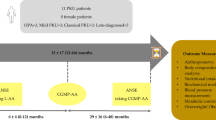
Abstract
Phenylketonuria’s (PKU) treatment based on low-protein diet may affect other metabolic pathways, such as that of asymmetric dimethylarginine (ADMA). The aim of this study was to evaluate the reliability of ADMA as a biomarker of adequate metabolic control and possible nutritional risk in a long-term PKU patient population. One hundred and six dietary-treated PKU patients from four hospitals in Spain were enrolled in this cross-sectional study. Their lipid profile, total homocysteine, ADMA, and symmetric dimethylarginine (SDMA) concentrations were analyzed and compared with a control group. Sensitivity, specificity, and likelihood ratios of the proposed biomarker were calculated. PKU patients had statistically significant lower plasmatic ADMA, SDMA, and arginine concentrations as compared with the control group (p < 0.001). Significant correlations were found between ADMA, phenylalanine, and total homocysteine levels. The ADMA/creatinine ratio correlated with phenylalanine levels as metabolic control and nutritional risk in PKU patients. Its reliability as a management biomarker was studied with positive results. The ADMA/creatinine ratio might serve as an independent biomarker in the management of PKU patients, different from blood phenylalanine levels. It could be of particular usefulness to detect those who are following an unbalanced diet that could have long-term negative effects.
Conclusion: In this study, we have evaluated the reliability of ADMA as a potential biomarker of adequate metabolic control and possible nutritional risk in a long-term PKU patient population.
What is Known: • Although PKU individuals have lower values of ADMA even with blood Phe levels in the recommended range, little attention is payed to other metabolic pathways. | |
What is New: • ADMA could be used as new biomarker for PKU management and follow-up of the diet, after evaluating their reliability in a long-term PKU patient population. |




Similar content being viewed by others
Abbreviations
- ADMA:
-
Asymmetric dimethylarginine
- Arg:
-
Arginine
- Crn:
-
Creatinine
- DDAH:
-
Dimethylarginine dimethylamino hydrolase
- HPA:
-
Hyperphenylalaninaemia
- LR:
-
Likelihood ratios
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- Phe:
-
Phenylalanine
- PKU:
-
Phenylketonuria
- PV:
-
Predictive values
- SDMA:
-
Symmetric dimethylarginine
- tHcys:
-
Total homocysteine
References
Aldámiz-Echevarría L, Andrade F (2012) Asymmetric dimethylarginine, endothelial dysfunction and renal disease. Int J Mol Sci 13:11288–11311
Andrade F, Llarena M, Lage S, Aldámiz-Echevarría L (2015) Quantification of arginine and its methylated derivatives in healthy children by liquid chromatography-tandem mass spectrometry. J Chromatogr Sci 53(5):787–792
Antoniades C, Tousoulis D, Marinou K, Vasiliadou C, Tentolouris C, Bouras G, Pitsavos C, Stefanadis C (2006) Asymmetrical dimethylarginine regulates endothelial function in methionine-induced but not in chronic homocystinemia in humans: effect of oxidative stress and proinflammatory cytokines. Am J Clin Nutr 84(4):781–788
Böger RH, Bode-Böger SM, Szuba A, Tsao PS, Chan JR, Tangphao O, Blaschke TF, Cooke JP (1998) Asymmetric dimethylarginine (ADMA): a novel risk factor for endothelial dysfunction. Circulation 98:1842–1847
Bossuyt PM (2010) Clinical validity: defining biomarker performance. Scand J Clin Lab Invest Suppl 242:46–52
