Abstract
Main conclusion
dnal7, a novel allelic variant of the OsHSP40, affects rice plant architecture and grain yield by coordinating auxins, cytokinins, and gibberellic acids.
Abstract
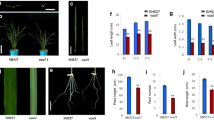
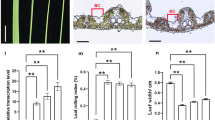
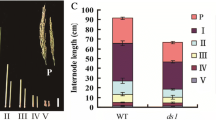
Plant height and leaf morphology are the most important traits of the ideal plant architecture (IPA), and discovering related genes is critical for breeding high-yield rice. Here, a dwarf and narrow leaf 7 (dnal7) mutant was identified from a γ-ray treated mutant population, which exhibits pleiotropic effects, including dwarfing, narrow leaves, small seeds, and low grain yield per plant compared to the wild type (WT). Histological analysis showed that the number of veins and the distance between adjacent small veins (SVs) were significantly reduced compared to the WT, indicating that DNAL7 controls leaf size by regulating the formation of veins. Map-based cloning and transgenic complementation revealed that DNAL7 is allelic to NAL11, which encodes OsHSP40, and the deletion of 2 codons in dnal7 destroyed the His-Pro-Asp (HPD) motif of OsHSP40. In addition, expression of DNAL7 in both WT and dnal7 gradually increased with the increase of temperature in the range of 27–31 °C. Heat stress significantly affected the seedling height and leaf width of the dnal7 mutant. A comparative transcriptome analysis of WT and dnal7 revealed that DNAL7 influenced multiple metabolic pathways, including plant hormone signal transduction, carbon metabolism, and biosynthesis of amino acids. Furthermore, the contents of the cytokinins in leaf blades were much higher in dnal7 than in the WT, whereas the contents of auxins were lower in dnal7. The contents of bioactive gibberellic acids (GAs) including GA1, GA3, and GA4 in shoots were decreased in dnal7. Thus, DNAL7 regulates rice plant architecture by coordinating the balance of auxins, cytokinins, and GAs. These results indicate that OsHSP40 is a pleiotropic gene, which plays an important role in improving rice yield and plant architecture.








Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article and Supplementary materials, further inquiries can be directed to the corresponding author. The raw data of RNA-seq generated are available in the Sequencing Read Archive (SRA) of NCBI (PRJNA1016282).
Abbreviations
- DAG:
-
Day after germination
- GA:
-
Gibberellic acid
- HSP:
-
Heat shock protein
References
Cho S, Yoo S, Zhang H et al (2013) The rice narrow leaf2 and narrow leaf3 loci encode WUSCHEL-related homeobox 3A (OsWOX3A) and function in leaf, spikelet, tiller and lateral root development. New Phytol 198:1071–1084. https://doi.org/10.1111/nph.12231
Davière J-M, Achard P (2013) Gibberellin signaling in plants. Development 140:1147–1151. https://doi.org/10.1242/dev.087650
Ding Z, Lin Z, Li Q et al (2015) DNL1, encodes cellulose synthase-like D4, is a major QTL for plant height and leaf width in rice (Oryza sativa L.). Biochem Biophys Res Commun 457:133–140. https://doi.org/10.1016/j.bbrc.2014.12.034
Fang Z, **a K, Yang X et al (2013) Altered expression of the PTR/NRT1 homologue OsPTR9 affects nitrogen utilization efficiency, growth and grain yield in rice. Plant Biotechnol J 11:446–458. https://doi.org/10.1111/pbi.12031
Fu**o K, Matsuda Y, Ozawa K et al (2008) NARROW LEAF 7 controls leaf shape mediated by auxin in rice. Mol Genet Genom 279:499–507. https://doi.org/10.1007/s00438-008-0328-3
