Abstract
Key message
Here, we report potential transcripts involved in the biosynthesis of therapeutic metabolites in Swertia japonica , the first report of transcriptome assembly, and characterization of the medicinal plant from Swertia genus.
Abstract
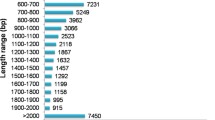
Swertia genus, representing over 170 plant species including herbs such as S. chirata, S. hookeri, S. longifolia, S. japonica, among others, have been used as the traditional medicine in China, India, Korea, and Japan for thousands of years. Due to the lack of genomic and transcriptomic resources, little is known about the molecular basis involved in the biosynthesis of characteristic key bioactive metabolites. Here, we performed deep-transcriptome sequencing for the aerial tissues and the roots of S. japonica, generating over 2 billion raw reads with an average length of 101 bps. Using a combined approach of three popular assemblers, de novo transcriptome assembly for S. japonica was obtained, yielding 81,729 unigenes having an average length of 884 bps and N50 value of 1452 bps, of which 46,963 unigenes were annotated based on the sequence similarity against NCBI-nr protein database. Annotation of transcriptome assembly resulted in the identification of putative genes encoding all enzymes from the key therapeutic metabolite biosynthesis pathways. Transcript abundance analysis, gene ontology enrichment analysis, and KEGG pathway enrichment analysis revealed metabolic processes being up-regulated in the aerial tissues with respect to the roots of S. japonica. We also identified 37 unigenes as potential candidates involved in the glycosylation of bioactive metabolites. Being the first report of transcriptome assembly and annotation for any of the Swertia species, this study will be a valuable resource for future investigations on the biosynthetic pathways of therapeutic metabolites and their regulations.










Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Basnet P, Kadota S, Shimizu M, Namba T (1994) Bellidifolin: a potent hypoglycemic agent in streptozotocin (STZ)-induced diabetic rats from Swertia japonica. Planta Med 60:507–511
Benning C, Ohta H (2005) Three enzyme systems for galactoglycerolipid biosynthesis are coordinately regulated in plants. J Biol Chem 280:2397–2400
Bertrand G (1926) The importance of minute chemical constituents (infiniment petits chimiques) of biological products: nickel, cobalt and insulin. Science 64:629–630
Bogs J, Jaffe FW, Takos AM, Walker AR, Robinson SP (2007) The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development. Plant Physiol 143:1347–1361
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120
Bradnam KR, Fass JN, Alexandrov A, Baranay P, Bechner M, Birol I, Boisvert S, Chapman JA, Chapuis G, Chikhi R, Chitsaz H, Chou WC, Corbeil J, Del Fabbro C, Docking TR, Durbin R, Earl D, Emrich S, Fedotov P, Fonseca NA, Ganapathy G, Gibbs RA, Gnerre S, Godzaridis E, Goldstein S, Haimel M, Hall G, Haussler D, Hiatt JB, Ho IY, Howard J, Hunt M, Jackman SD, Jaffe DB, Jarvis ED, Jiang H, Kazakov S, Kersey PJ, Kitzman JO, Knight JR, Koren S, Lam TW, Lavenier D, Laviolette F, Li Y, Li Z, Liu B, Liu Y, Luo R, Maccallum I, Macmanes MD, Maillet N, Melnikov S, Naquin D, Ning Z, Otto TD, Paten B, Paulo OS, Phillippy AM, Pina-Martins F, Place M, Przybylski D, Qin X, Qu C, Ribeiro FJ, Richards S, Rokhsar DS, Ruby JG, Scalabrin S, Schatz MC, Schwartz DC, Sergushichev A, Sharpe T, Shaw TI, Shendure J, Shi Y, Simpson JT, Song H, Tsarev F, Vezzi F, Vicedomini R, Vieira BM, Wang J, Worley KC, Yin S, Yiu SM, Yuan J, Zhang G, Zhang H, Zhou S, Korf IF (2013) Assemblathon 2: evaluating de novo methods of genome assembly in three vertebrate species. GigaScience 2:10
