Abstract
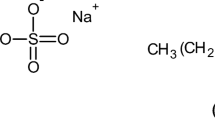
The mixed micellization behavior of (1-dodecyl-1-methylpiperidinium chloride) [C12mpip]Cl and hydrocarbon surfactants, sodium dodecylbenzene sulfonate (SDBS) as an anionic surfactant, cetyltrimethylammonium bromide (CTAB) as a cationic surfactant, and octylphenol ethoxylate (Titron X-100) as a non-ionic surfactant in water was investigated using dynamic light scattering (DLS), conductivity, UV spectroscopy, and surface tension measurements. In addition, critical micellar concentration (CMC), the degree of counterion binding (ꞵ), and various thermodynamic parameters of micellization have been evaluated from conductivity measurements. The different interfacial properties were calculated from surface tension measurement. The experimental results of the mix system in an aqueous solution indicated that CMC values decreased with the addition of different mole fractions of surfactant in the surface active ionic liquid (SAIL) mixture. The mix system’s hydrodynamic radius was less for the higher concentration of the surfactant in the mix system, which was assessed using the DLS methodology. The micellar mole fractions (Xm), ideal CMC (CMC*), interaction parameter (βm), and activity coefficients of components (f1 and f2) in the mixed micelle were calculated using various theoretical models (Clint, Rubingh, and Motomura). The negative values of the interaction parameter (βm) suggest the synergistic effects in a mixed state. The mixed micellar parameters (CMC* and Xm) ensure the non-ideal behavior of the mixed system.
Graphical abstract








Similar content being viewed by others
References
Martins MAP, Frizzo CP, Moreira DN et al (2008) Ionic liquids in heterocyclic synthesis. Chem Rev 108:2015–2050. https://doi.org/10.1021/cr078399y
Tsubone K (2003) The interaction of an anionic gemini surfactant with conventional anionic surfactants. J Colloid Interface Sci 261:524–528. https://doi.org/10.1016/S0021-9797(03)00088-2
Moud AA (2022) Rheology and microscopy analysis of polymer – surfactant complexes. Colloid Polym Sci. https://doi.org/10.1007/s00396-022-04982-2
Lochhead BRY, Huisinga LR (2020) A review of polymer–surfactant interactions. handbook of surface and colloid chemistry 654–699. https://doi.org/10.1201/b18633-17
Khan N, Brettmann B (2019) Intermolecular interactions in polyelectrolyte and surfactant complexes in solution. Polymers 11:1–28. https://doi.org/10.3390/polym11010051
Jabbari M, Izadmanesh Y, Ghavidel H (2019) Synthesis of ionic liquids as novel emulsifier and demulsifiers. J Mol Liq. https://doi.org/10.1016/j.molliq.2019.111512
Wu J, Shu Q, Niu Y et al (2018) Preparation, characterization, and antibacterial effects of chitosan nanoparticles embedded with essential oils synthesized in an ionic liquid containing system. J Agric Food Chem 66:7006–7014. https://doi.org/10.1021/acs.jafc.8b01428
Clint JH (1975) Micellization of mixed nonionic surface active agents. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases 71:1327–1334. https://doi.org/10.1039/F19757101327
Azum N, Asiri AM, Rub MA et al (2013) Mixed micellization of gemini surfactant with nonionic surfactant in aqueous media: a fluorometric study. Colloid J 75:235–240. https://doi.org/10.1134/S1061933X13030022
Wu Z, Li Y, Li J et al (2019) Study on the properties and self-assembly of fatty alcohol ether carboxylic ester anionic surfactant and cationic surfactant in a mixed system. New J Chem 43:12494–12502. https://doi.org/10.1039/c9nj02407g
Kume G, Gallotti M, Nunes G (2008) Review on anionic/cationic surfactant mixtures. J Surfactants Deterg 11:1–11. https://doi.org/10.1007/s11743-007-1047-1
