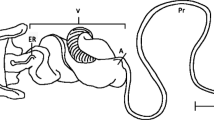
Abstract
Male genitalia are rapidly evolving structures, often driven by sexual selection to increase fertilisation success. Although sexual selection on females can be strong in systems where males provide offspring care or feed their mates, sometimes resulting in the evolution of female ornamentation, there are no actual estimates of direct sexual selection on female genitalia. In a New Zealand ground weta, Hemiandrus pallitarsis (Orthoptera: Ensifera, Anostostomatidae), females possess a genitalic device (the accessory organ) that is necessary for successful copulation and the acquisition of glandular food-gifts from males. These nutritious gifts are known to result in sexual competition among females in other ensiferan species. In ground weta, the gifts are probably important in avoiding starvation during a months-long period when caring for (their lifetime production of) eggs and offspring. Here, we test the hypothesis that the accessory organ is a sexually selected device in H. pallitarsis by measuring the female Bateman gradient, Jones index, and directional sexual selection on the accessory organ. Using newly developed and characterised microsatellite loci, we analyse offspring and/or stored sperm to estimate female mating frequency for the first time in ground weta. As predicted, we found both a positive Bateman gradient and Jones index for females, and evidence of directional sexual selection on accessory organ length. Although organ length does not correlate well with female fecundity, it may increase mating success by indicating her condition and thus quality of her offspring care.
Significance statement
Female genitalia have largely been studied in the context of exerting selection on males, while the potential for selection on females via mate acquisition or manipulation of males is virtually unstudied. Such selection may be relevant in systems where males make investments in reproduction (offspring-care; mate-feeding), and females are subject to strong sexual selection, occasionally possessing sexual ornaments. Using microsatellite analysis of offspring and stored sperm to estimate mating frequency, we provide the first evidence of directional sexual selection on a female genital device (accessory organ) found in species of maternal care-providing ground weta. In our focal species, Hemiandrus pallitarsis, we found that females with more mates produced more offspring and females with longer accessory organs obtained more mates. These findings suggest that the female genital device is a secondary sexual trait, although may have additional mechanical functions.



Similar content being viewed by others
Data availability
All data generated or analysed during this study are available in the Dryad repository: https://doi.org/10.5061/dryad.s1rn8pkbh.
References
Andersson M (1994) Sexual selection. Princeton University Press, New Jersey
Anthes N, Häderer IK, Michiels NK, Janicke T (2017) Measuring and interpreting sexual selection metrics: evaluation and guidelines. MEE 8:918–931. https://doi.org/10.1111/2041-210X.12707
Arnold SJ (1994) Bateman’s principles and the measurement of sexual selection in plants and animals. Am Nat 144:126–149. https://doi.org/10.1086/285656
Arnqvist G, Rowe L (2005) Sexual conflict. Princeton University Press, New Jersey
Aronsen T, Berglund A, Mobley KB, Ratikainen II, Rosenqvist G (2013) Sex ration and density affect sexual selection in a sex-role reversed fish. Evolution 67:3243–3257. https://doi.org/10.1111/evo.12201
Banks HJ, Williams DJ (1972) Use of the surfactant, Decon 90, in the preparation of coccids and other insects for microscopy. J Aust Entomol Soc 11:347–448. https://doi.org/10.1111/j.1440-6055.1972.tb01639.x
Barry KL (2015) Sexual deception in a cannibalistic mating system? Testing the Femme Fatale hypothesis. Proc R Soc B 282:20141428. https://doi.org/10.1098/rspb.2014.1428
