Abstract
Purpose
Whole-body positron emission tomography/magnetic resonance imaging (wbPET/MRI) is a promising diagnostic tool of recurrent prostate cancer (PC), but its role in primary staging of high-risk PC (hrPC) is not well defined. Thus, the aim was to compare the diagnostic accuracy for T-staging of PET-blinded reading (PBR) and PET/MRI.
Methods
In this prospective study, hrPC patients scheduled to radical prostatectomy (RPx) with extended lymphadenectomy (eLND) were staged with wbPET/MRI and either 68Ga-PSMA-11 or 11C-choline including simultaneous multiparametric MRI (mpMRI). Images were assessed in two sessions, first as PBR (mpMRI and wbMRI) and second as wbPET/MRI. Prostate Imaging Reporting and Data System criteria (PIRADS v2) were used for T-staging. Results were correlated with the exact anatomical localization and extension as defined by histopathology. Diagnostic accuracy of cTNM stage according to PBR was compared to pathological pTNM stage as reference standard.
Results
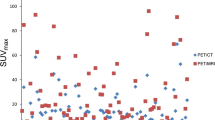
Thirty-four patients underwent wbPET/MRI of 68Ga-PSMA-11 (n = 17) or 11C-choline (n = 17). Twenty-four patients meeting the inclusion criteria of localized disease ± nodal disease based on imaging results underwent RPx and eLND, whereas ten patients were excluded from analysis due to metastatic disease. T-stage was best defined by mpMRI with underestimation of tumor lesion size by PET for both tracers. N-stage yielded a per patient sensitivity/specificity comparable to PBR.
Conclusion
MpMRI is the primary modality for T-staging in hrPC as PET underestimated T-stage in direct comparison to final pathology. In this selected study, cohort MRI shows no inferiority compared to wbPET/MRI considering N-staging.




Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J (2012) Jemal A (2015) Global cancer statistics. CA Cancer J Clin 65(2):87–108. https://doi.org/10.3322/caac.21262
Siegel RL, Miller KD (2016) Jemal A (2016) Cancer statistics. CA Cancer J Clin 66(1):7–30. https://doi.org/10.3322/caac.21332
Cooperberg MR, Cowan J, Broering JM, Carroll PR (2008) High-risk prostate cancer in the United States, 1990–2007. World J Urol 26(3):211–218. https://doi.org/10.1007/s00345-008-0250-7
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, Tomaszewski JE, Renshaw AA, Kaplan I, Beard CJ, Wein A (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280(11):969–974
Oesterling JE (1993) Using PSA to eliminate the staging radionuclide bone scan. Significant economic implications. Urol Clin N Am 20(4):705–711
Briganti A, Blute ML, Eastham JH, Graefen M, Heidenreich A, Karnes JR, Montorsi F, Studer UE (2009) Pelvic lymph node dissection in prostate cancer. Eur Urol 55(6):1251–1265. https://doi.org/10.1016/j.eururo.2009.03.012
Checcucci E, Amparore D, De Luca S, Autorino R, Fiori C, Porpiglia F (2019) Precision prostate cancer surgery: an overview of new technologies and techniques. Minerva Urol Nefrol 71(5):487–501. https://doi.org/10.23736/s0393-2249.19.03365-4
Jazayeri SB, Weissman B, Samadi DB (2018) Outcomes following robotic-assisted laparoscopic prostatectomy: Pentafecta and Trifecta achievements. Minerva Urol Nefrol 70(1):66–73. https://doi.org/10.23736/s0393-2249.17.02909-5
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, Fossati N, Gross T, Henry AM, Joniau S, Lam TB, Mason MD, Matveev VB, Moldovan PC, van den Bergh RCN, Van den Broeck T, van der Poel HG, van der Kwast TH, Rouviere O, Schoots IG, Wiegel T, Cornford P (2017) EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 71(4):618–629. https://doi.org/10.1016/j.eururo.2016.08.003
Hovels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL, Severens JL, Barentsz JO (2008) The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol 63(4):387–395. https://doi.org/10.1016/j.crad.2007.05.022
Albisinni S, Aoun F, Marcelis Q, Jungels C, Al-Hajj Obeid W, Zanaty M, Tubaro A, Roumeguere T, De Nunzio C (2018) Innovations in imaging modalities for recurrent and metastatic prostate cancer: a systematic review. Minerva Urol Nefrol 70(4):347–360. https://doi.org/10.23736/s0393-2249.18.03059-x
