Abstract
Objective
To investigate the feasibility of using amide proton transfer (APT) magnetic resonance imaging (MRI) in the liver and to evaluate its ability to characterize focal liver lesions (FLLs).
Methods
A total of 203 patients with suspected FLLs who underwent APT imaging at 3T were included. APT imaging was obtained using a single-slice turbo spin-echo sequence to include FLLs through five breath-holds, and its acquisition time was approximately 1 min. APT signals in the background liver and FLL were measured with magnetization transfer ratio asymmetry (MTRasym) at 3.5 ppm. The technical success rate of APT imaging and the reasons for failure to obtain meaningful MTRasym values were assessed. The Mann Whitney U test was used to compare MTRasym values between different FLLs.
Results
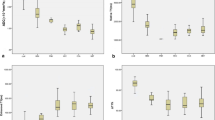
The technical success rate of APT imaging in the liver was 62.1% (126/203). The reasons for failure were a too large B0 inhomogeneity (n = 43), significant respiratory motion (n = 12), and these two factors together (n = 22), respectively. Among 59 FLLs with analyzable APT images, MTRasym values were compared between 27 patients with liver metastases and 23 patients with hepatocellular carcinomas (HCCs). The MTRasym values of metastases were significantly higher than those of HCC (0.13 ± 2.15% vs. − 1.41 ± 3.68%, p = 0.027).
Conclusions
APT imaging could be an imaging biomarker for the differentiation of FLLs. However, further technical improvement is required before APT imaging can be clinically applied to liver MRI.
Key Points
• Liver APT imaging was technically feasible, but with a relatively low success rate (62.1%).
• Liver metastases showed higher APT values than hepatocellular carcinomas.






Similar content being viewed by others
Abbreviations
- APT:
-
Amide proton transfer
- CEST:
-
Chemical exchange saturation transfer
- CoV:
-
Coefficient of variation
- FLL:
-
Focal liver lesion
- FNH:
-
Focal nodular hyperplasia
- HCC:
-
Hepatocellular carcinoma
- MRI:
-
Magnetic resonance imaging
- MTR asym :
-
Magnetization transfer ratio asymmetry
- RF:
-
Radiofrequency
- ROI:
-
Region of interest
- TSE:
-
Turbo spin-echo
References
van Zijl PC, Yadav NN (2011) Chemical exchange saturation transfer (CEST): what is in a name and what isn’t? Magn Reson Med 65:927–948
Ward KM, Aletras AH, Balaban RS (2000) A new class of contrast agents for MRI based on proton chemical exchange dependent saturation transfer (CEST). J Magn Reson 143:79–87
Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090
Jones KM, Pollard AC, Pagel MD (2018) Clinical applications of chemical exchange saturation transfer (CEST) MRI. J Magn Reson Imaging 47:11–27
Park JE, Lee JY, Kim HS et al (2018) Amide proton transfer imaging seems to provide higher diagnostic performance in post-treatment high-grade gliomas than methionine positron emission tomography. Eur Radiol 28:3285–3295
Jeong HK, Han K, Zhou J et al (2017) Characterizing amide proton transfer imaging in haemorrhage brain lesions using 3T MRI. Eur Radiol 27:1577–1584
Jones CK, Schlosser MJ, van Zijl PC, Pomper MG, Golay X, Zhou J (2006) Amide proton transfer imaging of human brain tumors at 3T. Magn Reson Med 56:585–592
Joo B, Han K, Choi YS et al (2018) Amide proton transfer imaging for differentiation of benign and atypical meningiomas. Eur Radiol 28:331–339
Zhou J, Heo HY, Knutsson L, van Zijl PCM, Jiang S (2019) APT-weighted MRI: techniques, current neuro applications, and challenging issues. J Magn Reson Imaging 50:347–364
Deng M, Chen SZ, Yuan J, Chan Q, Zhou J, Wang YX (2016) Chemical exchange saturation transfer (CEST) MR technique for liver imaging at 3.0 Tesla: an evaluation of different offset number and an after-meal and over-night-fast comparison. Mol Imaging Biol 18:274–282
Chen SZ, Yuan J, Deng M, Wei J, Zhou J, Wang YX (2016) Chemical exchange saturation transfer (CEST) MR technique for in-vivo liver imaging at 3.0 tesla. Eur Radiol 26:1792–1800
Lin Y, Luo X, Yu L et al (2019) Amide proton transfer-weighted MRI for predicting histological grade of hepatocellular carcinoma: comparison with diffusion-weighted imaging. Quant Imaging Med Surg 9:1641–1651
Choi YS, Ahn SS, Lee SK et al (2017) Amide proton transfer imaging to discriminate between low- and high-grade gliomas: added value to apparent diffusion coefficient and relative cerebral blood volume. Eur Radiol 27:3181–3189
Zhou J, Lal B, Wilson DA, Laterra J, van Zijl PC (2003) Amide proton transfer (APT) contrast for imaging of brain tumors. Magn Reson Med 50:1120–1126
Chung SR, Lee SS, Kim N et al (2015) Intravoxel incoherent motion MRI for liver fibrosis assessment: a pilot study. Acta Radiol 56:1428–1436
Kim HC, Seo N, Chung YE, Park MS, Choi JY, Kim MJ (2019) Characterization of focal liver lesions using the stretched exponential model: comparison with monoexponential and biexponential diffusion-weighted magnetic resonance imaging. Eur Radiol 29:5111–5120
Danet IM, Semelka RC, Leonardou P et al (2003) Spectrum of MRI appearances of untreated metastases of the liver. AJR Am J Roentgenol 181:809–817