Brahmachari G, Mondal S, Gangopadhyay A, Gorai D, Mukhopadhyay B, Saha S, Brahmachari AK (2004) Swertia (Gentianaceae): chemical and pharmacological aspects. Chem Biodivers 1:1627–1651
Cao TW, Geng CA, Ma YB, He K, Wang HL, Zhou NJ, Zhang XM, Tao YD, Chen JJ (2013) Xanthones with anti-hepatitis B virus activity from Swertia mussotii. Planta Med 79:697–700
Conesa A, Gotz S (2008) Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int J Plant Genom 2008:619832
Conesa A, Gotz S, Garcia-Gomez JM, Terol J, Talon M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
Duan J, **a C, Zhao G, Jia J, Kong X (2012) Optimizing de novo common wheat transcriptome assembly using short-read RNA-Seq data. BMC Genom 13:392
Dutt B, Srivastava LJ, Singh JM (1996) Swertia spp: a source of bitter compounes for medicinal use. Anc Sci Life 15:226–229
Eddouks M, Chattopadhyay D, De Feo V, Cho WC (2014) Medicinal plants in the prevention and treatment of chronic diseases 2013. Evid Based Complement Alter Med eCAM 2014:180981
Edgar R, Domrachev M, Lash AE (2002) Gene expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30:207–210
Fu L, Niu B, Zhu Z, Wu S, Li W (2012) CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics 28:3150–3152
Fukushima A, Nakamura M, Suzuki H, Saito K, Yamazaki M (2015) High-throughput sequencing and de novo assembly of red and green forms of the Perilla frutescens var. crispa transcriptome. PLoS One 10:e0129154
Gene Ontology C (2015) Gene ontology consortium: going forward. Nucleic Acids Res 43:D1049–D1056
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Gruenheit N, Deusch O, Esser C, Becker M, Voelckel C, Lockhart P (2012) Cutoffs and k-mers: implications from a transcriptome study in allopolyploid plants. BMC Genom 13:92
Hancock JM (1996) Simple sequences in a “minimal” genome. Nat Genet 14:14–15
Hase K, Li J, Basnet P, **ong Q, Takamura S, Namba T, Kadota S (1997) Hepatoprotective principles of Swertia japonica Makino on d-galactosamine/lipopolysaccharide-induced liver injury in mice. Chem Pharm Bull 45:1823–1827
Hiramatsu N, **ufen W, Takechi R, Itoh Y, Mamo J, Pal S (2004) Antimutagenicity of Japanese traditional herbs, gennoshoko, yomogi, senburi and iwa-tobacco. BioFactors 22:123–125
Hou B, Lim EK, Higgins GS, Bowles DJ (2004) N-glucosylation of cytokinins by glycosyltransferases of Arabidopsis thaliana. J Biol Chem 279:47822–47832
Ikeshiro Y, Tomita Y (1985) Iridoid glucoside of Swertia japonica. Planta Med 51:390–393
Ikeshiro Y, Tomita Y (1987) Senburiside II, a new iridoid glucoside from Swertia japonica. Planta Med 53:158–161
Jarvis DE, Kopp OR, Jellen EN, Mallory MA, Pattee J, Bonifacio A, Coleman CE, Stevens MR, Fairbanks DJ, Maughan PJ (2008) Simple sequence repeat marker development and genetic map** in quinoa (Chenopodium quinoa Willd.). J Genet 87:39–51
Jiang W, Zhu DL, Wang MF, Yang QS, Zuo MY, Zeng L, Li GP (2015) Xanthones from the herb of Swertia elata and their anti-TMV activity. Nat Product Res 1–6
