gilbert RE, Buffolano W, Zufferey J, Petersen E, Jenum PA, Foulon W, Semprini AE, Dunn DT (2000) Sources of Toxoplasma infection in pregnant women: European multicentre case-control study. BMJ 321(7254):142–147
Dabritz HA, Conrad PA (2010) Cats and Toxoplasma implications for public health. Zoonoses Public Health 57:34–52
Dabritz HA, Gardner IA, Miller MA, Lappin MR, Atwill ER, Packham AE, Melli AC, Conrad PA (2007) Evaluation of two Toxoplasma gondii serological tests used in a serosurvey of domestic cats in California. J Parasitol 93(4):806–816
Davis SW, Dubey JP (1995) Mediation of immunity to Toxoplasma gondii oocyst shedding in cats. J Parasitol 81(6):882–886
Dryden MW, Payne PA, Ridley R, Smith V (2005) Comparison of common fecal flotation techniques for the recovery of parasite eggs and oocysts. Vet Ther 6(1):15–28
Dubey JP (2010) Toxoplasmosis of animals and humans. CRC Press Inc, Boca Raton, FL
Dubey JP, Darrington C, Tiao N, Ferreira L, Choudhary S, Molla B, Saville W, Tilahun G, Kwok O, Gebreyes G (2013) Isolation of viable Toxoplasma gondii from tissues and feces of cats from Addis Ababa, Ethiopia. J Parasitol 99(1):56–58
Elmore SA, Jones JL, Conrad PA, Patton S, Lindsay DS, Dubey JP (2010) Toxoplasma gondii: epidemiology, feline clinical aspects, and prevention. Trends Parasitol 26(4):190–196
Fallahi S, Kazemi B, Seyyed Tabaei SJ, Bandehpour M, Lasjerdi Z, Taghipour N, Zebardast N, Nikmanesh B, Omrani VF, Ebrahimzadeh F (2014) Comparison of the RE and B1 gene for detection of Toxoplasma gondii infection in children with cancer. Parasitol Int 63(1):37–41
Feng XC, Milhausen M (1999) Localization of Toxoplasma gondii in feline intestinal tissue using PCR. J Parasitol 85(6):1041–1046
Homan WL, Vercammen M, De Braekeleer J, Verschueren H (2000) Identification of a 200- to 300-fold repetitive 529 bp DNA fragment in Toxoplasma gondii, and its use for diagnostic and quantitative PCR. Int J Parasitol 30(1):69–75
Kitano T, Umetsu K, Tian W, Osawa M (2007) Two universal primer sets for species identification among vertebrates. Int J Legal Med 121:423–427
Lappin MR (2010) Update on the diagnosis and management of Toxoplasma gondii infection in cats. Top Companion Anim Med 25(3):136–141
Lilly EL, Wortham CD (2013) High prevalence of Toxoplasma gondii oocyst shedding in stray and pet cats (Felis catus) in Virginia, United States. Parasit Vectors 6:266
Lin MH, Chen TC, Cuo TT, Tseng CC, Tseng CP (2000) Real-time PCR for quantitative detection of Toxoplasma gondii. J Clin Microbiol 38(11):4121–4125
Mancianti F, Nardoni S, Mugnaini L, Zambernardi L, Guerrini A, Gazzola V, Papini RA (2015) A retrospective molecular study of select intestinal protozoa in healthy pet cats from Italy. J Feline Med Surg 17(2):163–167
Okay TS, Yamamoto L, Oliveira LC, Manuli ER, Andrade Junior HF, Del Negro GM (2009) Significant performance variation among PCR systems in diagnosing congenital toxoplasmosis in São Paulo, Brazil: analysis of 467 amniotic fluid samples. Clinics (Sao Paulo) 64(3):171–176
Poulle ML, Forin-Wiart MA, Josse-Dupuis E, Villena I, Aubert D (2016) Detection of Toxoplasma gondii DNA by qPCR in the feces of a cat that recently ingested prey does not necessary imply oocyst shedding. Parasite 23:29
Reischl U, Bretagne S, Kruger D, Ernault P, Costa JM (2003) Comparison of two DNA targets for the diagnosis of toxoplasmosis by real-time PCR using fluorescence resonance energy transfer hybridization probes. BMC Infect Dis 3:7
Salant H, Marcovics A, Spira DT, Hamburger J (2007) The development of a molecular approach for coprodiagnosis of Toxoplasma gondii. Vet Parasitol 146(3–4):214–220
Salant H, Spira D, Hamburger J (2010) A comparative analysis of coprologic diagnostic methods for detection of Toxoplasma gondii in cats. AmJTrop Med Hyg 82(5):865–870
Schares G, Globokar Vhrovec M, Pantchev N, Herrmann DC, Conraths FJ (2008) Occurrence of Toxoplasma gondii and Hammondia hammondi oocysts in the faeces of cats from Germany and other European countries. Vet Parasitol 152(1–2):34–45
Switaij K, Master A, Skrzypczak M, Zaborowski P (2005) Recent trends in molecular diagnostics for Toxoplasma gondii infections. Clin Microbiol Infect 11(3):170–176
Thrusfield M (2007) Veterinary epidemiology. Blackwell Science Ltd, Oxford, United Kingdom
Veronesi F, Santoro A, Milardi GL, Diaferia M, Branciari R, Miraglia D, Cioffi A, Gabrielli S, Ranucci D (2017) Comparison of PCR assays targeting the multi-copy targets B1 gene and 529 bp repetitive element for detection of Toxoplasma gondii in swine muscle. Food Microbiol 63:213–216
Wahab T, Edvinsson B, Palm D, Lindh J (2010) Comparison of the AF146527 and B1 repeated elements, two real-time PCR targets used for detection of Toxoplasma gondii. J Clin Microbiol 48(2):591–592
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests, and that the conceptual design, conduct, interpretation of the results and all the scientific aspects of the study were not influenced by any third party.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Additional information
Fabrizia Veronesi and Azzurra Santoro contributed equally to the work.
Rights and permissions
About this article
Cite this article
Veronesi, F., Santoro, A., Milardi, G.L. et al. Detection of Toxoplasma gondii in faeces of privately owned cats using two PCR assays targeting the B1 gene and the 529-bp repetitive element. Parasitol Res 116, 1063–1069 (2017). https://doi.org/10.1007/s00436-017-5388-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5388-z