Bruix J, Sherman M (2011) Management of hepatocellular carcinoma: an update. Hepatology 53:1020–1022
Seale MK, Catalano OA, Saini S, Hahn PF, Sahani DV (2009) Hepatobiliary-specific MR contrast agents: role in imaging the liver and biliary tree. Radiographics 29:1725–1748
Zhou J (2011) Amide proton transfer imaging of the human brain. Methods Mol Biol 711:227–237
Zhu H, Jones CK, van Zijl PC, Barker PB, Zhou J (2010) Fast 3D chemical exchange saturation transfer (CEST) imaging of the human brain. Magn Reson Med 64:638–644
Zhou J, Zhu H, Lim M et al (2013) Three-dimensional amide proton transfer MR imaging of gliomas: initial experience and comparison with gadolinium enhancement. J Magn Reson Imaging 38:1119–1128
Wen Z, Hu S, Huang F et al (2010) MR imaging of high-grade brain tumors using endogenous protein and peptide-based contrast. Neuroimage 51:616–622
Zhou J, Blakeley JO, Hua J et al (2008) Practical data acquisition method for human brain tumor amide proton transfer (APT) imaging. Magn Reson Med 60:842–849
Yuan J, Chen S, King AD et al (2014) Amide proton transfer-weighted imaging of the head and neck at 3 T: a feasibility study on healthy human subjects and patients with head and neck cancer. NMR Biomed 27:1239–1247
Wei W, Jia G, Flanigan D, Zhou J, Knopp MV (2014) Chemical exchange saturation transfer MR imaging of articular cartilage glycosaminoglycans at 3 T: accuracy of B0 field inhomogeneity corrections with gradient echo method. Magn Reson Imaging 32:41–47
Kuai ZX, Sang XQ, Yao YF, Chu CY, Zhu YM (2019) Evaluation of non-monoexponential diffusion models for hepatocellular carcinoma using b values up to 2000 s/mm(2): a short-term repeatability study. J Magn Reson Imaging 50:297–304
Barnhart HX, Barboriak DP (2009) Applications of the repeatability of quantitative imaging biomarkers: a review of statistical analysis of repeat data sets. Transl Oncol 2:231–235
Hallgren KA (2012) Computing inter-rater reliability for observational data: an overview and tutorial. Tutor Quant Methods Psychol 8:23–34
Sun PZ, Benner T, Kumar A, Sorensen AG (2008) Investigation of optimizing and translating pH-sensitive pulsed-chemical exchange saturation transfer (CEST) imaging to a 3T clinical scanner. Magn Reson Med 60:834–841
Kim J, Wu Y, Guo Y, Zheng H, Sun PZ (2015) A review of optimization and quantification techniques for chemical exchange saturation transfer MRI toward sensitive in vivo imaging. Contrast Media Mol Imaging 10:163–178
Wang QB, Zhu H, Liu HL, Zhang B (2012) Performance of magnetic resonance elastography and diffusion-weighted imaging for the staging of hepatic fibrosis: a meta-analysis. Hepatology 56:239–247
Lee JB, Park JE, Jung SC et al (2020) Repeatability of amide proton transfer-weighted signals in the brain according to clinical condition and anatomical location. Eur Radiol 30:346–356
Feng L, Benkert T, Block KT, Sodickson DK, Otazo R, Chandarana H (2017) Compressed sensing for body MRI. J Magn Reson Imaging 45:966–987
Heo HY, Zhang Y, Lee DH, Jiang S, Zhao X, Zhou J (2017) Accelerating chemical exchange saturation transfer (CEST) MRI by combining compressed sensing and sensitivity encoding techniques. Magn Reson Med 77:779–786
Seo N, Chung YE, Park YN, Kim E, Hwang J, Kim MJ (2018) Liver fibrosis: stretched exponential model outperforms mono-exponential and bi-exponential models of diffusion-weighted MRI. Eur Radiol 28:2812–2822
Ohno Y, Yui M, Koyama H et al (2016) Chemical exchange saturation transfer MR imaging: preliminary results for differentiation of malignant and benign thoracic lesions. Radiology 279:578–589
Nishie A, Asayama Y, Ishigami K et al (2019) Amide proton transfer imaging to predict tumor response to neoadjuvant chemotherapy in locally advanced rectal cancer. J Gastroenterol Hepatol 34:140–146
Ray KJ, Simard MA, Larkin JR et al (2019) Tumor pH and protein concentration contribute to the signal of amide proton transfer magnetic resonance imaging. Cancer Res 79:1343–1352
van Zijl PC, Zhou J, Mori N, Payen JF, Wilson D, Mori S (2003) Mechanism of magnetization transfer during on-resonance water saturation. A new approach to detect mobile proteins, peptides, and lipids. Magn Reson Med 49:440–449
Heo HY, Xu X, Jiang S et al (2019) Prospective acceleration of parallel RF transmission-based 3D chemical exchange saturation transfer imaging with compressed sensing. Magn Reson Med 82:1812–1821
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yong Eun Chung.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
Nieun Seo performed statistical analysis, who is one of the coauthors.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 516 kb)
Rights and permissions
About this article
Cite this article
Seo, N., Jeong, HK., Choi, JY. et al. Liver MRI with amide proton transfer imaging: feasibility and accuracy for the characterization of focal liver lesions. Eur Radiol 31, 222–231 (2021). https://doi.org/10.1007/s00330-020-07122-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07122-y