Jones P, Messner B, Nakajima J, Schaffner AR, Saito K (2003) UGT73C6 and UGT78D1, glycosyltransferases involved in flavonol glycoside biosynthesis in Arabidopsis thaliana. J Biol Chem 278:43910–43918
Jones P, Binns D, Chang HY, Fraser M, Li W, McAnulla C, McWilliam H, Maslen J, Mitchell A, Nuka G, Pesseat S, Quinn AF, Sangrador-Vegas A, Scheremetjew M, Yong SY, Lopez R, Hunter S (2014) InterProScan 5: genome-scale protein function classification. Bioinformatics 30:1236–1240
Kakiuchi N, Iwaki N, Mikage M, **ao H, Wang Z, Hattori M (2014) Phylogenetic examination of crude drugs derived from Yunnanese Swertia plants. J Nat Med 68:206–210
Kanehisa M, Goto S, Sato Y, Kawashima M, Furumichi M, Tanabe M (2014) Data, information, knowledge and principle: back to metabolism in KEGG. Nucleic Acids Res 42:D199–D205
Kavimani S, Manisenthlkumar KT (2000) Effect of methanolic extract of Enicostemma littorale on Dalton’s ascitic lymphoma. J Ethnopharmacol 71:349–352
Kelly RJ, Vincent DE, Friedberg I (2010) IPRStats: visualization of the functional potential of an InterProScan run. BMC Bioinform 11(Suppl 12):S13
Khanal S, Shakya N, Thapa K, Pant DR (2015) Phytochemical investigation of crude methanol extracts of different species of Swertia from Nepal. BMC Res Notes 8:821
Kikuchi M, Kikuchi M (2004) Studies on the constituents of Swertia japonica MAKINO I. On the structures of new secoiridoid diglycosides. Chem Pharm Bull 52:1210–1214
Kikuchi M, Kikuchi M (2005) Studies on the constituents of Swertia japonica Makino II. On the structures of new glycosides. Chem Pharm Bull 53:48–51
Kimura Y, Sumiyoshi M (2011) Effects of Swertia japonica extract and its main compound swertiamarin on gastric emptying and gastrointestinal motility in mice. Fitoterapia 82:827–833
Komatsu M, Tomimori T, Makiguchi Y, Asano K (1968) Studies on the constituents of Swertia japonica. 3. On the flavonoid constituents of the plants of Swertia spp. Yakugaku zasshi J Pharm Soc Jpn 88:832–837
Komatsu M, Tomimori T, Mikuriya N (1969) Studies on the constituents of Swertia japonica. IV. Isolation and structure of xanthones. Chem Pharm Bull 17:155–162
Kshirsagar PR, Pai SR, Nimbalkar MS, Gaikwad NB (2016) RP-HPLC analysis of seco-iridoid glycoside swertiamarin from different Swertia species. Nat Prod Res 30:865–868
Kumar S, Blaxter ML (2010) Comparing de novo assemblers for 454 transcriptome data. BMC Genom 11:571
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform 12:323
Li W, Godzik A (2006) Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22:1658–1659
Liang JZ (1984) The active constituents of Swertia patens. Zhong yao tong bao 9:34–36
Licciardi PV, Underwood JR (2011) Plant-derived medicines: a novel class of immunological adjuvants. Int Immunopharmacol 11:390–398
Lim CE, Choi JN, Kim IA, Lee SA, Hwang YS, Lee CH, Lim J (2008) Improved resistance to oxidative stress by a loss-of-function mutation in the Arabidopsis UGT71C1 gene. Mol Cells 25:368–375
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Medda S, Mukhopadhyay S, Basu MK (1999) Evaluation of the in vivo activity and toxicity of amarogentin, an antileishmanial agent, in both liposomal and niosomal forms. J Antimicrob Chemother 44:791–794
Menkovic N, Savikin-Fodulovic K, Bulatovic V, Aljancic I, Juranic N, Macura S, Vajs V, Milosavljevic S (2002) Xanthones from Swertia punctata. Phytochemistry 61:415–420