Fujita D, Trijatmiko KR, Tagle AG et al (2013) NAL1 allele from a rice landrace greatly increases yield in modern indica cultivars. Proc Natl Acad Sci USA 110:20431–20436. https://doi.org/10.1073/pnas.1310790110
Guo T, Wang D, Fang J et al (2019) Mutations in the rice OsCHR4 gene, encoding a CHD3 family chromatin remodeler, induce narrow and rolled leaves with increased cuticular wax. Int J Mol Sci 20:2567. https://doi.org/10.3390/ijms20102567
Jambunathan N (2010) Determination and detection of reactive oxygen species (ROS), lipid peroxidation, and electrolyte leakage in plants. Methods Mol Biol 639:292–298. https://doi.org/10.1007/978-1-60761-702-0_18
Jiang D, Fang J, Lou L et al (2015) Characterization of a null allelic mutant of the rice NAL1 gene reveals its role in regulating cell division. PLoS One 10:e0118169. https://doi.org/10.1371/journal.pone.0118169
Kang Y, Lee K, Hoshikawa K et al (2022) Molecular bases of heat stress responses in vegetable crops with focusing on heat shock factors and heat shock proteins. Front Plant Sci 13:837152. https://doi.org/10.3389/fpls.2022.837152
Kusnandar AS, Itoh J-I, Sato Y et al (2022) NARROW AND DWARF LEAF 1, the ortholog of Arabidopsis ENHANCER OF SHOOT REGENERATION1/DORNRÖSCHEN, mediates leaf development and maintenance of the shoot apical meristem in Oryza sativa L. Plant Cell Physiol 63:265–278. https://doi.org/10.1093/pcp/pcab169
Li W, Wu C, Hu G et al (2013) Characterization and fine map** of a novel rice narrow leaf mutant nal9. J Integr Plant Biol 55:1016–1025. https://doi.org/10.1111/jipb.12098
Lin L, Zhao Y, Liu F et al (2019) Narrow leaf 1 (NAL1) regulates leaf shape by affecting cell expansion in rice (Oryza sativa L.). Biochem Biophys Res Commun 516:957–962. https://doi.org/10.1016/j.bbrc.2019.06.142
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lo S-F, Yang S-Y, Chen K-T et al (2008) A novel class of gibberellin 2-oxidases control semidwarfism, tillering, and root development in rice. Plant Cell 20:2603–2618. https://doi.org/10.1105/tpc.108.060913
Luo L, **e Y, Yu S et al (2023) The DnaJ domain-containing heat-shock protein NAL11 determines plant architecture by mediating gibberellin homeostasis in rice (Oryza sativa). New Phytol 237:2163–2179. https://doi.org/10.1111/nph.18696
Micol JL (2009) Leaf development: time to turn over a new leaf? Curr Opin Plant Biol 12:9–16. https://doi.org/10.1016/j.pbi.2008.11.001
Muradoglu F, Gundogdu M, Ercisli S et al (2015) Cadmium toxicity affects chlorophyll a and b content, antioxidant enzyme activities and mineral nutrient accumulation in strawberry. Biol Res 48:11. https://doi.org/10.1186/s40659-015-0001-3
Nakajima Y, Suzuki S (2013) Environmental stresses induce misfolded protein aggregation in plant cells in a microtubule-dependent manner. Int J Mol Sci 14:7771–7783. https://doi.org/10.3390/ijms14047771
Qi J, Qian Q, Bu Q et al (2008) Mutation of the rice Narrow leaf1 gene, which encodes a novel protein, affects vein patterning and polar auxin transport. Plant Physiol 147:1947–1959. https://doi.org/10.1104/pp.108.118778
Vinoth BV, Rajan PD (2009) Arabidopsis thaliana J-class heat shock proteins: cellular stress sensors. Funct Integr Genom 9:433–446. https://doi.org/10.1007/s10142-009-0132-0
Tamadaddi C, Verma AK, Zambare V et al (2022) J-like protein family of Arabidopsis thaliana: the enigmatic cousins of J-domain proteins. Plant Cell Rep 41:1343–1355. https://doi.org/10.1007/s00299-022-02857-y
Tsukaya H (2006) Mechanism of leaf-shape determination. Annu Rev Plant Biol 57:477–496. https://doi.org/10.1146/annurev.arplant.57.032905.105320