Camp KM, Parisi MA, Acosta PB, Berry GT, Bilder DA, Blau N, Bodamer OA, Brosco JP, Brown CS, Burlina AB, Burton BK, Chang CS, Coates PM, Cunningham AC, Dobrowolski SF, Ferguson JH, Franklin TD, Frazier DM, Grange DK, Greene CL, Groft SC, Harding CO, Howell RR, Huntington KL, Hyatt-Knorr HD, Jevaji IP, Levy HL, Lichter-Konecki U, Lindegren ML, Lloyd-Puryear MA, Matalon K, MacDonald A, McPheeters ML, Mitchell JJ, Mofidi S, Moseley KD, Mueller CM, Mulberg AE, Nerurkar LS, Ogata BN, Pariser AR, Prasad S, Pridjian G, Rasmussen SA, Reddy UM, Rohr FJ, Singh RH, Sirrs SM, Stremer SE, Tagle DA, Thompson SM, Urv TK, Utz JR, van Spronsen F, Vockley J, Waisbren SE, Weglicki LS, White DA, Whitley CB, Wilfond BS, Yannicelli S, Young JM (2014) Phenylketonuria Scientific Review Conference: state of the science and future research needs. Mol Genet Metab 112:87–122
Cleary M, Trefz F, Muntau AC, Feillet F, van Spronsen FJ, Burlina A, Bélanger-Quintana A, Giżewska M, Gasteyger C, Bettiol E, Blau N, MacDonald A (2013) Fluctuations in phenylalanine concentrations in phenylketonuria: a review of possible relationships with outcomes. Mol Genet Metab 110(4):418–423
Colomé C, Artuch R, Lambruschini N, Cambra FJ, Campistol J, Vilaseca M (2001) Is there a relationship between plasma phenylalanine and cholesterol in phenylketonuric patients under dietary treatment? Clin Biochem 34(5):373–376
Couce ML, Vitoria I, Aldámiz-Echevarría L, Fernández-Marmiesse A, Roca I, Llarena M, Sánchez-Pintos P, Leis R, Hermida A (2016) Lipid profile status and other related factors in patients with hyperphenylalaninaemia. Orphanet J Rare Dis 11(1):123
Daelman L, Sedel F, Tourbah A (2014) Progressive neuropsychiatric manifestations of phenylketonuria in adulthood. Rev Neurol (Paris) 170(4):280–287
Donlon J, Sarkissian C, Levy H, Scriver CR (2014) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In The online metabolic and molecular bases of inherited disease (eds Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Antonarakis SE, Ballabio A, Gibson K, Mitchell G) (McGraw-Hill, New York, NY, USA) http://ommbid.mhmedical.com/content.aspx?bookid=971&Sectionid=62673211 [accessed January 23, 2017]
Feillet F, Agostoni C (2010) Nutritional issues in treating phenylketonuria. J Inherit Metab Dis 33(6):659–664
Finkelstein JD (1998) The metabolism of homocysteine: pathways and regulation. Eur J Pediatr 157:S40–S44
Hermida-Ameijeiras A, Crujeiras V, Roca I, Calvo C, Leis R, Couce ML (2017) Arterial stiffness assessment in patients with phenylketonuria. Medicine (Baltimore) 96(51):e9322
Huemer M, Simma B, Mayr D, Möslinger D, Mühl A, Schmid I, Ulmer H, Bodamer OA (2012) Free asymmetric dimethylarginine (ADMA) is low in children and adolescents with classical phenylketonuria (PKU). J Inherit Metab Dis 35(5):817–821
Kanzelmeyer N, Tsikas D, Chobanyan-Jürgens K, Beckmann B, Vaske B, Illsinger S, Das AM, Lücke T (2012) Asymmetric dimethylarginine in children with homocystinuria or phenylketonuria. Amino Acids 42(5):1765–1772
Loscalzo J (2004) L-arginine and atherothrombosis. J Nutr 34(10 Suppl):2798S–2800S discussion 2818S-2819S
Nijveldt RJ, Teerlink T, van Guldener C, Prins HA, van Lambalgen AA, Stehouwer CD, Rauwerda JA, van Leeuwen PA (2003) Handling of asymmetrical dimethylarginine and symmetrical dimethylarginine by the rat kidney under basal conditions and during endotoxaemia. Nephrol Dial Transpl 18:2542–2550
Özcan Ö, Ipcioglu OM, Gultepe M (2012) Unexpectedly low asymmetric dimethylarginine (ADMA) and homocysteine levels in patients with phenylketonuria (PKU). J Inherit Metab Dis 35(6):1153
Rocha JC, Almeida MF, Carmona C, Cardoso ML, Borges N, Soares I, Salcedo G, Lima MR, Azevedo I, van Spronsen FJ (2010) The use of prealbumin concentration as a biomarker of nutritional status in treated phenylketonuric patients. Ann Nutr Metab 56(3):207–211
Rocha JC, van Spronsen FJ, Almeida MF, Soares G, Quelhas D, Ramos E, Guimarães JT, Borges N (2012) Dietary treatment in phenylketonuria does not lead to increased risk of obesity or metabolic syndrome. Mol Genet Metab 107(4):659–663