Bateman AJ (1948) Intra-sexual selection in Drosophila. Heredity 2:349–368. https://doi.org/10.1038/hdy.1948.21
Berglund A, Rosenqvist G, Bernet P (1997) Ornamentation predicts reproductive success in female pipefish. Behav Ecol Sociobiol 40:145–150 (https://www.jstor.org/stable/4601312)
Bohme W (1983) The Tucano Indians of Columbia and the iguanid lizard Plica plica: ethnological herpetological and ethological implications. Biotropica 15:148–150. https://doi.org/10.2307/2387961
Bonduriansky R (2001) The evolution of male mate choice in insects: a synthesis of ideas and evidence. Biol Rev 76:305–339. https://doi.org/10.1017/S1464793101005693
Bretman A, Tregenza T (2005) Measuring polyandry in wild population: a case study using promiscuous crickets. Mol Ecol 14:2169–2179. https://doi.org/10.1111/j.1365-294X.2005.02556.x
Briceño R, Wegrzynek DD, Chinea-Cano E, Eberhard WG, dos Santos RT (2010) Movements and morphology under sexual selection: tsetse fly genitalia. Ethol Ecol Evol 22:385–391. https://doi.org/10.1080/03949370.2010.505581
Brooks R, Caithness N (1995) Female choice in a feral guppy population: are there multiple cues? Anim Behav 50:301–307. https://doi.org/10.1006/anbe.1995.0246
Browne JH, Gwynne DT (2022) Paternity sharing in insects with female competition for nuptial gifts. Ecol Evol 12:E9463. https://doi.org/10.1002/ece3.9463
Buzkova P (2016) Interaction testing: residuals-based permutations and parametric bootstrap in continuous count, and binary data. Epidemiol Methods 5:119–128. https://doi.org/10.1515/em-2015-0010
Buzkova P, Lumley T, Rice K (2011) Permutation and parametric bootstrap tests for gene-gene and gene-environment interactions. Ann Hum Genet 75:36–45. https://doi.org/10.1111/j.1469-1809.2010.00572.x
Chappell EM, Trewick SA, Morgan-Richards M (2012) Shape and sound reveal genetic cohesion not speciation in the New Zealand orthopteran, Hemiandrus pallitarsis, despite high mitochondrial DNA divergence. Biol J Linn Soc 105:169–186. https://doi.org/10.1111/j.1095-8312.2011.01777.x
Chenoweth SF, Doughty P, Kokko H (2006) Can non-directional male mating preferences facilitate honest female ornamentation? Ecol Lett 9:179–184. https://doi.org/10.1111/j.1461-0248.2005.00867.x
Clutton-Brock T (2007) Sexual selection in males and females. Science 318:1882–1885 (https://www.science.org/doi/10.1126/science.1133311)
Collet JM, Dean RF, Worley K, Richardson DS, Pizzari T (2014) The measure and significance of Bateman’s principles. Proc R Soc B 281:20132973. https://doi.org/10.1098/rspb.2013.2973
Cumming JM (1994) Sexual selection and the evolution of dance fly mating systems (Diptera: Empididae: Empidinae). Can Entomol 126:907–920. https://doi.org/10.4039/Ent126907-3
Dakin EE, Avise JC (2004) Microsatellite null alleles in parentage analysis. Heredity 93:504–509. https://doi.org/10.1038/sj.hdy.6800545
Darwin C (1871) The decent of man and selection in relation to sex. John Murray, London
Downes JA (1970) The feeding and mating behaviour of the specialized Empidinae (Diptera); observations on four species of Rhamphomyia in the high arctic and general discussion. Can Entomol 102:769–791. https://doi.org/10.4039/Ent102769-7
Eberhard WG (1985) Sexual selection and animal genitalia. Harvard University Press, Massachusetts
Eberhard WG (1990) Animal genitalia and female choice. Am Sci 78:134–141
Eberhard WG, Huber BA, Rodriguez RL, Briceño RD, Salas I, Rodriguez V (1998) One size fits all? Relationships between the size and degree of variation in genitalia and other body parts in twenty species of insects and spiders. Evolution 52:415–431. https://doi.org/10.2307/2411078
Fitzpatrick S, Berglund A, Rosenqvist G (1995) Ornaments or offspring: costs to reproductive success restrict sexual selection processes. Biol J Linn Soc 55:251–260. https://doi.org/10.1111/j.1095-8312.1995.tb01063.x