Miettinen K, Dong L, Navrot N, Schneider T, Burlat V, Pollier J, Woittiez L, van der Krol S, Lugan R, Ilc T, Verpoorte R, Oksman-Caldentey KM, Martinoia E, Bouwmeester H, Goossens A, Memelink J, Werck-Reichhart D (2014) The seco-iridoid pathway from Catharanthus roseus. Nat Commun 5:3606
Misra A, Shasany AK, Shukla AK, Darokar MP, Singh SC, Sundaresan V, Singh J, Bagchi GD, Jain SP, Saikia D, Khanuja SP (2010) AFLP markers for identification of Swertia species (Gentianaceae). Genet Mol Res GMR 9:1535–1544
Mulder N, Apweiler R (2007) InterPro and InterProScan: tools for protein sequence classification and comparison. Methods Mol Biol 396:59–70
Muranaka T, Saito K (2013) Phytochemical genomics on the way. Plant Cell Physiol 54:645–646
Nakasugi K, Crowhurst R, Bally J, Waterhouse P (2014) Combining transcriptome assemblies from multiple de novo assemblers in the allo-tetraploid plant Nicotiana benthamiana. PLoS One 9:e91776
Neerja Pant DC, Jain Bhakuni RS (2000) Phytochemicals from genus Swertia and their biological activities. Indian J Chem 39B:565–586
Negi Jagmohan S, Singh Pramod, Rawat B (2011) Chemical constituents and biological importance of Swertia: a review. Curr Res Chem 3(1):1–15
Padhan JK, Kumar V, Sood H, Singh TR, Chauhan RS (2015) Contents of therapeutic metabolites in Swertia chirayita correlate with the expression profiles of multiple genes in corresponding biosynthesis pathways. Phytochemistry 116:38–47
Pal D, Sur S, Mandal S, Das A, Roy A, Das S, Panda CK (2012) Prevention of liver carcinogenesis by amarogentin through modulation of G1/S cell cycle check point and induction of apoptosis. Carcinogenesis 33:2424–2431
Pan SY, Litscher G, Gao SH, Zhou SF, Yu ZL, Chen HQ, Zhang SF, Tang MK, Sun JN, Ko KM (2014) Historical perspective of traditional indigenous medical practices: the current renaissance and conservation of herbal resources. Evid Based Complement Alter Med eCAM 2014:525340
Paquette SM, Jensen K, Bak S (2009) A web-based resource for the Arabidopsis P450, cytochromes b5, NADPH-cytochrome P450 reductases, and family 1 glycosyltransferases (http://www.P450.kvl.dk). Phytochemistry 70:1940–1947
Peres V, Nagem TJ, de Oliveira FF (2000) Tetraoxygenated naturally occurring xanthones. Phytochemistry 55:683–710
Phoboo S, Pinto Mda S, Barbosa AC, Sarkar D, Bhowmik PC, Jha PK, Shetty K (2013) Phenolic-linked biochemical rationale for the anti-diabetic properties of Swertia chirayita (Roxb. ex Flem.) Karst. Phytother Res PTR 27:227–235
Priest DM, Jackson RG, Ashford DA, Abrams SR, Bowles DJ (2005) The use of abscisic acid analogues to analyse the substrate selectivity of UGT71B6, a UDP-glycosyltransferase of Arabidopsis thaliana. FEBS Lett 579:4454–4458
Rai A, Saito K (2016) Omics data input for metabolic modeling. Curr Opin Biotechnol 37:127–134
Rai A, Yamazaki M, Takahashi H, Nakamura M, Kojoma M, Suzuki H, Saito K (2016) RNA-seq transcriptome analysis of Panax japonicus, and its comparison with other Panax species to identify potential genes involved in the saponins biosynthesis. Front Plant Sci 7
Saito K (2013) Phytochemical genomics—a new trend. Curr Opin Plant Biol 16:373–380
Saravanan S, Islam VI, Babu NP, Pandikumar P, Thirugnanasambantham K, Chellappandian M, Raj CS, Paulraj MG, Ignacimuthu S (2014) Swertiamarin attenuates inflammation mediators via modulating NF-kappaB/I kappaB and JAK2/STAT3 transcription factors in adjuvant induced arthritis. Eur J Pharm Sci Off J Eur Fed Pharm Sci 56:70–86
