Abstract
Objectives
This study investigated the discriminability of quantitative radiomics features extracted from cardiac magnetic resonance (CMR) images for hypertrophic cardiomyopathy (HCM), dilated cardiomyopathy (DCM), and healthy (NOR) patients.
Methods
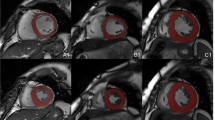
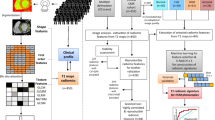
The data of two hundred and eighty-three patients with HCM (n = 48) or DCM (n = 52) and NOR (n = 123) were extracted from two publicly available datasets. Ten feature selection methods were first performed on twenty-one different sets of radiomics features extracted from the left ventricle, right ventricle, and myocardium segmented from CMR images in the end-diastolic frame, end-systolic frame, and a combination of both; then, nine classical machine learning methods were trained with the selected radiomics features to distinguish HCM, DCM, and NOR. Ninety classification models were constructed based on combinations of the ten feature selection methods and nine classifiers. The classification models were evaluated, and the optimal model was selected. The diagnostic performance of the selected model was also compared to that of state-of-the-art methods.
Results
The random forest minimum redundancy maximum relevance model with features based on LeastAxisLength, Maximum2DDiameterSlice, Median, MinorAxisLength, Sphericity, VoxelVolume, Kurtosis, Flatness, and Skewness was the highest performing model, achieving 91.2% classification accuracy. The cross-validated areas under the curve on the test dataset were 0.938, 0.966, and 0.936 for NOR, DCM, and HCM, respectively. Furthermore, compared with those of the state-of-the-art methods, the sensitivity and accuracy of this model were greatly improved.
Conclusions
A predictive model was proposed based on CMR radiomics features for classifying HCM, DCM, and NOR patients. The model had good discriminability.
Key Points
• The first-order features and the features extracted from the LOG-filtered images have potential in distinguishing HCM patients from DCM patients.
• The features extracted from the RV play little role in distinguishing DCM from HCM.
• The VoxelVolume of the myocardium in the ED frame is important in the recognition of DCM.






Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve
- CMR:
-
Cardiac magnetic resonance
- DCM:
-
Dilated cardiomyopathy
- DT:
-
Decision tree
- ED:
-
End-diastole
- EL:
-
Ensemble learning
- ES:
-
End-systole
- EUDT:
-
Euclidean distance
- EUDT:
-
Euclidean distance
- FAOV:
-
F-ANOVA
- GINI:
-
Gini index
- GLCM:
-
Gray level co-occurrence matrix
- GLRLM:
-
Gray level run length matrix
- GLSZM:
-
Gray level size zone matrix
- GNRO:
-
Gain ratio
- HCM:
-
Hypertrophic cardiomyopathy
- ICC:
-
Intraclass correlation coefficient
- IFGN:
-
Information gain
- JMI:
-
Joint mutual information
- KNN:
-
K-nearest neighbor
- LOG:
-
Laplacian of Gaussian-filtered
- LR:
-
Logistic regression
- LV:
-
Left ventricle
- MIM:
-
Mutual information maximization
- MLP:
-
Multilayer perceptron
- MRMR:
-
Minimum redundancy maximum relevance
- MUIF:
-
Mutual information feature selection
- MYO:
-
Myocardium
- NB:
-
Naive Bayes
- NGDTM:
-
Neighboring gray tone difference
- ROI:
-
Region of interest
- SSFP:
-
Steady-state free procession
References
Santulli G (2013) Epidemiology of cardiovascular disease in the 21st century: updated updated numbers and updated facts. J Cardiovasc Dis Res 1:1–2