Flanagan SP, Johnson JB, Rose E, Jones G (2014) Sexual selection on female ornaments in the sex-role-reversed pipefish (Syngnathus scovelli). J Evol Biol 27:2457–2467. https://doi.org/10.1111/jeb.12487
Foster JC, Nan B, Shen L, Kaciroti N, Taylor JMG (2016) Permutation testing for treatment-covariate interactions and subgroup identification. Stat in Biosci 8:77–98. https://doi.org/10.1007/s12561-015-9125-9
Fritzsche K, Arnqvist G (2013) Homage to Bateman: sex roles predict sex differences in sexual selection. Evolution 67:1926–1936. https://doi.org/10.1111/evo.12086
Fromonteil S, Marie-Orleach L, Winkler L, Janicke T (2023) Sexual selection in females and the evolution of polyandry. PLoS Biol 21:e3001916. https://doi.org/10.1371/journal.pbio.3001916
Gerlach NM, McGlothlin JW, Parker PG, Ketterson ED (2012) Interpreting Bateman gradients: multiple mating and selection in both sexes of a songbird species. Behav Ecol 23:1078–1088. https://doi.org/10.1093/beheco/ars077
Gwynne DT (1981) Sexual difference theory: Mormon crickets show role reversal in mate choice. Science 213:779–780 (https://www.science.org/doi/10.1126/science.213.4509.779)
Gwynne DT (1984) Sexual selection and sexual differences in Mormon crickets (Orthoptera: Tettigoniidae, Anabrus simplex). Evolution 38:1011–1022. https://doi.org/10.2307/2408435
Gwynne DT (1995) Phylogeny of the Ensifera (Orthoptera): a hypothesis supporting multiple origins of acoustical signalling, complex spermatophores and maternal care in crickets, katydids and weta. JOR 4:203–218. https://doi.org/10.2307/3503478
Gwynne DT (2001) Katydids and bush-crickets: reproductive behavior and evolution of the Tettigoniidae. Cornell University Press, New York
Gwynne DT (2002) A secondary copulatory structure in a female insect: a clasp for a nuptial meal? Naturwissenschaften 89:125–127. https://doi.org/10.1007/s00114-002-0298-y
Gwynne DT (2004) Reproductive behavior of ground weta (Orthoptera: Anostostomatidae): drumming behavior, nuptial feeding, post-copulatory guarding and maternal care. J Kans Entomol Soc 77:414–428 (https://www.jstor.org/stable/25086232)
Gwynne DT (2005) The secondary copulatory organ in female ground weta (Hemiandrus pallitarsis, Orthoptera: Anostostomatidae): a sexually selected device in females? Biol J Linn Soc 85:463–469. https://doi.org/10.1111/j.1095-8312.2005.00510.x
Gwynne DT (2016) Sexual selection: roles evolving. Curr Biol 26:913–936. https://doi.org/10.1016/j.cub.2016.08.063
Gwynne DT, Bailey WJ (1999) Female-female competition in katydids: sexual selection for increased sensitivity to a male signal? Evolution 53:546–551. https://doi.org/10.1111/j.1558-5646.1999.tb03789.x
Gwynne DT, Simmons LW (1990) Experimental reversal of courtship roles in an insect. Nature 346:172–174. https://doi.org/10.1038/346172a0
Hare RM, Simmons LW (2018) Sexual selection and its evolutionary consequences in female animals. Biol Rev 94:929–956. https://doi.org/10.1111/brv.12484
Hare RM, Simmons LW (2021) Sexual selection maintains a female-specific character in a species with dynamic sex roles. Behav Ecol 32:609–616. https://doi.org/10.1093/beheco/arab005
Hare RM, Simmons LW (2022) Bateman gradients reflect variation in sexual selection in a species with dynamic sex roles. J Evol Biol 35:1206–1217. https://doi.org/10.1111/jeb.14070
Henshaw JM, Kahn AT, Fritzsche K (2016) A rigorous comparison of sexual selection indexes via simulations of diverse mating systems. Proc Natl Acad Sci USA 113:E300–E308. https://doi.org/10.1073/pnas.1518067113
Herridge EJ, Murray RL, Gwynne DT, Bussière LF (2016) Diversity in mating and parental sex roles. In: Kliman RM (ed) Encyclopedia of evolutionary biology. Elsevier, Oxford, pp 453–458
Herridge EJ (2016) Polyandry rates and reproductive success in a nuptial gift giving dance fly Rhamphomyia longicauda, Dissertation. University of Stirling.