Senthilvel S, Jayashree B, Mahalakshmi V, Kumar PS, Nakka S, Nepolean T, Hash C (2008) Development and map** of simple sequence repeat markers for pearl millet from data mining of expressed sequence tags. BMC Plant Biol 8:119
Steijger T, Abril JF, Engstrom PG, Kokocinski F, Hubbard TJ, Guigo R, Harrow J, Bertone P, Consortium R (2013) Assessment of transcript reconstruction methods for RNA-seq. Nat Methods 10:1177–1184
Takei H, Nakauchi K, Yoshizaki F (2001) Analysis of swertiamarin in Swertia herb and preparations containing this crude drug by capillary electrophoresis. Anal Sci Int J Jpn Soc Anal Chem 17:885–888
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Thiel T, Michalek W, Varshney RK, Graner A (2003) Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum vulgare L.). TAG Theor Appl Genet 106:411–422
Tomimori T, Komatsu M (1969) Studies on the constituents of Swertia japonica. V. On the xanthone constituents of the plants of Swertia ssp. Yakugaku zasshi J Pharm Soc Jpn 89:410–417
Vaidya H, Goyal RK, Cheema SK (2013) Anti-diabetic activity of swertiamarin is due to an active metabolite, gentianine, that upregulates PPAR-gamma gene expression in 3T3-L1 cells. Phytother Res PTR 27:624–627
Varshney RK, Thiel T, Stein N, Langridge P, Graner A (2002) In silico analysis on frequency and distribution of microsatellites in ESTs of some cereal species. Cell Mol Biol Lett 7:537–546
Varshney RK, Graner A, Sorrells ME (2005) Genic microsatellite markers in plants: features and applications. Trends Biotechnol 23:48–55
Veeresham C (2012) Natural products derived from plants as a source of drugs. J Adv Pharm Technol Res 3:200–201
Visser EA, Wegrzyn JL, Steenkmap ET, Myburg AA, Naidoo S (2015) Combined de novo and genome guided assembly and annotation of the Pinus patula juvenile shoot transcriptome. BMC Genom 16:1057
Wang J, Zhao C, Liu C, **a G, **ang F (2011) Introgression of Swertia mussotii gene into Bupleurum scorzonerifolium via somatic hybridization. BMC Plant Biol 11:71
Weeks AM, Chang MC (2011) Constructing de novo biosynthetic pathways for chemical synthesis inside living cells. Biochemistry 50:5404–5418
**e Y, Wu G, Tang J, Luo R, Patterson J, Liu S, Huang W, He G, Gu S, Li S, Zhou X, Lam TW, Li Y, Xu X, Wong GK, Wang J (2014) SOAPdenovo-Trans: de novo transcriptome assembly with short RNA-Seq reads. Bioinformatics 30:1660–1666
Yamahara J, Konoshima T, Sawada T, Fujimura H (1978) Biologically active principles of crude drugs: pharmacological actions of Swertia japonica extracts, swertiamarin and gentianine (author’s transl). Yakugaku zasshi J Pharm Soc Jpn 98:1446–1451
Yamahara J, Kobayashi M, Matsuda H, Aoki S (1991) Anticholinergic action of Swertia japonica and an active constituent. J Ethnopharmacol 33:31–35
Yamazaki Y, Kitajima M, Arita M, Takayama H, Sudo H, Yamazaki M, Aimi N, Saito K (2004) Biosynthesis of camptothecin. In silico and in vivo tracer study from [1-13C]glucose. Plant Physiol 134:161–170
Yonekura-Sakakibara K, Hanada K (2011) An evolutionary view of functional diversity in family 1 glycosyltransferases. Plant J Cell Mol Biol 66:182–193
Zhao Y, Chow TF, Puckrin RS, Alfred SE, Korir AK, Larive CK, Cutler SR (2007) Chemical genetic interrogation of natural variation uncovers a molecule that is glycoactivated. Nat Chem Biol 3:716–721
