Abstract
Purpose
Treatment options for patients with relapsed and refractory multiple myeloma have improved since the introduction of immune-modulating agents such as lenalidomide and thalidomide. However, almost all patients relapse and suffer from an increasing amount of adverse events due to multiple lines of therapy that eventually lead to a reduced quality of life.
Methods
In this bicentric retrospective analysis, 58 patients who had been treated with either bendamustine monotherapy (62 % of the patients) or combined steroid therapy were included. Further inclusion criteria were at least relapsed disease. Patients had previously been treated with a mean of four lines of therapy (range 1–10). They received a median of three cycles of treatment. Dosage varied from 60 to 300 mg/m2 (median 120 mg/m2) and was administered intravenously on day 1 and 2 of a 28-day cycle.
Results
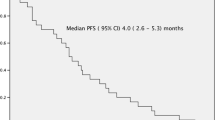
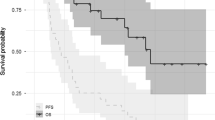
Observed toxicity was mild and most commonly led to hematological side effects such as thrombopenia and anemia. Response rates were as follows: no complete response, 20 % partial response, 39 % minimal response, 27 % stable disease and 14 % progressive disease. Median overall survival (OS) was 17 months. Median event-free survival was 7 months. Patients who had not received a concomitant steroid had a median OS of 17 months compared to 13 months median OS for patients who had received a concomitant steroid.
Conclusion
Bendamustine monotherapy is an effective treatment option for heavily pre-treated myeloma patients due to its favorable response rate and mild toxicity.


Similar content being viewed by others
References
Anderson KC, Kyle RA, Rajkumar SV, Stewart AK, Weber D, Richardson P (2008) Clinically relevant end points and new drug approvals for myeloma. Leukemia 22(2):231–239. doi:10.1038/sj.leu.2405016
Berenson JR, Yellin O, Bessudo A, Boccia RV, Noga SJ, Gravenor DS et al (2013) Phase I/II trial assessing bendamustine plus bortezomib combination therapy for the treatment of patients with relapsed or refractory multiple myeloma. Br J Haematol 160(3):321–330. doi:10.1111/bjh.12129
Durie BGM, Harousseau J-L, Miguel JS, Bladé J, Barlogie B, Anderson K et al (2006) International uniform response criteria for multiple myeloma. Leukemia 20(9):1467–1473. doi:10.1038/sj.leu.2404284
Grey-Davies E, Bosworth JL, Boyd KD, Ebdon C, Saso R, Chitnavis D et al (2012) Bendamustine, thalidomide and dexamethasone is an effective salvage regimen for advanced stage multiple myeloma. Br J Haematol 156(4):552–555. doi:10.1111/j.1365-2141.2011.08887.x
Kalaycio M (2009) Bendamustine: a new look at an old drug. Cancer 115(3):473–479. doi:10.1002/cncr.24057
Lentzsch S, O’Sullivan A, Kennedy RC, Abbas M, Dai L, Pregja SL et al (2012) Combination of bendamustine, lenalidomide, and dexamethasone (BLD) in patients with relapsed or refractory multiple myeloma is feasible and highly effective: results of phase 1/2 open-label, dose escalation study. Blood 119(20):4608–4613. doi:10.1182/blood-2011-12-395715
Ludwig H, Kasparu H, Leitgeb C, Rauch E, Linkesch W, Zojer N et al (2014) Bendamustine–bortezomib–dexamethasone is an active and well-tolerated regimen in patients with relapsed or refractory multiple myeloma. Blood 123(7):985–991. doi:10.1182/blood-2013-08-521468
Michael M, Bruns I, Bölke E, Zohren F, Czibere A, Safaian NN et al (2010) Bendamustine in patients with relapsed or refractory multiple myeloma. Eur J Med Res 15(1):13–19
Niesvizky R, Richardson PG, Rajkumar SV, Coleman M, Rosiñol L, Sonneveld P et al (2008) The relationship between quality of response and clinical benefit for patients treated on the bortezomib arm of the international, randomized, phase 3 APEX trial in relapsed multiple myeloma. Br J Haematol 143(1):46–53. doi:10.1111/j.1365-2141.2008.07303.x
Offidani M, Corvatta L, Maracci L, Liberati AM, Ballanti S, Attolico I et al (2013) Efficacy and tolerability of bendamustine, bortezomib and dexamethasone in patients with relapsed-refractory multiple myeloma: a phase II study. Blood Cancer J 3(11):e162. doi:10.1038/bcj.2013.58
Pönisch W, Mitrou PS, Merkle K, Herold M, Assmann M, Wilhelm G et al (2006) Treatment of bendamustine and prednisone in patients with newly diagnosed multiple myeloma results in superior complete response rate, prolonged time to treatment failure and improved quality of life compared to treatment with melphalan and prednisone—a randomized phase III study of the East German Study Group of Hematology and Oncology (OSHO). J Cancer Res Clin Oncol 132(4):205–212. doi:10.1007/s00432-005-0074-4
Pönisch W, Heyn S, Beck J, Wagner I, Mohren M, Hoffmann FA et al (2013) Lenalidomide, bendamustine and prednisolone exhibits a favourable safety and efficacy profile in relapsed or refractory multiple myeloma: final results of a phase 1 clinical trial OSHO-#077. Br J Haematol. doi:10.1111/bjh.12361
Rodon P, Hulin C, Pegourie B, Tiab M, Anglaret B, Benboubker L et al (2015) Phase II study of bendamustine, bortezomib and dexamethasone as second-line treatment for elderly patients with multiple myeloma: the Intergroupe Francophone du Myelome 2009-01 trial. Haematologica 100(2):e56–e59. doi:10.3324/haematol.2014.110890
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that no conflicts of interest exist. There is no conflict to study participants. There is no study sponsor.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethics approval for this retrospective study had been obtained according to the guidelines of the host institution.
Rights and permissions
About this article
Cite this article
Stöhr, E., Schmeel, F.C., Schmeel, L.C. et al. Bendamustine in heavily pre-treated patients with relapsed or refractory multiple myeloma. J Cancer Res Clin Oncol 141, 2205–2212 (2015). https://doi.org/10.1007/s00432-015-2014-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-015-2014-2