Abstract
Background
Various second-generation transcatheter heart valve (THV) prostheses with high clinical efficacy and safety are available, but there is limited large-scale data available comparing their hemodynamic performance and clinical implications.
Objective
To compare the hemodynamic performance and short-term clinical outcome of four second-generation THV prostheses.
Methods
24,124 patients out of the German Aortic Valve Registry who underwent transfemoral transcatheter aortic valve implantation (TAVI) (Evolut™ R n = 7028, Acurate neo™ n = 2922, Portico n = 878 and Sapien 3 n = 13,296) were included in this analysis. Propensity-score weighted analysis was performed to control for differences in age, left ventricular function, STS score and sex. Primary endpoint was survival at one-year, secondary endpoints were 30 days survival, pre-discharge transvalvular gradients, paravalvular leakage and peri-procedural complications.
Results
Thirty-day and one-year survival were not significantly different between the four patient groups. Transvalvular gradients in Evolut™ R and Acurate neo™ were significantly lower as compared to Portico and Sapien 3 at hospital discharge. This difference exists across all annulus sizes. Paravalvular leakage ≥ II occurred significantly less often in the Sapien 3 group (1.2%, p < 0.0001). Rate of severe procedural complications was low and comparable in all groups. Permanent pacemaker implantation rate at one year was lowest in the ACUARATE neo group (13.0%) and highest in the Evolut™ R group (21.9%).
Conclusion
Albeit comparable short-term clinical outcomes there are certain differences regarding hemodynamic performance and permanent pacemaker implantation rate between currently available THV prostheses which should be considered for individual prosthesis selection.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transcatheter aortic valve implantation (TAVI) has become a safe and standardized procedure. With the development of second-generation transcatheter heart valve (THV) prostheses significantly lower rates of procedure-related complications and higher clinical success could be observed after TAVI as compared to first-generation THV prostheses. One balloon-expandable THV prosthesis (Sapien 3) and three self-expanding THV prostheses (Acurate neo™, Evolut™ R and Portico) are frequently used. Due to the different prosthesis design (intra-annular versus supra-annular) and distinct implantation mode a difference in hemodynamic performance, clinical outcome as well as safety profile could be hypothesized.
Previous studies suggest there might be differences in transvalvular gradients, paravalvular leakage rates, permanent pacemaker implantation, and cerebrovascular event rates [1,2,3,4,5,6,7,8]. However, CE mark studies and post-market registries of these four prostheses showed an excellent safety profile [4, 9,10,11]. Clinical outcomes as well as 30-day and 1-year mortality seem to be similar for the four THV prostheses, however, there is limited direct large-scale comparison, in particular in all-comers populations.
To fill this gap of knowledge we analyzed the data of all patients included in the German Aortic Valve Registry (GARY) who were treated with a transfemoral TAVI with either the balloon-expandable Sapien 3 or the self-expanding EvolutTMR, Acurate neo™ or Portico THV prosthesis.
Methods
Data of all patients who underwent transfemoral TAVI between 2014 and 2019 were extracted from the database of GARY which is a nationwide multicenter all-comers registry. The registry design has been previously published [12]. Prostheses studied in this analysis included the balloon-expandable SAPIEN 3 (Edwards Lifesciences, Irvine, CA, USA) and the self-expanding Acurate neo™ (Boston Scientific, Marlborough, MA, USA), Evolut™ R (Medtronic, Minneapolis, MN, USA) and Portico (Abbott). Prostheses selection was at the discretion of the operating physician.
Transvalvular gradients and paravalvular leakage were assessed pre-discharge by transthoracic echocardiography. Procedural data, procedural success and severe intraprocedural complications were analyzed.
Patients were followed-up at 30 days and one year regarding adverse clinical events and NYHA classification by phone interviews. One-year follow-up was not available in patients undergoing transfemoral TAVI in 2016 and 2017. The primary outcome was the mortality rate at one year.
