Abstract
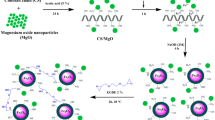
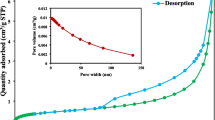
In this work, a new quadruple nanocomposite of chitosan/polyvinyl alcohol/magnesium oxide/Fe3O4 (Chit-PVA/MgO/Fe3O4) was prepared for the adsorption of remazol brilliant blue R (RBBR) dye in batch process. Several advanced analytical techniques including BET, XRD, FTIR, SEM–EDX, potentiometric titrations, and pHpzc were performed for characterizing of the synthesized nanocomposite (Chit-PVA/MgO/Fe3O4). A multi-step experimental design was derived from response surface methodology (RSM) for optimizing the factors affecting RBBR dye adsorption, namely A: dose (0.02–0.1 g), B: pH (4–10), C: temperature (30–60 °C), and time D: (10–60 min). The obtained kinetic data and adsorption isotherm for the studied Chit-PVA/MgO/Fe3O4-RBBR system followed pseudo-second-order and Langmuir isotherm model, respectively. The maximum adsorption capacity of Chit-PVA/MgO/Fe3O4 toward RBBR was found to be 163.7 mg/g. The adsorption mechanism of RBBR dye by Chit-PVA/MgO/Fe3O4 possibly comprises an electrostatic interaction, hydrogen bonding, n-π interactions, and Yoshida H-bonding. This work reveals that Chit-PVA/MgO/Fe3O4 nanocomposite is an efficient and recoverable biohybrid adsorbent with a great ability to remove organic dye from an aqueous environment.









Similar content being viewed by others
References
Rafaie HA, Yusop NFM, Azmi NF, Abdullah NS, Ramli NIT (2021) Photocatalytic degradation of methylene blue dye solution using different amount of ZnO as a photocatalyst. Sci Lett 15(1):1–12
Malek NNA, Mohammed IA, Reghioua A, Yousif E, Jawad AH (2020) Removal of reactive red 4 dye using chitosan-epichlorohydrin/ TiO2 nanocomposite: application of response surface methodology. Sci Lett 14(1):96–108
Malek NNA, Yousif E, Jawad HJ (2020) Optimization of adsorption parameters for reactive red 4 (RR4) removal by cross-linked chitosan-epichlorohydrin using box-behnken design. Sci Lett 14(1):83–95
Mohammed IA, Jawad AH, Abdulhameed AS, Mastuli MS (2020) Physicochemical modification of chitosan with fly ash and tripolyphosphate for removal of reactive red 120 dye: statistical optimization and mechanism study. Int J Biol Macromol 161:503–513
Bonetto L, Crespo J, Guégan R, Esteves V, Giovanela M (2021) Removal of methylene blue from aqueous solutions using a solid residue of the apple juice industry: full factorial design, equilibrium, thermodynamics and kinetics aspects. J Mol Struct 1224:129296–129310
Medrano-Rodríguez F, Picos-Benítez A, Brillas E, Bandala ER, Pérez T, Peralta-Hernández JM (2020) Electrochemical advanced oxidation discoloration and removal of three brown diazo dyes used in the tannery industry. J Electroanal Chem 873:114360–114368
Tahira I, Aslam Z, Abbas A, Monim-ul-Mehboob M, Ali S, Asghar A (2019) Adsorptive removal of acidic dye onto grafted chitosan: a plausible grafting and adsorption mechanism. Int J Biol Macromol 136:1209–1218
Mcyotto F, Wei Q, Macharia DK, Huang M, Shen C, Chow CW (2021) Effect of dye structure on color removal efficiency by coagulation. Chem Eng J 405:126674–126687
Pereira MFR, Soares SF, Órfão JJ, Figueiredo JL (2003) Adsorption of dyes on activated carbons: influence of surface chemical groups. Carbon 41:811–821
Gupta V (2009) Application of low-cost adsorbents for dye removal–a review. J Environ Manage 90:2313–2342
Kahdestani SA, Shahriari MH, Abdouss M (2021) Synthesis and characterization of chitosan nanoparticles containing teicoplanin using sol–gel. Polym Bull 78:1133–1148
Liu Xq, Zhao Xx, Liu Y, Zhang T (2021) Review on preparation and adsorption properties of chitosan and chitosan composites. Polym Bull 79:2633–2665
Mahmoodi NM, Oveisi M, Taghizadeh A, Taghizadeh M (2020) Synthesis of pearl necklace-like ZIF-8@ chitosan/PVA nanofiber with synergistic effect for recycling aqueous dye removal. Carbohydr Polym 227:115364–115376
Zhang L, Zeng Y, Cheng Z (2016) Removal of heavy metal ions using chitosan and modified chitosan: a review. J Mol Liq 214:175–191
Keshvardoostchokami M, Majidi M, Zamani A, Liu B (2021) A review on the use of chitosan and chitosan derivatives as the bio-adsorbents for the water treatment: removal of nitrogen-containing pollutants. Carbohydr Polym 273:118625–118637
Jawad AH, Abdulhameed AS, Kashi E, Yaseen ZM, ALOthman ZA, Khan MR, (2022) Cross-linked chitosan-glyoxal/kaolin clay composite: parametric optimization for color removal and COD reduction of remazol brilliant blue R dye. J. Polym. Environ. 30:164–178
Malek NNA, Jawad AH, Abdulhameed AS, Ismail K, Hameed B (2020) New magnetic schiff’s base-chitosan-glyoxal/fly ash/Fe3O4 biocomposite for the removal of anionic azo dye: an optimized process. Int J Biol Macromol 146:530–539
Nga NK, Chau NTT, Viet PH (2020) Preparation and characterization of a chitosan/MgO composite for the effective removal of reactive blue 19 dye from aqueous solution. J Sci-Adv Mater Dev 5:65–72