Zheng HH, Luo CT, Chen H, Lin JN, Ye CL, Mao SS, Li YL (2014) Xanthones from Swertia mussotii as multitarget-directed antidiabetic agents. ChemMedChem 9:1374–1377
Acknowledgments
This study, in part, was supported by the “Health and Labour Sciences Research Grant” on the enhancement of “Comprehensive Medicinal Plant Database” by the Grants-in-Aid for the Scientific Research of the Japan Society for the Promotion of Science (JSPS), and by the “Strategic Priority Research Promotion Program” of the Chiba University. HT was partially supported by MEXT KAKENHI (no. 221S0002). The super-computing resources were provided by the National Institute of Genetics (NIG), and Research Organization of Information and Systems, Japan. The computing resources were provided by the Medical Mycology Research Center, Chiba University, Japan. We also thank Mr. Tsutomu Hosouchi, and Ms. Sayaka Shinpo from the Kazusa DNA Research Institute for technical support in Illumina sequencing. We would also like to thank Dr. Ashfaq Mahmood, Megha from Chiba University, Dr. Gourvendu Saxena from the National University of Singapore, and two anonymous reviewers for their helpful and critical comments to improve this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Communicated by P. P. Kumar.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1 Summary for the InterProScan results of the S. japonica transcriptome assembly (EPS 1687 kb)
299_2016_2021_MOESM2_ESM.eps
Fig. S2 Distribution of the FPKM values of unigenes across the aerial tissues and the roots of S. japonica. The number of unigenes with the transcript expression levels in the FPKM values within different ranges for the aerial tissues and the roots of S. japonica is shown here (EPS 844 kb)
299_2016_2021_MOESM3_ESM.eps
Fig. S3 Distribution of unigenes, differentially expressed in the aerial tissues with respect to the roots in S. japonica, to the KEGG pathways database. A number of differentially expressed unigenes assigned to KEGG pathways are shown here. Red and green colour bar represents up- or down-regulated unigenes in the aerial tissues with respect to the roots of S. japonica (EPS 1352 kb)
299_2016_2021_MOESM4_ESM.eps
Fig. S4 Proposed pathways for the biosynthesis of mangiferin, and expression of unigenes assigned to the different enzyme coding genes in the S. japonica. Biosynthetic pathways for mangiferin, and candidate unigenes of S. japonica annotated as the enzyme coding genes at different biochemical reactions, and their expression levels are shown here. Red or green colour represents high or low expression levels, respectively, for a specific unigenes across the aerial tissues and the roots of S. japonica, while asterisks identify the false discovery rate, (FDR) <0.05, with the DESeq2 package. Abbreviation: DAHPS (3-deoxy-d-arabinoheptulosonate-7-phosphate synthase), DHQS (3-dehydroquinate synthase), DHQD (3-dehydroquinate dehydratase), SDH shikimate dehydrogenase), SAK (shikimate kinase), EPSPS (5-enolpyruvylshikimate-3-phosphate synthase), CS (chorismate synthase), CM (chorismate mutase), ADT (prephenate dehydratase), PAT (aspartate–prephenate aminotransferase), PHAT (phenylalanine-histidine aminotransferase), ADH (arogenate dehydrogenase), PAL (phenylalanine ammonia lyase), TAL (tyrosine ammonia lyase), C4H (trans-cinnamate 4-hydroxylase), and C3H (p-coumarate 3-hydroxylase) (EPS 2056 kb)
299_2016_2021_MOESM5_ESM.docx