Banjare MK, Kurrey R, Yadav T et al (2017) A comparative study on the effect of imidazolium-based ionic liquid on self-aggregation of cationic, anionic and nonionic surfactants studied by surface tension, conductivity, fluorescence and FTIR spectroscopy. J Mol Liq 241:622–632. https://doi.org/10.1016/j.molliq.2017.06.009
Javadian S, Nasiri F, Heydari A et al (2014) Modifying effect of imidazolium-based ionic liquids on surface activity and self-assembled nanostructures of sodium dodecyl sulfate. J Phys Chem B 118:4140–4150. https://doi.org/10.1021/jp5010049
Chat OA, Maswal M, Hassan PA et al (2015) Effect of mixed micellization on dimensions of 1-butyl-3-methylimidazolium dodecylsulfate micelles in presence of electrolytes. Colloids Surf, A 484:498–507. https://doi.org/10.1016/j.colsurfa.2015.08.032
Kumar H, Kaur G, Priya (2020) Influence of tetra ethyl ammonium bromide (C2H5)4NBr on the aggregation behavior of surface active ionic liquid 1-tetradecyl-3-methylimidazolium bromide [C14mim][Br]. J Mol Liq 313:113431. https://doi.org/10.1016/j.molliq.2020.113431
Kumar H, Kaur G (2021) Aggregation behavior of mixed micellar system of dodecyl sulfate-based surface-active ionic liquids and anionic surfactant in aqueous media. J Surfactants Deterg 24:209–227. https://doi.org/10.1002/jsde.12486
Mandal B, Mondal S, Pan A et al (2015) Colloids and surfaces A : physicochemical and engineering aspects physicochemical study of the interaction of lysozyme with surface active ionic liquid 1-butyl-3-methylimidazolium octylsulfate [BMIM ] [OS ] in aqueous and buffer media. Colloids Surf, A 484:345–353. https://doi.org/10.1016/j.colsurfa.2015.07.052
Pal A, Punia R, Dubey GP (2021) Formation of mixed micelles in an aqueous mixture of a biamphiphilic surface active ionic liquid and an anionic surfactant: Experimental and theoretical study. J Mol Liq 337:116355. https://doi.org/10.1016/j.molliq.2021.116355
Chabba S, Kumar S, Kumar V et al (2015) Colloids and surfaces A : physicochemical and engineering aspects interfacial and aggregation behavior of aqueous mixtures of imidazolium based surface active ionic liquids and anionic surfactant sodium dodecylbenzenesulfonate. Colloids Surf, A 472:9–20. https://doi.org/10.1016/j.colsurfa.2015.02.032
Ali A, Farooq U, Uzair S, Patel R (2016) Conductometric and tensiometric studies on the mixed micellar systems of surface-active ionic liquid and cationic surfactants in aqueous medium. J Mol Liq 223:589–602. https://doi.org/10.1016/j.molliq.2016.08.082
Jafari-Chashmi P, Bagheri A (2018) The strong synergistic interaction between surface active ionic liquid and anionic surfactant in the mixed micelle using the spectrophotometric method. J Mol Liq 269:816–823. https://doi.org/10.1016/j.molliq.2018.08.094
Carnero Ruiz C, Molina-Bolívar JA, Aguiar J et al (2001) Thermodynamic and structural studies of Triton X-100 micelles in ethylene glycol-water mixed solvents. Langmuir 17:6831–6840. https://doi.org/10.1021/la010529p
Pal A, Punia R (2019) Mixed micellization behaviour of tri-substituted surface active ionic liquid and cationic surfactant in aqueous medium and salt solution: experimental and theoretical study. J Mol Liq 296:111831. https://doi.org/10.1016/j.molliq.2019.111831
Némethy G, Scheraga HA (1962) Thermodynamic properties of liquid water. J Chem Phys 36:3382–3400. https://doi.org/10.1063/1.1732472
Hait SK, Majhi PR, Blume A, Moulik SP (2003) A critical assessment of micellization of sodium dodecyl benzene sulfonate ( SDBS ) and its interaction with poly (vinyl pyrrolidone) and hydrophobically modified polymers, JR 400 and LM 200. J Phys Chem B 2003:3650–3658. https://doi.org/10.1021/jp027379r