**a C, Li X, Wang X et al (2019) A multi-modality network for cardiomyopathy death risk prediction with CMR images and clinical information. Medical image computing and computer assisted intervention – MICCAI 2019, Shenzhen, China, 13-17 Oct 2019. Available via https://cse.buffalo.edu/~siweilyu/papers/miccai19.pdf
Sundaram DSB, Arunachalam SP, Damani DN et al (2021) Natural language processing based machine learning model using cardiac MRI reports to identify hypertrophic cardiomyopathy patients. 2021 design of medical devices conference, Minneapolis, USA. https://doi.org/10.1115/DMD2021-1076
Alis D, Yergin M, Asmakutlu O et al (2021) The influence of cardiac motion on radiomics features: radiomics features of non-enhanced CMR cine images greatly vary through the cardiac cycle. Eur Radiol 31:2706–2715
Luo C, Shi CH, Li XJ, Wang X, Chen YC, Gao DR, Yin YB, Song Q, Wu X, Zhou JL (2020) Multi-task learning using attention-based convolutional encoder–decoder for dilated cardiomyopathy CMR segmentation and classification. Cmc-Comput Mater Con 63:995–1012
Karamitsos TD, Francis JM, Myerson S, Selvanayagam JB, Neubauer S (2009) The role of cardiovascular magnetic resonance imaging in heart failure. J Am Coll Cardiol 54:1407–1424
Loecher M, Perotti LE, Ennis DB (2021) Using synthetic data generation to train a cardiac motion tag tracking neural network. Med Image Anal 74:102223
Antonopoulos AS, Boutsikou M, Simantiris S et al (2021) Machine learning of native T1 map** radiomics for classification of hypertrophic cardiomyopathy phenotypes. Sci Rep 11:1–11
Śpiewak M, Kłopotowski M, Ojrzyńska N et al (2021) Impact of cardiac magnetic resonance on the diagnosis of hypertrophic cardiomyopathy-a 10-year experience with over 1000 patients. Eur Radiol 31:1194–1205
Farahani NZ, Sundaram DSB, Enayati M, Arunachalam SP, Pasupathy K, Arruda-Olson AM (2020) Explanatory analysis of a machine learning model to identify hypertrophic cardiomyopathy patients from EHR using diagnostic codes. 2020 IEEE international conference on bioinformatics and biomedicine (BIBM), Seoul, Korea (South), 16-19 Dec 2020. https://doi.org/10.1109/BIBM49941.2020.9313231
Luo C, **n W, Li XJ, Chen YC, Zhou JL, Cao KL, Yin YB, Song Q, Wu X (2019) ACNET: attention-based convolution network with additional discriminative features for DCM classification (S). The 31st international conference on software engineering and knowledge engineering (SEKE 2019), Lisbon, Portugal, 10-12 July 2019. Available via http://ksiresearch.org/seke/seke19paper/seke19paper_155.pdf
Wolterink J, Leiner T, Viergever M, Isgum I (2017) Automatic segmentation and disease classification using cardiac cine MR images. Statistical atlases and computational models of the heart. ACDC and MMWHS challenges. STACOM 2017, Quebec City, Canada, 10-14 September 2017. Available via https://arxiv.org/pdf/1708.01141.pdf
Isensee F, Jaeger P, Full P, Wolf I, Engelhardt S, Maier-Hein K (2017) Automatic cardiac disease assessment on cine-MRI via time-series segmentation and domain specific features. Statistical atlases and computational models of the heart. ACDC and MMWHS challenges. STACOM 2017, Quebec City, Canada, 10-14 September 2017. Available via https://arxiv.org/pdf/1707.00587.pdf
Khened M, Alex V, Krishnamurthi G (2017) Densely connected fully convolutional network for short-axis cardiac cine MR image segmentation and heart diagnosis using random forest. Statistical atlases and computational models of the heart. ACDC and MMWHS challenges. STACOM 2017, Quebec City, Canada, 10-14 September 2017. https://doi.org/10.1007/978-3-319-75541-0_1
Zheng Q, Delingette H, Ayache N (2019) Explainable cardiac pathology classification on cine MRI with motion characterization by semi-supervised learning of apparent flow. Med Image Anal 56:80–95