Table S1 Output summary of the Trimmomatic program-based pre-processing of raw reads of the aerial tissues and the roots of S. japonica (DOCX 12 kb)
299_2016_2021_MOESM6_ESM.xlsx
Table S2 Blastx search summary for the unigenes of the S. japonica transcriptome assembly with sequence similarity against NCBI-non-redundant (nr) database (XLSX 5674 kb)
299_2016_2021_MOESM7_ESM.xlsx
Table S3 InterProScan result summary for the unigenes of the S. japonica transcriptome assembly with a hit against InterPro protein signature databases (XLSX 1255 kb)
299_2016_2021_MOESM8_ESM.xlsx
Table S4 Summary of S. japonica transcriptome assembly annotation with assigned gene ontology terms, EC numbers, and KEGG pathways annotation based on Blastx and InterProScan search results using Blast2GO program (XLSX 9805 kb)
299_2016_2021_MOESM9_ESM.xlsx
Table S5 List of KEGG pathways identified, and the number of unigenes assigned from the S. japonica transcriptome assembly (XLSX 11 kb)
299_2016_2021_MOESM10_ESM.xlsx
Table S6 Identified simple sequence repeats (SSRs) motifs and their frequencies in the S. japonica transcriptome assembly (XLSX 23 kb)
299_2016_2021_MOESM11_ESM.xlsx
Table S7 Transcript expression values (FPKM values) for all unigenes for the aerial tissues and the roots of S. japonica (XLSX 2133 kb)
299_2016_2021_MOESM12_ESM.xlsx
Table S8 Annotation summary and transcript expression values for the unigenes annotated as UDP-glycosyltransferase and up-regulated in the aerial tissues with respect to the roots of S. japonica (XLSX 15 kb)
299_2016_2021_MOESM13_ESM.xlsx
Table S9 Annotation summary and transcript expression values for the unigenes annotated as UDP-glycosyltransferase and down-regulated in the aerial tissues with respect to the roots of S. japonica (XLSX 19 kb)
299_2016_2021_MOESM14_ESM.docx
Data set S1 Nucleotide sequences for the unigenes of S. japonica, annotated as enzyme coding genes associated with the methylerythritol phosphate (MEP) biosynthesis pathways (DOCX 19 kb)
299_2016_2021_MOESM15_ESM.docx
Data set S2 Nucleotide sequences for the unigenes of S. japonica, annotated as enzyme coding genes associated with the mevalonate (MVA) biosynthesis pathways (DOCX 19 kb)
299_2016_2021_MOESM16_ESM.docx
Data set S3 Nucleotide sequences for the unigenes of S. japonica, annotated as enzyme coding genes associated with the secoiridoid biosynthesis pathways (DOCX 26 kb)
299_2016_2021_MOESM17_ESM.docx
Data set S4 Nucleotide sequences for the unigenes of S. japonica, annotated as enzyme coding genes associated with the mangiferin biosynthetic pathways (DOCX 31 kb)
299_2016_2021_MOESM18_ESM.docx
Data set S5 Nucleotide sequences for the unigenes up-regulated in the aerial tissues with respect to the roots of the S. japonica, and annotated as UDP-glycosyltransferase coding genes (DOCX 27 kb)
299_2016_2021_MOESM19_ESM.docx
Data set S6 Nucleotide sequences for the unigenes down-regulated in the aerial tissues with respect to the roots of the S. japonica, and annotated as UDP-glycosyltransferase coding genes (DOCX 38 kb)
Rights and permissions
About this article
Cite this article
Rai, A., Nakamura, M., Takahashi, H. et al. High-throughput sequencing and de novo transcriptome assembly of Swertia japonica to identify genes involved in the biosynthesis of therapeutic metabolites. Plant Cell Rep 35, 2091–2111 (2016). https://doi.org/10.1007/s00299-016-2021-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-016-2021-z