Ethical statement
The study complies with the Declaration of Helsinki. Subjects included in the registry gave informed consent, and an ethics body at participating institutions approved the use of patient data for research purposes.
Statistics
Continuous variables are reported as mean ± standard deviation (SD) for the total patient cohort or mean ± standard error (SE) for group comparisons. Categorical variables are reported as frequencies and percentages. Comparisons between the different prosthesis groups were made using unweighted and weighted linear and generalized linear models. For adjusted comparisons with weighted regression analysis, a propensity score model from boosted logistic regression analysis was used to determine weights to estimate the average treatment effect of the group receiving the Acurate neo™ devices as a reference. Variables included in the propensity score model were age, gender, LVEF (≤ 30%, 31–50%, > 50%) and STS score. Adjusted analysis according to this propensity score model was also used to compare 30-day and one-year mortality in the patient groups with a weighted Cox proportional hazard model.
Tests with a two-sided p-value of ≤ 0.05 were considered statistically significant. Statistical analysis was performed with R (R Foundation for Statistical Computing, Vienna, Austria). The packages “twang”, “gbm” and “survey” were used for calculating propensity score weights.
Results
Baseline characteristics
Overall, 24,124 patients who underwent transfemoral TAVI have been included in this retrospective analysis. Mean age ± SD was 80.4 ± 5.9 years and there was a female predominance, in particular in self-expanding prostheses (Table 1). The percentage of comorbidities is summarized in Table 1. Occurrence of peripheral and coronary artery disease was significantly different between the four groups as well as left ventricular function. Patients treated with Sapien 3 showed a more severe aortic valve calcification (Supplementary Table 1).
Only Evolut™ R and Sapien 3 valves are available for annulus sizes > 27 mm, thus mean annulus sizes are significantly larger in these groups (24.2 mm and 24.7 mm for Evolut™ R and Sapien 3, versus 23.9 mm and 23.8 mm for Acurate neo™ and Portico, p < 0.001, Table 1). To account for systematic data differences, weighted analysis with weights from a propensity score model using adjustments for age, gender, STS score and LV function was also performed. See Table 1 and Supplemental Fig. 1 for the adjustment effects.
Clinical outcome
Overall survival in the study cohort was 97.2% (95% CI 97.0–97.4%) at 30 days and 83.0% (95% CI 82.3–83.7%) at 1 year, respectively. There was no significant difference between the four groups in the propensity score-adjusted analysis (Fig. 1A and B). To explore the effect of aortic annulus size this parameter was also included as a covariate. Furthermore, as sensitivity analysis, we restricted the data to a subset with an aortic annulus diameter between 21 and 27 mm (n = 19,504, Supplemental Fig. 2). Both approaches yielded no significant differences between the four groups.
Mortality after 30 days (A) and one year (B) with propensity score derived weights adjusting for age, gender, left ventricular ejection fraction and STS score. Mortality was comparable for all studied TAVI prostheses with no significant differences even in not significance-adjusted pairwise comparisons
Pre-procedurally, patients were symptomatic with dyspnoea mainly in NYHA class III and IV and improved significantly after 1 year, regardless of the implanted TAVI prosthesis (Fig. 2 for unadjusted comparisons, results were even more comparable between the groups after weighting). In adjusted comparisons, there was no significant difference between the groups regarding stroke rate, TIA rate in patients without stroke, rehospitalization, reintervention and myocardial infarction.
NYHA class at baseline (pre) and after 1 year (post) showing a clear improvement following TAVI without any significant difference between the four groups. Results are shown for all 6.606 patients with 1 year follow-up assessment. Supplemental Fig. 2 shows NYHA class at baseline in all 24,124 patients
Permanent pacemaker implantation rate at 1 year was lowest in the Acurate neo™ group (13.0%) and highest in the Evolut™ R group (23.0%, Table 2, p < 0.0001). Results for adjusted weighted comparisons were similar (Table 2).