Uzair M, Long H, Zafar SA et al (2021) Narrow Leaf21, encoding ribosomal protein RPS3A, controls leaf development in rice. Plant Physiol 186:497–518. https://doi.org/10.1093/plphys/kiab075
Wang J, Xu H, Li N et al (2015) Artificial selection of Gn1a plays an important role in improving rice yields across different ecological regions. Rice 8:37. https://doi.org/10.1186/s12284-015-0071-4
Wang J, Wu B, Lu K et al (2019) The amino acid permease 5 (OsAAP5) regulates tiller number and grain yield in rice. Plant Physiol 180:1031–1045. https://doi.org/10.1104/pp.19.00034
Wang J, Jiang X, Zhao C et al (2020) Transcriptomic and metabolomic analysis reveals the role of CoA in the salt tolerance of Zygophyllum spp. BMC Plant Biol 20:9. https://doi.org/10.1186/s12870-019-2226-8
Wang F, Tang Z, Wang Y et al (2022) Leaf Mutant 7 encoding heat shock protein OsHSP40 regulates leaf size in rice. Int J Mol Sci 23:4446. https://doi.org/10.3390/ijms23084446
Woo Y-M, Park H-J, Suudi M et al (2007) Constitutively wilted 1, a member of the rice YUCCA gene family, is required for maintaining water homeostasis and an appropriate root to shoot ratio. Plant Mol Biol 65:125–136. https://doi.org/10.1007/s11103-007-9203-6
Wu Y, Luo L, Chen L et al (2016) Chromosome map**, molecular cloning and expression analysis of a novel gene response for leaf width in rice. Biochem Biophys Res Commun 480:394–401. https://doi.org/10.1016/j.bbrc.2016.10.061
Xu Y, Yan S, Jiang S et al (2023) Identification of a rice leaf width gene Narrow Leaf 22 (NAL22) through genome-wide association study and gene editing technology. Int J Mol Sci 24:4073. https://doi.org/10.3390/ijms24044073
You J, **ao W, Zhou Y et al (2022) The APC/CTAD1-WIDE LEAF 1-NARROW LEAF 1 pathway controls leaf width in rice. Plant Cell 34:4313–4328. https://doi.org/10.1093/plcell/koac232
Zhang G-H, Li S-Y, Wang L et al (2014) LSCHL4 from japonica cultivar, which is allelic to NAL1, increases yield of indica super rice 93–11. Mol Plant 7:1350–1364. https://doi.org/10.1093/mp/ssu055
Zhang T, Wang J, Luo R et al (2023) OsHLS1 regulates plant height and development by controlling active gibberellin accumulation in rice (Oryza sativa L.). Plant Sci 326:111508. https://doi.org/10.1016/j.plantsci.2022.111508
Acknowledgements
This research was supported by the fund for China Agriculture Research System (CARS-01-08), National Natural Science Foundation of China (31960400), Major Discipline Academic and Technical Leaders Training Program of Jiangxi Province (20213BCJL22044), Hubei Province Natural Science Foundation of China (2022CFB017), Jiangxi Technology Innovation Guidance Program (20223AEI91010), and Jiangxi Province Natural Science Foundation (20232BAB205033, 20232BAB205017, and 20224BAB215008).
Author information
Authors and Affiliations
Contributions
JW and HX designed the experiments and analyzed the data; JW, WL, KL, JP, XZ, LL, MQ, WC, LL, WX, YL, and YC performed most of the experiments; JW and HX completed the writing. HX agreed to serve as the author responsible for contact and ensures communication. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Informed consent
Not applicable.
Institutional review bosard statement
Not applicable.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Long, W., Pan, J. et al. DNAL7, a new allele of NAL11, has major pleiotropic effects on rice architecture. Planta 259, 93 (2024). https://doi.org/10.1007/s00425-024-04376-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-024-04376-4