Hosken DJ, House CM (2011) Sexual Selection Current Biol 21:62–65. https://doi.org/10.1016/j.cub.2010.11.053
Johns PM (2001) Distribution and conservation status of ground weta, Hemiandrus species (Orthoptera: Anostostomatidae). Science for Conservation Report 180, Department of Conservation, Wellington, New Zealand
Jones AG (2005) GERUD 2.0: a computer program for the reconstruction of parental genotypes from half-sib progeny arrays with known or unknown parents. Mol Ecol Notes 5:708–711. https://doi.org/10.1111/j.1471-8286.2005.01029.x
Jones SG (2009) On the opportunity for sexual selection, the Bateman gradient and the maximum intensity of sexual selection. Evolution 63:1673–1684. https://doi.org/10.1111/j.1558-5646.2009.00664.x
Jones AG (2015) BATEMANATOR: a computer program to estimate and bootstrap mating system variables based on Bateman’s principles. Mol Ecol Resour 15:1396–1402. https://doi.org/10.1111/1755-0998.12397
Kelly CD, Gwynne DT (2023) Mating assortment and the strength of sexual selection in a polyandrous population of Cook Strait giant weta. Behav Ecol 34:506–513. https://doi.org/10.1093/beheco/arad017
Lande R, Arnold ST (1983) The measurement of selection on correlated characters. Evolution 37:1210–1226. https://doi.org/10.2307/2408842
Lefèvre BM, Catté D, Courtier-Orgogoza V, Lang M (2021) Male genital lobe morphology affects the chance to copulate in Drosophila pachea. BMC Ecol Evol 21:23. https://doi.org/10.1186/s12862-021-01759-z
Lloyd JE (1979) Mating behaviour and natural selection. Fla Entomol 62:17–34. https://doi.org/10.2307/3494039
Mayr E (1963) Animal species and evolution. Harvard University Press, Cambridge
Mobley KB, Jones AG (2012) Overcoming statistical bias to estimate genetic mating systems in open populations: a comparison of Bateman’s principles between the sexes in a sex-role-reversed pipefish. Evolution 67:646–660. https://doi.org/10.1111/j.1558-5646.2012.01819.x
Morrow EH, Arnqvist G (2003) Costly traumatic insemination and female counter-adaptation in bed bugs. Proc R Soc B 270:2377–2381. https://doi.org/10.1098/rspb.2003.2514
Murray RL, Gwynne DT, Bussière LF (2022) Mating and sexual selection in empidine dance flies. Insects 13:839. https://doi.org/10.3390/insects13090839
Nason SE, Kelly CD (2020) Benefits of multiple mating in a sexually dimorphic polygynandrous insect. Anim Behav 164:65–72. https://doi.org/10.1016/j.anbehav.2020.03.018
Parker GA, Simmons LW (1996) Parental investment and the control of sexual selection: predicting the direction of sexual competition. Proc R Soc 263:315–321 (https://www.jstor.org/stable/50614)
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software from exact tests and ecumenicism. J Hered 86:248–249. https://doi.org/10.1093/oxfordjournals.jhered.a111573
Rentz DCF (1993) Tettigoniidae of Australia Volume 2: The Austrosaginae, Zaprochilinae and Phasmodinae. CSIRO, Canberra
Robson LJ, Gwynne DT (2010) Measuring sexual selection on females in sex-role reversed Mormon crickets (Anabrus simplex, Orthoptera: Tettigoniidae). J Evol Ecol 23:1528–1537. https://doi.org/10.1111/j.1420-9101.2010.02021.x
Salmon JT (1950) A revision of the of the New Zealand weta Anostostomatidae (Orthoptera: Stenopelmatidae). Dominion Museum Records in Entomology, ISSN 0373–7233
Schlupp I (2021) Male choice, female competition, and female ornaments in sexual selection. Oxford University Press, New York
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Shuker DM, Kvarnemo C (2021) The definition of sexual selection. Behav Ecol 32:781–794. https://doi.org/10.1093/beheco/arab055
Simmons LW (1992) Quantification of role reversal in relative parental investment in a bush cricket. Nature 358:61–63. https://doi.org/10.1038/358061a0
Simmons LW (2001) Sperm competition and its evolutionary consequences in the insects. Princeton University Press, New Jersey
Simmons LW, Bailey WJ (1990) Resource influenced sex roles of zaprochiline tettigoniids (orthoptera: Tettigoniidae). Evolution 44:1853–1868. https://doi.org/10.1111/j.1558-5646.1990.tb05255.x
Sloan NS, Simmons LW (2019) The evolution of female genitalia. J Evol Biol 32:882–899. https://doi.org/10.1111/jeb.13503