Cetin I, Sanroma G, Petersen S, Napel S, Camara O, Ballester M, Lekadir K (2017) A radiomics approach to computer-aided diagnosis with cardiac cine-MRI. Statistical atlases and computational models of the heart. ACDC and MMWHS challenges. STACOM 2017, Quebec City, Canada, 10-14 September 2017. Available via https://arxiv.org/pdf/1909.11854.pdf
Chang YK, Jung C (2020) Automatic cardiac MRI segmentation and permutation-invariant pathology classification using deep neural networks and point clouds. Neurocomputing 418:270–279
Thermos S, Liu X, O’Neil A et al (2021) Controllable cardiac synthesis via disentangled anatomy arithmetic. Proc. In: 24th Int Conf on Medical Image Computing and Computer-Assisted Intervention, p, pp 160–170
Kolossváry M, Kellermayer M, Merkely B, Maurovich-Horvat P (2018) Cardiac computed tomography radiomics: a comprehensive review on radiomic techniques. J Thorac Imaging 33:26–34
Al-Mallah MH (2019) Radiomics in hypertrophic cardiomyopathy: the new tool. JACC-Cardiovasc Imag 12:1955–1957
Xu P, Xue Y, Schoepf UJ, Varga-Szemes A, Griffith J, Yacoub B, Zhou F, Zhou C, Yang Y, **ng W, Zhang L (2021) Radiomics: the next frontier of cardiac computed tomography. Circ Cardiovasc Imag 14:e011747
Leiner T (2020) Radiomics in cardiac MRI: Sisyphean Struggle or Close to the Summit of Olympus? Radiol Cardiothorac Imag 2:e200244
Ponsiglione A, Stanzione A, Cuocolo R, Ascione R, Gambardella M, De Giorgi M, Nappi C, Cuocolo A, Imbriaco M (2021) Cardiac CT and MRI radiomics: systematic review of the literature and radiomics quality score assessment. Eur Radiol 32:2629–2638
Tautz L, Zhang H, Hüllebrand M, Ivantsits M, Kelle S, Kuehne T, Falk V, Hennemuth A (2020) Cardiac radiomics: an interactive approach for 4D data exploration. Curr Dir Biomed Eng 6:20200008
Fei JL, Pu CL, Xu FY, Wu Y, Hu HJ (2021) Progress in radiomics of common heart disease based on cardiac magnetic resonance imaging. J Mol Clin Med 4:29–38
Martin-Isla C, Campello VM, Izquierdo C, Raisi-Estabragh Z, Baeßler B, Petersen SE, Lekadir K (2020) Image-based cardiac diagnosis with machine learning: a review. Front Cardiovasc Med 7:1
Schofield R, Ganeshan B, Fontana M, Nasis A, Castelletti S, Rosmini S, Treibel TA, Manisty C, Endozo R, Groves A, Moon JC (2019) Texture analysis of cardiovascular magnetic resonance cine images differentiates aetiologies of left ventricular hypertrophy. Clin Radiol 74:140–149
Hassani C, Saremi F, Varghese BA, Duddalwar V (2020) Myocardial radiomics in cardiac MRI. AJR Am J Roentgenol 214:536–545
Di Noto T, von Spiczak J, Mannil M, Gantert E, Soda P, Manka R, Alkadhi H (2019) Radiomics for distinguishing myocardial infarction from myocarditis at late Gadolinium enhancement at MRI: comparison with subjective visual analysis. Radiol Cardiothorac Imag 1:e180026
Larroza A, López-Lereu MP, Monmeneu JV, Bodí V, Moratal D (2017) Texture analysis for infarcted myocardium detection on delayed enhancement MRI. 2017 IEEE 14th international symposium on biomedical imaging (ISBI 2017), Melbourne, Australia, 18-21 April 2017. https://doi.org/10.1109/ISBI.2017.7950700
Avard E, Shiri I, Hajianfar G, Abdollahi H, Kalantari KR (2022) Non-contrast cine cardiac magnetic resonance image radiomics features and machine learning algorithms for myocardial infarction detection. Comput Biol Med 141:105145
Raisi-Estabragh Z, Izquierdo C, Campello VM, Martin-Isla C, Jaggi A, Harvey NC, Lekadir K, Petersen SE (2020) Cardiac magnetic resonance radiomics: basic principles and clinical perspectives. Eur Heart J Cardiovasc Imaging 21:349–356