Hemodynamic performance
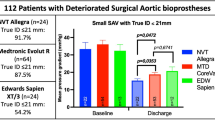
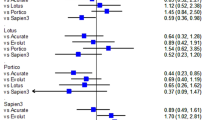
Pre-procedural mean transvalvular gradient was comparable between the four groups (Table 1), whereas post-procedurally mean transvalvular gradient was significantly lower in the Acurate neo™ group (mean ± SE 8.60 ± 0.14 mmHg, p < 0.0001, Table 3) versus the overall cohort. The differences remained significant in the weighted analysis and in a sensitivity analysis in a subgroup of patients with aortic annulus diameter of < 23 mm, 23–25 mm and > 25 mm. Data on effective orifice areas have not been collected in GARY.
The observed overall rate of clinically relevant paravalvular leakage (grade II +) at discharge was 1.6% ranging from 1.2% in the Sapien 3 group to 2.1% and 3.1% in the Acurate neo™ and Portico groups, respectively (Table 3, p < 0.0001). Again, the differences remained similar in extent and significance in the weighted analysis and in the sensitivity subset of patients with aortic annulus diameter of < 23 mm, 23–25 mm and > 25 mm. Only 32 out of 23,652 patients with available data displayed grade III or IV paravalvular leakage.
Procedural data
Procedural characteristics are listed in Table 4. Radiation time and amount of contrast dye were significantly lower in the ACUARTE neo™ and the Sapien 3 group (Table 4). Pre-dilatation rate was highest in Acurate neo™ and Portico (84.7% and 82.0%) and lowest in Evolut™ R (55.2%). Post-dilatation rate was significantly lower in Sapien 3 (13.1%). Overall procedural success rate was high (98.3%) without significant difference between the groups (Table 4).
The rate of vascular complications during the procedure was 2.9% with a significantly lower rate in the Sapien 3 group (2.0%, p = 0.003) which was also confirmed in adjusted analysis using propensity score weights. Severe intraprocedural complications occurred very infrequently (Table 4). Conversion rate to open heart surgery was reported in 0.4% of patients without differences between the groups. Coronary obstruction was reported in 36 out of 24,124 patients (0.1%). An immediate valve-in-valve implantation was performed in 134 out of 21,458 patients (0.6%) without any difference between the four groups.
Discussion
This study comprises a large-scale multi-center comparison of hemodynamic performance and short-term clinical outcome of the four most implanted second-generation THV prostheses. The main findings in this all-comers cohort are (1) one-year survival rates are not different between the various second-generation THV prostheses; (2) short-term outcome and hemodynamic performance are excellent for all THV prostheses with some certain differences of lower transvalvular gradients in supra-annular self-expanding valves and less paravalvular leakage in balloon-expandable valves; (3) a high procedural success rate combined with a low rate of severe intraprocedural complications, which confirms excellent efficacy and safety of all studied devices.
Clinical outcome
30-day and one-year survival rates were high in all studied second-generation THV prostheses (97.2% and 83.0%, respectively) in this intermediate-risk cohort and are in line with data from previous publications [4, 5, 7, 9,10,11, 13,14,15]. In contrast to one recent study from the French TAVI registry there was no difference in survival between balloon- and self-expanding THV prostheses [16] This might be explained by the fact that in the French registry patients received only first-generation self-expanding THVs which are more prone to a higher rate of clinically significant paravalvular regurgitation, periprocedural complications including stroke and permanent pacemaker implantation [17].
Short- and intermediate-term clinical event rates were low which underlines the high safety of all four THV prostheses.
Rehospitalisation within a year was reported in approximately 40% of the patients in the current analysis mainly due to cardiovascular reasons in about 15%. There was a significant improvement in clinical symptoms which is comparable to previous studies in an intermediate-risk cohort [18].
The stroke rate at one year was only 1.6%, which is significantly lower as compared to previous publications [7, 13, 15, 17, 19,20,21]. This might in part be due to underreporting in the registry-based data acquisition.