Taylor Smith BL, Morgan-Richards M, Trewick SA (2013) New Zealand ground weta (Anostostomatidae: Hemiandrus): descriptions of two new species with notes on their biology. N Z J Zool 40:314–329. https://doi.org/10.1080/03014223.2013.804422
Taylor Smith BL (2015) Evolution of diversity: analysis of species and speciation in Hemiandrus ground weta. Dissertation, Massey University
Thornhill R (1983) Cryptic female choice and its implications in the scorpionfly Harpobittacus nigriceps. Am Nat 122:765–788 (https://www.jstor.org/stable/2460916)
Tobias J, Montgomerie R, Lyon BE (2012) The evolution of female ornaments and weaponry: social selection, sexual selection and ecological competition. Philos Trans R Soc B 367:2274–2293. https://doi.org/10.1098/rstb.2011.0280
Trewick SA, Bland KJ (2011) Fire and slice: palaeogeography for biogeography at New Zealand’s North Island/South Island juncture. JRSNZ 42:153–183. https://doi.org/10.1080/03036758.2010.549493
Trewick SA, Taylor-Smith B, Morgan-Richards M (2020) Ecology and systematics of the wine weta and allied species, with description of four new Hemiandrus species. N Z J Zool 48:47–80. https://doi.org/10.1080/03014223.2020.1790396
Tripet F, Touré YT, Taylor CE, Norris DE, Dolo G, Lanzaro GC (2001) DNA analysis of transferred sperm reveals significant levels of gene flow between molecular forms of Anopheles gambiae. Mol Ecol 10:1725–1732. https://doi.org/10.1046/j.0962-1083.2001.01301.x
Trivers RL (1972) Parental investment and sexual selection. In: Campbell B (ed) Sexual selection and the decent of man. Aldine, Chicago, USA, pp 136–179
Vahed K, Gilbert FS (1996) Differences across taxa in nuptial gift size correlate with differences in sperm number and ejaculate volume in bushcrickets (Orthoptera: Tettigoniidae). Proc R Soc B 263:1257–1265. https://doi.org/10.1098/rspb.1996.0185
Ward RD, Skibinski DOF, Woodwark M (1992) Protein heterozygosity, protein structure, and taxonomic differentiation. In: Hecht MK, Wallace B, Macintyre RJ (eds) Evolutionary Biology, vol 26. Springer, Boston, MA
Wearing-Wilde J (1996) Mate choice and competition in the barklouse Lepinotus patruelis (Psocoptera: Trogiidae): the effect of diet quality and sex ratio. J Insect Behav 9:599–612. https://doi.org/10.1007/BF02213883
Weissman DB (2001) Communication and reproductive behaviour in North American Jerusalem crickets. In: Field LH (ed) The biology of wetas, king crickets and their allies. CAB International, New York, pp 351–373
Wheeler J, Gwynne DT, Bussière LF (2012) Stabilizing sexual selection for female ornaments in a dance fly. J Evol Biol 25:1233–1242. https://doi.org/10.1111/j.1420-9101.2012.02522.x
Williams GC (1975) Sex and evolution. Princeton University Press, New Jersey
Wulff NC, Lehmann GUC (2016) Function of male genital titillators in mating and spermatophore transfer in the tettigoniid bushcricket Metrioptera roeselii. Biol J Linn Soc 117:206–216. https://doi.org/10.1111/bij.12661
Yoshizawa K, Ferreira RL, Kamimura Y, Lienhard C (2014) Female penis, male vagina, and their correlated evolution in a cave insect. Curr Biol 24:1006–1010. https://doi.org/10.1016/j.cub.2014.03.022
Yoshizawa K, Ferreira RL, Yao I, Lienhard C, Kamimura Y (2018) Independent origins of female penis and its coevolution with male vagina in cave insects (Psocodea: Prionoglarididae). Biol Lett 14:20180533. https://doi.org/10.1098/rsbl.2018.0533
Acknowledgements
We thank Mary Morgan Richards and Lindsay Coome for their help collecting data; Doug Currie, Marc Johnson, Rosalind Murray, and Clint Kelly for comments on the research and manuscript; and the New Zealand Department of Conservation (for permits) and anonymous referees for valuable comments on the manuscript.
Funding
This research was supported by a Natural Sciences and Engineering Research Council of Canada Discovery Grant to DTG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by N. Wedell
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Browne, J.H., Gwynne, D.T. Sexual selection on a female copulatory device in an insect with nuptial gifts. Behav Ecol Sociobiol 77, 138 (2023). https://doi.org/10.1007/s00265-023-03415-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03415-6