Schulpis KH, Karakonstantakis T, Bartzeliotou A, Karikas GA, Papassotiriou I (2004) The association of serum lipids, lipoproteins and apolipoproteins with selected trace elements and minerals in phenylketonuric patients on diet. Clin Nutr 23(3):401–407
Schulpis KH, Papassotiriou I, Tsakiris S, Vounatsou M, Chrousos GP (2005) Increased plasma adiponectin concentrations in poorly controlled patients with phenylketonuria normalize with a strict diet: evidence for catecholamine-mediated adiponectin regulation and a complex effect of phenylketonuria diet on atherogenesis risk factors. Metabolism 54(10):1350–1355
Singh RH, Cunningham AC, Mofidi S, Douglas TD, Frazier DM, Hook DG, Jeffers L, McCune H, Moseley KD, Ogata B, Pendyal S, Skrabal J, Splett PL, Stembridge A, Wessel A, Rohr F (2016) Updated, web-based nutrition management guideline for PKU: an evidence and consensus based approach. Mol Genet Metab 118(2):72–83
Sirtori LR, Dutra-Filho CS, Fitarelli D, Sitta A, Haeser A, Barschak AG, Wajner M, Coelho DM, Llesuy S, Belló-Klein A et al (2005) Oxidative stress in patients with phenylketonuria. Biochim Biophys Acta 15 1740(1):68–73
Stühlinger MC, Tsao PS, Her JH, Kimoto M, Balint RF, Cooke JP (2001) Homocysteine impairs the nitric oxide synthase pathway: role of asymmetric dimethylarginine. Circulation 104:2569–2575
Ueland PM (1983) Pharmacological and biochemical aspects of S-adenosylhomocysteine and S-adenosylhomocysteine hydrolase. Pharmacol Rev 34:223–253
Vallance P, Leiper J (2004) Cardiovascular biology of the asymmetric dimethylarginine:dimethylarginine dimethylaminohydrolase pathway. Arterioscler Thromb Vasc Biol 24(6):1023–1030
van Spronsen FJ, van Wegberg AM, Ahring K, Belanger-Quintana A, Blau N, Bosch AM, Burlina A, Campistol J, Feillet F, Giżewska M et al (2017) Key European guidelines for the diagnosis and management of patients with phenylketonuria. Lancet Diabetes Endocrinol 5(9):743–756
van Wegberg AMJ, MacDonald A, Ahring K, Bélanger-Quintana A, Blau N, Bosch AM, Burlina A, Campistol J, Feillet F, Giżewska M, Huijbregts SC, Kearney S, Leuzzi V, Maillot F, Muntau AC, van Rijn M, Trefz F, Walter JH, van Spronsen FJ (2017) The complete European guidelines on phenylketonuria: diagnosis and treatment. Orphanet J Rare Dis 12(1):162
Verduci E, Banderali G, Moretti F, Lassandro C, Cefalo G, Radaelli G, Salvatici E, Giovannini M (2016) Diet in children with phenylketonuria and risk of cardiovascular disease: a narrative overview. Nutr Metab Cardiovasc Dis 26(3):171–177
Acknowledgements
We thank all patients for kindly participating in the study. This work was supported in part by NUTRICIA S.L.R. (Madrid, Spain). Research in this paper was partially financed by Biocruces Bizkaia Health Research Institute and Carlos III Health Research Institute.
Author information
Authors and Affiliations
Contributions
LAE, MLC, MAB, CA, JH, and LC designed the study, conducted the research, and corrected the final version of the manuscript. FA, OV, SNM, and PSN analyzed samples and data and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The study was approved by Clinical Research Ethics Committees at the hospitals involved and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Communicated by Peter de Winter
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 27 kb)
Rights and permissions
About this article
Cite this article
Andrade, F., Villate, O., Couce, M.L. et al. Asymmetric dimethylarginine as a potential biomarker for management and follow-up of phenylketonuria. Eur J Pediatr 178, 903–911 (2019). https://doi.org/10.1007/s00431-019-03365-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-019-03365-0