Rosenkrantz AB, Friedman K, Chandarana H, Melsaether A, Moy L, Ding YS, Jhaveri K, Beltran L, Jain R (2016) Current status of hybrid PET/MRI in oncologic imaging. AJR Am J Roentgenol 206(1):162–172. https://doi.org/10.2214/AJR.15.14968
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, Holland-Letz T, Hadaschik BA, Giesel FL, Debus J, Haberkorn U (2014) Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging 41(1):11–20. https://doi.org/10.1007/s00259-013-2525-5
Giesel FL, Sterzing F, Schlemmer HP, Holland-Letz T, Mier W, Rius M, Afshar-Oromieh A, Kopka K, Debus J, Haberkorn U, Kratochwil C (2016) Intra-individual comparison of (68)Ga-PSMA-11-PET/CT and multi-parametric MR for imaging of primary prostate cancer. Eur J Nucl Med Mol Imaging 43(8):1400–1406. https://doi.org/10.1007/s00259-016-3346-0
Schwenck J, Rempp H, Reischl G, Kruck S, Stenzl A, Nikolaou K, Pfannenberg C, la Fougere C (2017) Comparison of (68)Ga-labelled PSMA-11 and (11)C-choline in the detection of prostate cancer metastases by PET/CT. Eur J Nucl Med Mol Imaging 44(1):92–101. https://doi.org/10.1007/s00259-016-3490-6
Eissa A, Elsherbiny A, Coelho RF, Rassweiler J, Davis JW, Porpiglia F, Patel VR, Prandini N, Micali S, Sighinolfi MC, Puliatti S, Rocco B, Bianchi G (2018) The role of 68Ga-PSMA PET/CT scan in biochemical recurrence after primary treatment for prostate cancer: a systematic review of the literature. Minerva Urol Nefrol 70(5):462–478. https://doi.org/10.23736/s0393-2249.18.03081-3
Evangelista L, Guttilla A, Zattoni F, Muzzio PC, Zattoni F (2013) Utility of choline positron emission tomography/computed tomography for lymph node involvement identification in intermediate- to high-risk prostate cancer: a systematic literature review and meta-analysis. Eur Urol 63(6):1040–1048. https://doi.org/10.1016/j.eururo.2012.09.039
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69(1):16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Sobin LH, Gospodariwicz M, Wittekind C (2009) TNM classification of malignant tumors UICC International Union Against Cancer, vol 7. Wiley-Blackwell, New York, pp 243–248
Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, Beer AJ, Wester HJ, Gschwend J, Schwaiger M, Maurer T (2016) Simultaneous (68)Ga-PSMA HBED-CC PET/MRI improves the localization of primary prostate cancer. Eur Urol 70(5):829–836. https://doi.org/10.1016/j.eururo.2015.12.053
Rahbar K, Weckesser M, Huss S, Semjonow A, Breyholz HJ, Schrader AJ, Schafers M, Bogemann M (2016) Correlation of intraprostatic tumor extent with (6)(8)Ga-PSMA distribution in patients with prostate cancer. J Nucl Med 57(4):563–567. https://doi.org/10.2967/jnumed.115.169243
Fendler WP, Schmidt DF, Wenter V, Thierfelder KM, Zach C, Stief C, Bartenstein P, Kirchner T, Gildehaus FJ, Gratzke C, Faber C (2016) 68Ga-PSMA PET/CT detects the location and extent of primary prostate cancer. J Nucl Med 57(11):1720–1725. https://doi.org/10.2967/jnumed.116.172627
Afshar-Oromieh A, Malcher A, Eder M, Eisenhut M, Linhart HG, Hadaschik BA, Holland-Letz T, Giesel FL, Kratochwil C, Haufe S, Haberkorn U, Zechmann CM (2013) PET imaging with a [68Ga]gallium-labelled PSMA ligand for the diagnosis of prostate cancer: biodistribution in humans and first evaluation of tumour lesions. Eur J Nucl Med Mol Imaging 40(4):486–495. https://doi.org/10.1007/s00259-012-2298-2
Piert M, Park H, Khan A, Siddiqui J, Hussain H, Chenevert T, Wood D, Johnson T, Shah RB, Meyer C (2009) Detection of aggressive primary prostate cancer with 11C-choline PET/CT using multimodality fusion techniques. J Nucl Med 50(10):1585–1593. https://doi.org/10.2967/jnumed.109.063396
Park H, Meyer CR, Wood D, Khan A, Shah R, Hussain H, Siddiqui J, Seo J, Chenevert T, Piert M (2010) Validation of automatic target volume definition as demonstrated for 11C-choline PET/CT of human prostate cancer using multi-modality fusion techniques. Acad Radiol 17(5):614–623. https://doi.org/10.1016/j.acra.2010.01.003
Budaus L, Leyh-Bannurah SR, Salomon G, Michl U, Heinzer H, Huland H, Graefen M, Steuber T, Rosenbaum C (2016) Initial experience of (68)Ga-PSMA PET/CT imaging in high-risk prostate cancer patients prior to radical prostatectomy. Eur Urol 69(3):393–396. https://doi.org/10.1016/j.eururo.2015.06.010