The rate of new permanent pacemaker implantation (PPI) was 18.8% after one year in the overall cohort. Differences between different devices have previously been described [4, 6, 22]. However, the rate is significantly higher as compared to randomized studies in low- and intermediate-risk patients [2, 3, 22, 23]. As reported before [15], Acurate neo™ displayed the lowest rate of PPI among self-expanding vales (13.0%) which might be due to the lower radial force of the stent as well as to the different implantation mode with a top-down deployment resulting in lower mechanical stress on the left ventricular outflow tract and subsequentially on the AV conduction system.
Procedural data and hemodynamics
The incidence of severe intraoperative complications was extremely confirming a high safety for all TAVI platforms investigated. Radiation time and usage of contrast medium was significantly lower in Acurate neo™ and Sapien 3 which is mainly due to the distinct implantation modes.
Pre-dilatation rate was highest in the Acurate neo™ and Portico group since the official recommendation is to pre-dilate the calcified native valve before implantation At the time of data acquisition Sapien 3 was also mainly implanted with pre-dilatation, however, current data have shown that Sapien 3 can safely be implanted without pre-dilation [24,25,26]. Post-dilatation rate was significantly lower in the balloon-expandable Sapien 3 [4, 10,11,12] and most probably a result of the high radial force of this valve [27]. Pre- and post-ballooning did not impact stroke rate in this cohort.
Overall peri interventional vascular access complication rate was in line with other reports in intermediate-risk patients [4, 12]. Notably, vascular complication rate was lowest in balloon-expandable THV which might be partially explained by the lower proportion of female patients who are prone to access site complications.
Baseline transvalvular gradients were comparable and there was a significant reduction in all four groups. Of interest, transvalvular gradients were lowest in the self-expanding Evolut™ R and Acurate neo™ even after adjusting for various annulus sizes. This finding is mainly explained by the supra-annular valve design which translates into lower transvalvular gradients. Among the intra-annular prosthesis types, the self-expanding Portico shows lower gradients compared to the balloon-expandable Sapien 3. Previous publications have reported the same finding and, furthermore, showed a significantly higher effective orifice area and a lower rate of patient prosthesis mismatch in supra-annular valves [2, 3]. There is evidence that patient prosthesis mismatch is a strong predictor for cardiac remodelling, development of heart failure and even premature prosthesis degeneration [28]. Since effective orifice area has not been collected in GARY we are not able to make any statements regarding this issue from our data, but at least the results of the transvalvular gradients emphasize that there is a difference in hemodynamics between the studied THV devices which might impact long-term outcome.
Paravalvular leakage is a major limitation of TAVI and has been linked to reduced survival in various studies [16, 22]. As opposed to first-generation THV devices all studied THV showed an acceptably low rate of clinically relevant paravalvular leakage being lowest with the balloon-expandable Sapien 3 device (1.2% PVL grade II or greater) s [29]. Moderate and severe paravalvular leakage was a rare finding confirming the high efficacy of all second-generation devices to prevent this important limitation of TAVI.
Differentiated THV prosthesis choice
The presented data reflects that TAVI implanters already perform individual decisions for specific patient needs, e.g. more low profile THV use in patients with coronary heart disease and patients with more severe aortic valve calcification or more often use of smaller delivery devices in patients with PAD. The all-comers face-to-face comparison of these four second-generation THVs adds evidence which prosthesis can be chosen, if a lower gradient, less PVL, lower permanent pacemaker rate or less burden in terms of radiation, contrast or pre- and post-dilatation is attempted.
Study limitation
Due to the retrospective design and the registry-based data acquisition of the study typical limitations apply. To control for confounding baseline variables a propensity score-adjusted analysis was used, but bias due to unknown or unmeasured confounders cannot be excluded, in particular since patients were not randomized to the respective treatment group.