Yang L, Dong D, Fang M, Zhu Y, Zang Y, Liu Z, Zhang H, Ying J, Zhao X, Tian J (2018) Can CT-based radiomics signature predict KRAS/NRAS/BRAF mutations in colorectal cancer? Eur Radiol 28:058–2067
Peng H, Long F, Ding C (2005) Feature selection based on mutual information criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans Pattern Anal Mach Intell 27:1226–1238
Saeys Y, Inza I, Larranaga P (2007) A review of feature selection techniques in bioinformatics. Bioinformatics 23:2507–2517
Brown G, Pocock A, Zhao MJ, Luján M (2012) Conditional likelihood maximisation: a unifying framework for information theoretic feature selection. J Mach Learn Res 13:27–66
Zhao Z, Morstatter F, Sharma S, Alelyani S, Anand A, Liu H (2010) Advancing feature selection research. ASU feature selection repository. Tempe, AZ: School of Computing, Informatics, and Decision Systems Engineering, Arizona State University, 2010, 1-28. Available via https://www.researchgate.net/publication/305083748_Advancing_feature_selection_research
Parmar C, Grossmann P, Bussink J, Lambin P, Aerts HJ (2015) Machine learning methods for quantitative radiomic biomarkers. Sci Rep 5:1–11
Breiman L (2001) Random forests. Mach Learn 45:5–32
Weintraub RG, Semsarian C, Macdonald P (2017) Dilated cardiomyopathy. Lancet 390:400–414
Vidal-Sospedra I, Ruiz-España S, Piñeiro-Vidal T, Santabárbara JM, Maceira A, Moratal D (2020) Determination of image-based biomarkers for the diagnosis of hypertrophic cardiomyopathy, hypertensive cardiomyopathy and amyloidosis from texture analysis in cardiac MRI. 2020 IEEE 20th international conference on bioinformatics and bioengineering (BIBE). Virtual conference, America, 26-28 Oct. 2020. https://doi.org/10.1109/BIBE50027.2020.00045
Geske JB, Ommen SR, Gersh BJ (2018) Hypertrophic cardiomyopathy: clinical update. JACC Heart Fail 6:364–375
Lu P, Qiu H, Qin C, Bai W, Rueckert D, Noble JA (2020) Going deeper into cardiac motion analysis to model fine spatio-temporal features. Annual conference on medical image understanding and analysis. Oxford, United Kingdom, 15-17 July 2020. https://doi.org/10.1007/978-3-030-52791-4_23
Lu P, Bai W, Rueckert D, Nhearoble JA (2021) Dynamic spatio-temporal graph convolutional networks for cardiac motion analysis. 2021 IEEE 18th international symposium on biomedical imaging (ISBI). Virtual conference, French, 13-16 April 2021. https://doi.org/10.1109/ISBI48211.2021.9433890
Burrage MK, Ferreira VM (2020) Cardiovascular magnetic resonance for the differentiation of left ventricular hypertrophy. Curr Heart Fail Rep 17:192–204
Upendra RR, Wentz BJ, Simon R, Shontz SM, Linte CA (2021) CNN-based cardiac motion extraction to generate deformable geometric left ventricle myocardial models from cine MRI. International Conference on Functional Imaging and Modeling of the Heart. Virtual Conference, America, 21-25
Acknowledgements
The authors would like to express appreciation to American Journal Experts for providing linguistic assistance during the preparation of this paper.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 62172047, 61802020).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Shifeng Zhao.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
Shifeng Zhao and **aoxuan Zhang have significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• observational
• multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Cui, C., Zhao, S. et al. Cardiac magnetic resonance radiomics for disease classification. Eur Radiol 33, 2312–2323 (2023). https://doi.org/10.1007/s00330-022-09236-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-09236-x