Abstract
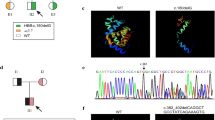
Most β-thalassemias are caused by mutations involving one or a limited number of nucleotides within the gene or its adjacent regions. They can be substitutions or deletions; in these cases, the loss ranges from a single nucleotide to even the entire HBB gene, so we wonder if the phenotype is due to the size of the deletion or the location of the mutation. To clarify this, we present two new deletions in the β-globin gene that cause β0-thalassemia. The hematological parameters were determined with an automated cell counter; the Hb A2 and Hb F levels were measured by performance liquid chromatography. Hemoglobins were analyzed by capillary zone electrophoresis (Sebia Capillarys Flex system) and ion-exchange HPLC (BioRad Variant II β-thalassemia Short Program). Molecular characterization was performed by automatic Sanger sequencing. The screening of common α-thalassemia point mutations and deletions in the world (21 in total) were carried out using multiplex PCR followed by reverse-hybridization with a commercial Alpha-Globin StripAssay kit. We have characterized two new mutations—(1) 1-bp deletion [CD61/62(–G)] [HBB:c.186_187delG], (2) 105-bp deletion [IVS-2-nt767-CD111] [HBB:c.316-84_333del]—and we have described, for first time in Spain, the 25-bp deletion [β nts 252 - 276 deleted] [HBB:c.93-22_95del] mutation. These mutations were classified as pathogenic by UniProt Variants confirmed according to the American College of Medical Genetics and Genomics guidelines. These mutations present a phenotype compatible with β0-thalassemia, supported by hematological parameters that correlate the degree of reduction in the synthesis of the β-globin chain. Identification of this type of mutation is important for genetic counselling of partners where both are carriers, so that they are aware of the genetic risk of having affected children, allowing them to take an informed decision about their reproductive choices.




Similar content being viewed by others
References
Steinberg MH, Forget BG, Higgs DR, Weatherall DJ (2009) Disorders of hemoglobin: genetics, pathophysiology, and clinical management. Cambridge University Press, New York
Taher Ali T, Musallam Khaled M, Cappellini M (2021) Domenica. β-Thalassemias. N Engl J Med 384:727–743. https://doi.org/10.1056/NEJMra2021838
Origa R (2000) Beta-Thalassemia. [Updated 2021 Feb 4]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2022. Bookshelf URL: https://www.ncbi.nlm.nih.gov/books/. Accessed March 2021
Thein SL (2013) The molecular basis of β-thalassemia. Cold Spring Harb Perspect Med 3(5):a011700. https://doi.org/10.1101/cshperspect.a011700
Galanello R, Origa R (2010) Beta-thalassemia. Orphanet J Rare Dis. 21(5):11. https://doi.org/10.1186/1750-1172-5-11
Giardine B, Borg J, Viennas E, Pavlidis C, Moradkhani K, Joly P et al (2014) Updates of the HbVar database of human hemoglobin variants and thalassemia mutations. Nucleic Acids Res. 42(D1):D1063–D1069. https://doi.org/10.1093/nar/gkt911]
Kountouris P, Lederer CW, Fanis P, Feleki X, Old J, Kleanthous M (2014) IthaGenes: an interactive database for haemoglobin variations and epidemiology. PLoS One 9(7):e103020. https://doi.org/10.1371/journal.pone.0103020
Thein SL (2018) Molecular basis of β thalassemia and potential therapeutic targets. Blood Cells Mol Dis 70:54–65. https://doi.org/10.1016/j.bcmd.2017.06.001
Losick R, Watson J, Tania AB, Bell S, Gann A, Levine MW (2004) Molecular biology of the gene. Pearson/Benjamin Cummings, San Francisco (978-0-8053-9592-1)
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–24. https://doi.org/10.1038/gim.2015.30
Orkin SH, Sexton JP, Goff SC, Kazazian HH Jr (1983) Inactivation of an acceptor RNA splice site by a short deletion in beta-thalassemia. J Biol Chem. 258(12):7249–51
Baker KE, Parker R (2004) Nonsense-mediated mRNA decay: terminating erroneous gene expressionr. Curr Opin Cell Biol 16(3):293–299
Weatherall DJ, Clegg JB (2001) The thalassemia syndromes. Blackwell Sciences, Oxford
Cao A, Galanello R (2010) Beta-thalassemia. Genet Med 12(2):61–76. https://doi.org/10.1097/GIM.0b013e3181cd68ed
Cao A, Galanello R, Rosatelli MC (1994) Genotype-phenotype correlations in beta-thalassemias. Blood Rev 8(1):1–12. https://doi.org/10.1016/0268-960x(94)90002-7
Orkin SH, Cheng TC, Antonarakis SE, Kazazian HH Jr (1985) Thalassemia due to a mutation in the cleavage-polyadenylation signal of the human beta-globin gene. EMBO J. 4(2):453–6
Author information
Authors and Affiliations
Contributions
PR, WT, and FG were involved in the design of the study. PR has developed the research. JN developed the methodology. VR, MAM, MJM, MS, and CF have sent us the cases. PR wrote the first draft of the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ropero, P., González Fernández, F.A., Nieto, J.M. et al. Does size matter? Two new deletions in the HBB gene cause β0-thalassemia. Ann Hematol 101, 1465–1471 (2022). https://doi.org/10.1007/s00277-022-04837-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-022-04837-4