Shah SK, Bhattarai A (2020) Interfacial and micellization behavior of cetyltrimethylammonium bromide (CTAB) in water and methanol-water mixture at 298.15 to 323.15 K. J Chem 2020:1–13. https://doi.org/10.1155/2020/4653092
Tikariha D, Ghosh KK, Barbero N et al (2011) Micellization properties of mixed cationic gemini and cationic monomeric surfactants in aqueous-ethylene glycol mixture. Colloids Surf, A 381:61–69. https://doi.org/10.1016/j.colsurfa.2011.03.027
Sinha S, Tikariha D, Lakra J et al (2015) Effect of polar organic solvents on self-aggregation of some cationic monomeric and dimeric surfactants. J Surfactants Deterg 18:629–640. https://doi.org/10.1007/s11743-015-1686-6
Mittal KL (1979) Solution chemistry of surfactants, Vol. I and II. (I-XI+519-:)
Holland PM (1986) Solutions pseudo-phase separation approach for treating nonideal mixed. Advances in Collodal and Interface Science 26:111–129. https://doi.org/10.1016/0001-8686(86)80018-5
Aratono M, Ikeguchi M, Takiue T et al (1995) Thermodynamic study on the miscibility of sodium perfluorooctanoate and sodium decyl sulfate in the adsorbed film and micelle. Journal of Colloid And Interface Science 174:156–161. https://doi.org/10.1006/jcis.1995.1377
Sohrabi B, Gharibi H, Tajik B et al (2008) Molecular interactions of cationic and anionic surfactants in mixed monolayers and aggregates. J Phys Chem B 112:14869–14876. https://doi.org/10.1021/jp803105n
Maeda H (1995) A simple thermodynamic analysis of the stability of ionic/nonionic mixed micelles. Journal of colloidal and interface science 172:98–105. https://doi.org/10.1006/jcis.1995.1230
Itoh H, Ishido S, Nomura M et al (1996) Estimation of the hydrophobicity in microenvironments by pyrene fluorescence measurements: n-yβ-octylglucoside micelles. J Phys Chem 100:9047–9053. https://doi.org/10.1021/jp953682z
Babu K, Pal N, Saxena VK, Mandal A (2016) Synthesis and characterization of a new polymeric surfactant for chemical enhanced oil recovery. Korean J Chem Eng 33:711–719. https://doi.org/10.1007/s11814-015-0186-8
Pillai P, Pal N, Mandal A (2017) Synthesis, characterization, surface properties and micellization behaviour of imidazolium-based ionic liquids. J Surfactants Deterg 20:1321–1335. https://doi.org/10.1007/s11743-017-2021-1
Sood AK, Aggarwal M (2018) Evaluation of micellar properties of sodium dodecylbenzene sulphonate in the presence of some salts. J Chem Sci 130:1–7. https://doi.org/10.1007/s12039-018-1446-z
Movchan TG, Soboleva IV, Plotnikova EV et al (2012) Dynamic light scattering study of cetyltrimethylammonium bromide aqueous solutions. Colloid J 74:239–247. https://doi.org/10.1134/S1061933X1202007X
Pal A, Pillania A (2016) Thermodynamic and micellization properties of aqueous cetyltrimethylammonium bromide solution in presence of 1-butyl-2,3-dimethylimidazolium bromide. Fluid Phase Equilib 412:115–122. https://doi.org/10.1016/j.fluid.2015.12.042
Acknowledgements
The authors are grateful to the Charotar University of Science and Technology (Charusat University) for providing research facilities to carry out the research work. In addition, Nidhi Patel and Deep Sharma acknowledge the Government of Gujarat for giving the “SHODH” Scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Patel, N., Patel, V., Sharma, D. et al. Effect of hydrocarbon surfactants on the properties of 1-dodecyl-1-methylpiperidinium chloride: synthesis, micellization, thermodynamics, and surface parameters. Colloid Polym Sci 300, 903–916 (2022). https://doi.org/10.1007/s00396-022-04996-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-022-04996-w