Conclusions
From our data, we conclude that all currently available THV devices can be safely and effectively used to treat patients with aortic stenosis. There are slight but significant differences in hemodynamics and permanent pacemaker implantation rate between the contemporary second-generation THV devices which imply that patient individual prosthesis selection is necessary to achieve the optimal short- and intermediate-term result. Whether these differences which are mainly due to the prosthesis design and implantation mode translate into significant long-term outcomes or effect prosthesis durability needs to be elucidated.
Perspectives
Impact on daily practice
This work demonstrates that despite the high overall clinical efficacy and safety of all investigated THV prostheses patient-based individual THV prostheses selection is necessary to achieve the best clinical result. Further studies are needed to elucidate the best selection criteria. In addition, long-term follow-up studies are needed to evaluate the impact of hemodynamic differences on THV prostheses longevity.
References
Abdel-Wahab M, Neumann F-J, Mehilli J, Frerker C, Richardt D, Landt M et al (2015) 1-year outcomes after transcatheter aortic valve replacement with balloon-expandable versus self-expandable valves. J Am Coll Cardiol 66:791–800
Mauri V, Kim WK, Abumayyaleh M, Walther T, Moellmann H, Schaefer U et al (2017) Short-term outcome and hemodynamic performance of next-generation self-expanding versus balloon-expandable transcatheter aortic valves in patients with small aortic annulus: a multicenter propensity-matched comparison. Circ Cardiovasc Interv 10:e005013
Husser O, Kim W-K, Pellegrini C, Holzamer A, Walther T, Mayr PN et al (2017) Multicenter comparison of novel self-expanding versus balloon-expandable transcatheter heart valves. JACC: Cardiovasc Interv 10:2078–2087
Grube E, Van Mieghem NM, Bleiziffer S, Modine T, Bosmans J, Manoharan G et al (2017) Clinical outcomes with a repositionable self-expanding transcatheter aortic valve prosthesis: the international FORWARD study. J Am Coll Cardiol 70:845–853
Möllmann H, Linke A, Holzhey DM, Walther T, Manoharan G, Schäfer U et al (2017) Implantation and 30-day follow-up on all 4 valve sizes within the portico transcatheter aortic bioprosthetic family. JACC: Cardiovasc Interv 10:1538–1547
Fujita B, Schmidt T, Bleiziffer S, Bauer T, Beckmann A, Bekeredjian R et al (2020) Impact of new pacemaker implantation following surgical and transcatheter aortic valve replacement on 1-year outcome. Eur J Cardiothorac Surg 57:151–159
Wendler O, Schymik G, Treede H, Baumgartner H, Dumonteil N, Neumann FJ et al (2017) SOURCE 3: 1-year outcomes post-transcatheter aortic valve implantation using the latest generation of the balloon-expandable transcatheter heart valve. Eur Heart J 38:2717–2726
Mauri V, Reimann A, Stern D, Scherner M, Kuhn E, Rudolph V et al (2016) Predictors of permanent pacemaker implantation after transcatheter aortic valve replacement with the SAPIEN 3. JACC: Cardiovasc Interv 9:2200–2209
Thourani VH, Kodali S, Makkar RR, Herrmann HC, Williams M, Babaliaros V et al (2016) Transcatheter aortic valve replacement versus surgical valve replacement in intermediate-risk patients: a propensity score analysis. Lancet 387:2218–2225
Möllmann H, Hengstenberg C, Hilker M, Kerber S, Schäfer U, Rudolph T et al (2018) Real-world experience using the Acurate neoTM prosthesis: 30-day outcomes of 1,000 patients enrolled in the SAVI TF registry. EuroIntervention 13:e1764–e1770
Linke A, Holzhey D, Möllmann H, Manoharan G, Schäfer U, Frerker C et al (2018) Treatment of aortic stenosis with a self-expanding, resheathable transcatheter valve. Circ Cardiovasc Interv 11:e005206