Dalvand A, Nabizadeh R, Ganjali MR, Khoobi M, Nazmara S, Mahvi AH (2016) Modeling of reactive blue 19 azo dye removal from colored textile wastewater using L-arginine-functionalized Fe3O4 nanoparticles: optimization, reusability, kinetic and equilibrium studies. J Magn Magn Mater 404:179–189
Vieira RS, Beppu MM (2006) Interaction of natural and crosslinked chitosan membranes with Hg (II) ions. Colloids Surf A Physicochem Eng Asp 279:196–207
Nawi M, Sabar S, Jawad AH, Ngah WW (2010) Adsorption of reactive red 4 by immobilized chitosan on glass plates: towards the design of immobilized TiO2–chitosan synergistic photocatalyst-adsorption bilayer system. Biochem Eng J 49:317–325
Sing KS (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (recommendations 1984). Pure Appl Chem 57:603–619
Khalil KD, Bashal AH, Khalafalla M, Zaki AA (2020) Synthesis, structural, dielectric and optical properties of chitosan-mgo nanocomposite. J Taibah Univ Sci 14:975–983
Reghioua A, Barkat D, Jawad AH, Abdulhameed AS, Al-Kahtani AA, ALOthman ZA, (2021) Parametric optimization by box–behnken design for synthesis of magnetic chitosan-benzil/ZnO/Fe3O4 nanocomposite and textile dye removal. J Environ Chem Eng 9:105166–105179
Mei Y, Runjun S, Yan F, Honghong W, Hao D, Chengkun L (2019) Preparation, characterization and kinetics study of chitosan/pva electrospun nanofiber membranes for the adsorption of dye from water. J Polym Eng 39:459–471
Lagergren S (1898) About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Handingarl 24:1–39
Ho YS, McKay G (1998) Sorption of dye from aqueous solution by peat. Chem Eng J 70:115–124
Vakili M, Rafatullah M, Salamatinia B, Ibrahim MH, Abdullah AZ (2015) Elimination of reactive blue 4 from aqueous solutions using 3-aminopropyl triethoxysilane modified chitosan beads. Carbohydr Polym 132:89–96
Freundlich H (1906) Over the adsorption in solution. J Phys Chem 57:384–471
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Temkin MI (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochim URSS 12:327–356
Malek NNA, Jawad AH, Ismail K, Razuan R, ALOthman ZA, (2021) Fly ash modified magnetic chitosan-polyvinyl alcohol blend for reactive orange 16 dye removal: adsorption parametric optimization. Int J Biol Macromol 189:464–476
Jawad AH, Abdulhameed AS, Malek NNA, ALOthman ZA (2020) Statistical optimization and modeling for color removal and cod reduction of reactive blue 19 dye by mesoporous chitosan-epichlorohydrin/kaolin clay composite. Int J Biol Macromol 164:4218–4230
Janaki V, Oh BT, Shanthi K, Lee KJ, Ramasamy A, Kamala-Kannan S (2012) Polyaniline/chitosan composite: an eco-friendly polymer for enhanced removal of dyes from aqueous solution. Synth Met 162:974–980
Li X, Sun C, Cao W, Hu C, Zhao Y (2019) Comparative adsorption of remazol brilliant blue R and copper in aqueous solutions by carbon nanotubes with different levels of carboxyl group and specific surface area. Mater Res Express 6:1050e2
Phan DN, Rebia RA, Saito Y, Kharaghani D, Khatri M, Tanaka T, Lee H, Kim IS (2020) Zinc oxide nanoparticles attached to polyacrylonitrile nanofibers with hinokitiol as gluing agent for synergistic antibacterial activities and effective dye removal. J Ind Eng Chem 85:258–268
Özcan A, Ömeroğlu Ç, Erdoğan Y, Özcan AS (2007) Modification of bentonite with a cationic surfactant: an adsorption study of textile dye reactive blue 19. J Hazard Mater 140:173–179
Nair V, Panigrahy A, Vinu R (2014) Development of novel chitosan–lignin composites for adsorption of dyes and metal ions from wastewater. Chem Eng J 254:491–502
Abbasi M (2017) Synthesis and characterization of magnetic nanocomposite of chitosan/SiO2/carbon nanotubes and its application for dyes removal. J Clean Prod 145:105–113
Saputra OA, Rachma AH, Handayani DS (2017) Adsorption of remazol brilliant blue R using amino-functionalized organosilane in aqueous solution. Indones J Chem 17:343–350
Mate CJ, Mishra S (2020) Synthesis of borax cross-linked jhingan gum hydrogel for remediation of remazol brilliant blue R (RBBR) dye from water: adsorption isotherm, kinetic, thermodynamic and biodegradation studies. Int J Biol Macromol 151:677–690
Acknowledgements
The authors would like to acknowledge Faculty of Applied Sciences, Universiti Teknologi MARA, Shah Alam, for facilitating this research work. The authors would like to thank the Research Management Center (RMC), Universiti Teknologi MARA (UiTM) for supporting this research by Strategic Research Partnership (SRP) grant (100-RMC 5/3/SRP (052/2021)).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jawad, A.H., Hameed, B.H. & Abdulhameed, A.S. Synthesis of biohybrid magnetic chitosan-polyvinyl alcohol/MgO nanocomposite blend for remazol brilliant blue R dye adsorption: solo and collective parametric optimization. Polym. Bull. 80, 4927–4947 (2023). https://doi.org/10.1007/s00289-022-04294-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04294-z