Rauscher I, Maurer T, Beer AJ, Graner FP, Haller B, Weirich G, Doherty A, Gschwend JE, Schwaiger M, Eiber M (2016) Value of 68Ga-PSMA HBED-CC PET for the assessment of lymph node metastases in prostate cancer patients with biochemical recurrence: comparison with histopathology after salvage lymphadenectomy. J Nucl Med 57(11):1713–1719. https://doi.org/10.2967/jnumed.116.173492
Maurer T, Gschwend JE, Rauscher I, Souvatzoglou M, Haller B, Weirich G, Wester HJ, Heck M, Kubler H, Beer AJ, Schwaiger M, Eiber M (2016) Diagnostic efficacy of (68)Gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol 195(5):1436–1443. https://doi.org/10.1016/j.juro.2015.12.025
Herlemann A, Wenter V, Kretschmer A, Thierfelder KM, Bartenstein P, Faber C, Gildehaus FJ, Stief CG, Gratzke C, Fendler WP (2016) (68)Ga-PSMA positron emission tomography/computed tomography provides accurate staging of lymph node regions prior to lymph node dissection in patients with prostate cancer. Eur Urol 70(4):553–557. https://doi.org/10.1016/j.eururo.2015.12.051
van Leeuwen PJ, Emmett L, Ho B, Delprado W, Ting F, Nguyen Q, Stricker PD (2017) Prospective evaluation of 68Gallium-prostate-specific membrane antigen positron emission tomography/computed tomography for preoperative lymph node staging in prostate cancer. BJU Int 119(2):209–215. https://doi.org/10.1111/bju.13540
Hacker A, Jeschke S, Leeb K, Prammer K, Ziegerhofer J, Sega W, Langsteger W, Janetschek G (2006) Detection of pelvic lymph node metastases in patients with clinically localized prostate cancer: comparison of [18F]fluorocholine positron emission tomography-computerized tomography and laparoscopic radioisotope guided sentinel lymph node dissection. J Urol 176(5):2014–2019. https://doi.org/10.1016/j.juro.2006.07.037
Rhee H, Thomas P, Shepherd B, Gustafson S, Vela I, Russell PJ, Nelson C, Chung E, Wood G, Malone G, Wood S, Heathcote P (2016) Prostate specific membrane antigen positron emission tomography may improve the diagnostic accuracy of multiparametric magnetic resonance imaging in localized prostate cancer. J Urol 196(4):1261–1267. https://doi.org/10.1016/j.juro.2016.02.3000
Zamboglou C, Drendel V, Jilg CA, Rischke HC, Beck TI, Schultze-Seemann W, Krauss T, Mix M, Schiller F, Wetterauer U, Werner M, Langer M, Bock M, Meyer PT, Grosu AL (2017) Comparison of (68)Ga-HBED-CC PSMA-PET/CT and multiparametric MRI for gross tumour volume detection in patients with primary prostate cancer based on slice by slice comparison with histopathology. Theranostics 7(1):228–237. https://doi.org/10.7150/thno.16638
Funding
No funding was received for this work.
Author information
Authors and Affiliations
Contributions
SK: protocol/project development; data collection or management; data analysis; manuscript writing/editing; statistical analysis. SK: protocol/project development; data collection or management; data analysis; manuscript writing/editing. SG: protocol/project development; data collection or management; data analysis; manuscript writing/editing; statistical analysis. TH: data analysis; manuscript writing/editing; statistical analysis. WMT: data analysis; manuscript writing/editing; statistical analysis. JH: data collection or management; manuscript writing/editing. JS: data analysis; manuscript writing/editing. MS: data collection or management; manuscript writing/editing. KN: data analysis; manuscript writing/editing; administrative, technical, or material support; supervision. AS: manuscript writing/editing; supervision. GR: protocol/project development; manuscript writing/editing; administrative, technical, or material support. CF: data analysis; manuscript writing/editing; administrative, technical, or material support; supervision. JB: protocol/project development; data analysis; manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee, approved by the local institutional review board (241/2012MPG23), and in line with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaufmann, S., Kruck, S., Gatidis, S. et al. Simultaneous whole-body PET/MRI with integrated multiparametric MRI for primary staging of high-risk prostate cancer. World J Urol 38, 2513–2521 (2020). https://doi.org/10.1007/s00345-019-03066-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-03066-1