Lanz J, Kim WK, Walther T, Burgdorf C, Möllmann H, Linke A et al (2019) Safety and efficacy of a self-expanding versus a balloon-expandable bioprosthesis for transcatheter aortic valve replacement in patients with symptomatic severe aortic stenosis: a randomised non-inferiority trial. Lancet 394:1619–1628
Hamm CW, Möllmann H, Holzhey D, Beckmann A, Veit C, Figulla HR et al (2014) The German Aortic Valve Registry (GARY): in-hospital outcome. Eur Heart J 35:1588–1598
Leon MB, Smith CR, Mack MJ, Makkar RR, Svensson LG, Kodali SK et al (2016) Transcatheter or surgical aortic-valve replacement in intermediate-risk patients. N Engl J Med 374:1609–1620
Reardon MJ, Van Mieghem NM, Popma JJ, Kleiman NS, Søndergaard L, Mumtaz M et al (2017) Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. N Engl J Med 376:1321–1331
Kim WK, Hengstenberg C, Hilker M, Kerber S, Schäfer U, Rudolph T et al (2018) The SAVI-TF Registry: 1-year outcomes of the european post-market registry using the acurate neoTM transcatheter heart valve under real-world conditions in 1,000 patients. JACC: Cardiovasc Interv 11:1368–1374
Van Belle E, Vincent F, Labreuche J, Auffret V, Debry N, Lefèvre T et al (2020) Balloon-expandable versus self-expanding transcatheter aortic valve replacement: a propensity-matched comparison from the FRANCE-TAVI registry. Circulation 141:243–259
Tamburino C, Barbanti M, D’Errigo P, Ranucci M, Onorati F, Covello RD et al (2015) 1-year outcomes after transfemoral transcatheter or surgical aortic valve replacement: results from the Italian OBSERVANT study. J Am Coll Cardiol 66:804–812
Baron SJ, Arnold SV, Wang K, Magnuson EA, Chinnakondepali K, Makkar R et al (2017) Health status benefits of transcatheter vs surgical aortic valve replacement in patients with severe aortic stenosis at intermediate surgical risk: results from the PARTNER 2 randomized clinical trial. JAMA Cardiol 2:837–845
Durko AP, Reardon MJ, Kleiman NS, Popma JJ, van Mieghem NM, Gleason TG et al (2018) Neurological complications after transcatheter versus surgical aortic valve replacement in intermediate-risk patients. J Am Coll Cardiol 72:2109–2119
Kapadia SR, Huded CP, Kodali SK, Svensson LG, Tuzcu EM, Baron SJ et al (2018) Stroke after surgical versus transfemoral transcatheter aortic valve replacement in the PARTNER trial. J Am Coll Cardiol 72:2415–2426
Søndergaard L, Popma JJ, Reardon MJ, van Mieghem NM, Deeb GM, Kodali S et al (2019) Comparison of a complete percutaneous versus surgical approach to aortic valve replacement and revascularization in patients at intermediate surgical risk: results from the randomized SURTAVI trial. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.118.039564
Kodali S, Pibarot P, Douglas PS, Williams M, Xu K, Thourani V et al (2015) Paravalvular regurgitation after transcatheter aortic valve replacement with the Edwards sapien valve in the PARTNER trial: characterizing patients and impact on outcomes. Eur Heart J 36:449–456
Vlastra W, Chandrasekhar J, Muñoz-Garcia AJ, Tchétché D, de Brito FS Jr, Barbanti M et al (2019) Comparison of balloon-expandable vs. self-expandable valves in patients undergoing transfemoral transcatheter aortic valve implantation: from the CENTER-collaboration. Eur Heart J 40:456–465
Butter C, Bramlage P, Rudolph T, Jacobshagen C, Rothe J, Treede H et al (2016) 2016) Balloon expandable transcatheter aortic valve implantation via the transfemoral route with or without pre-dilation of the aortic valve - rationale and design of a multicentre registry (EASE-IT TF. BMC Cardiovasc Disord 16:223
Ashauer JO, Bonaros N, Kofler M, Schymik G, Butter C, Romano M et al (2019) Balloon-expandable transcatheter aortic valve implantation with or without pre-dilation – results of a meta-analysis of 3 multicenter registries. BMC Cardiovasc Disord 19:172
Kim WK, Praz F, Blumenstein J, Liebetrau C, Gaede L, Van Linden A et al (2017) Transfemoral aortic valve implantation of Edwards SAPIEN 3 without predilatation. Catheter Cardiovasc Interv 89:E38–E43
Egron S, Fujita B, Gullón L, Pott D, Schmitz-Rode T, Ensminger S et al (2018) Radial force: an underestimated parameter in oversizing transcatheter aortic valve replacement prostheses. in vitro analysis with five commercialized valves. ASAIO J 64:536–543
Bilkhu R, Jahangiri M, Otto CM (2019) Patient-prosthesis mismatch following aortic valve replacement. Heart 105:s28–s33
Funding
Open Access funding enabled and organized by Projekt DEAL. The registry receives funding from unrestricted grants from medical device companies (Edwards Lifesciences, Medtronic, Abbott, Boston Scientific), the German Center for Cardiovascular Research (DZHK), the German Heart Foundation, the German Ministry of Health and donations from Dr Rolf M. Schwiete Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T. Rudolph is a proctor for Edwards Lifesciences and Boston Scientific and received speaker´s honoraria from Edwards Lifesciences, Medtronic, Abbott and Boston Scientific/Symetis SA. S. Ensminger is a consultant for Edwards LifeSciences and received speaker’s honoraria from Edwards LifeSciences and Medtronic. C. Frerker received speaker’s honoraria and travel support from Edwards Lifesciences, Medtronic, Boston Scientific, Abbott Vascular, Biotronik. H. Möllmann received speaker ‘s honoraria and / or consulting fees from Abbott, Boston Scientific, Edwards Lifesciences, and SMT. C. Hamm is an Advisory Board member of Medtronic. S. Baldus has received a research grant from Abbott and lecture fees from Abbott and Edwards. A. Böning has received speaker’s honoraria/consulting fees from Abiomed, Bayer, Edwards Lifesciences, Orion Pharma, Maquet, Smith & Nephew, Somahlution, Resuscitec, and Zoll, and is Advisory Board Member of Nordic Pharma and Guard Therapeutics. V. Rudolph has received research grants from Abbott Vascular and Edwards Lifesciences. S. Bleiziffer is a proctor for Medtronic and received speaker´s honoraria from Medtronic, Abbott, and Boston Scientific. E. Herrmann, D. Bon, T. Walther, T. Bauer, A. Beckmann, R. Bekeredijan, F. Beyersdorf, J. Gummert on have nothing to disclose.
Supplementary Information
Below is the link to the electronic supplementary material.
392_2023_2242_MOESM2_ESM.tif
Supplementary file2 Supplemental Figure 1 Standardized mean differences showing the effect of the propensity score adjustments on the variables included in the propensity score model (age, gender, left ventricular ejection fraction and STS score). (TIF 153 KB)
392_2023_2242_MOESM3_ESM.tif
Supplementary file3 Supplemental Figure 2 NYHA class at baseline (pre, n=24,124) and after 1 year (post, n=6,606) showing a clear improvement following TAVI without any significant difference between the four groups. (TIF 98 KB)
392_2023_2242_MOESM5_ESM.tif
Supplementary file5 Supplemental Figure 3 Mortality after 30 days (A) and one year (B) with propensity score derived weights adjusting for age, gender, left ventricular ejection fraction and STS score in a subset of patients with aortic annulus between 21 and 27 mm. Mortality was comparable for all studied TAVI prostheses. (TIF 89 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rudolph, T.K., Herrmann, E., Bon, D. et al. Comparison of contemporary transcatheter heart valve prostheses: data from the German Aortic Valve Registry (GARY). Clin Res Cardiol 113, 75–85 (2024). https://doi.org/10.1007/s00392-023-02242-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-023-02242-z