Abstract
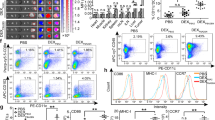
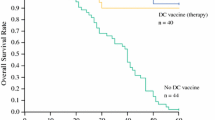
Hepatocellular carcinoma (HCC), a prevalent cause of cancer-related deaths, is insensitive to traditional treatments. At different time intervals, the combined antitumor effects of DC-TEX and the programmed death protein 1 (PD-1) antibody (Ab) have not been investigated. In this study, HCC models were established and treated at different time intervals with DC-TEX alone or in combination with PD-1 Ab. In addition, we developed an orthotopic HCC model in BALB/c nude mice and restored T cells. Results demonstrated that the PD-1 + CD8 + T-cell population also increased significantly after DC-TEX treatment, in addition to the increased number of CD8 + T cells. The number of CD8 + T cells increased 72 h after DC-TEX administration. Similar observations were made for PD-1 + CD8 + T cells. Subsequently, PD-1 Ab was administered in combination with DC-TEX at different time points (0, 24, 72, 96, 120, or 168 h). Surprisingly, the combination treatment demonstrated a strong antitumor effect, which was very prominent when PD-1 Ab was administered at 72 h. PD-1 Ab significantly reversed the proliferative ability of PD-1 + CD8 + T cells at 72 h in vitro. The combined antitumor effects of PD-1 Ab and DC-TEX occurred mainly by stimulating CD8 + T cell proliferation and inhibiting T cell exhaustion. In conclusion, our results indicate that the combination of DC-TEX and PD-1 Ab significantly inhibits tumor growth in a murine HCC model and that the timing of PD-1 Ab administration impacts the antitumor effect.






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69(1):7–34
Hou J, Zhang H, Sun B et al (2020) The immunobiology of hepatocellular carcinoma in humans and mice: basic concepts and therapeutic implications. J Hepatol 72(1):167–182
Schmidt TM, Liu LI, Abraham IE et al (2011) Efficacy and safety of sorafenib in a racially diverse patient population with advanced hepatocellular carcinoma. Anticancer Res 38(7):4027–4034
Juengpanich S, Topatana W, Chen Lu et al (2020) Role of cellular, molecular and tumor microenvironment in hepatocellular carcinoma: possible targets and future directions in the regorafenib era. Int J Cancer 147(7):1778–1792
Iñarrairaegui M, Melero I (2018) Immunotherapy of hepatocellular carcinoma: facts and hopes. Clin Cancer Res 24(7):1518–1524
Zhen Lu, Zuo B, **g R et al (2017) Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol 67(4):739–748
Zhang Q, Huang H, Zheng F et al (2020) Resveratrol exerts antitumor effects by downregulating CD8 + CD122 + Tregs in murine hepatocellular carcinoma. Oncoimmunology 9(1):1829346
Zhou H, Li Wu (2017) The development and function of dendritic cell populations and their regulation by miRNAs. Protein Cell 8(7):501–513
Merad M, Salmon H (2015) A dendritic-cell brake on antitumour immunity. Nature 523:294–295
Gurunathan S, Kang M-H, Jeyaraj M et al (2019) Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells 8(4):307
Wolfers J, Lozier A, Raposo G et al (2001) Tumor-derived exosomes in cancer metastasis risk diagnosis and metastasis therapy. Nat Med 7:297–303
Rao Q, Zuo B, Lu Z et al (2016) Tumor-derived exosomes elicit tumor suppression in murine hepatocellular carcinoma models and humans in vitro. Hepatology 64(2):456–472
Gholami MD, Kardar GA, Saeedi Y et al (2017) Exhaustion of T lymphocytes in the tumor microenvironment: significance and effective mechanisms. Cell Immunol 322:1–14
Celis-Gutierrez J, Blattmann P, Zhai Y et al (2019) Quantitative interactomics in primary T cells provides a rationale for concomitant PD-1 and BTLA coinhibitor blockade in cancer immunotherapy. Cell Rep 27(11):3315–3330
Kim H-D, Song G-W, Park S et al (2018) Association between expression level of PD1 by tumor-infiltrating CD8 + T cells and features of hepatocellular carcinoma. Gastroenterology 155(6):1936–1950
Chiu DK-C, Yuen VW-H, Cheu JW-S et al (2020) Hepatocellular carcinoma cells up-regulate PVRL1, stabilizing PVR and inhibiting the cytotoxic T-cell response via TIGIT to mediate tumor resistance to PD1 inhibitors in mice. Gastroenterology 159(2):609–623
Fourcade J, Sun Z, Benallaoua M et al (2010) Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med 207(10):2175–2186
Wang X, Yang X, Zhang C et al (2020) Tumor cell-intrinsic PD-1 receptor is a tumor suppressor and mediates resistance to PD-1 blockade therapy. Proc Natl Acad Sci U S A 117(12):6640–6650
Tumeh PC, Harview CL, Yearley JH et al (2014) PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515:568–571
Torre LA, Bray F, Siegel RL et al (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
Wculek SK, Cueto FJ, Mujal AM et al (2014) Dendritic cells in cancer immunology and immunotherapy. Curr Opin Immunol 27:26–32
Copier J, Dalgleish A (2020) Overview of tumor cell-based vaccines. Nat Rev Immunol 20(1):7–24
Li G, Liu D, Cooper TK et al (2017) Successful chemoimmunotherapy against hepatocellular cancer in a novel murine model. J Hepatol 66:75–85
Brunner SM, Rubner C, Kesselring R et al (2015) Tumor-infiltrating, interleukin-33-producing effector-memory CD8(+) T cells in resected hepatocellular carcinoma prolong patient survival. Hepatology 61:1957–1967
Di Blasio S, van Wigcheren GF, Becker A et al (2020) The tumour microenvironment shapes dendritic cell plasticity in a human organotypic melanoma culture. Nat Commun 11(1):2749
Chen Lu, Rong D, Zhang B et al (2019) Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: challenges and opportunities. Mol Cancer 18(1):130
Hendrickson PG, Olson M, Luetkens T et al (2019) The promise of adoptive cellular immunotherapies in hepatocellular carcinoma. Oncoimmunology 9(1):1673129
Flecken T, Schmidt N, Hild S et al (2014) Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology 59:1415–1426
Agata Y, Kawasaki A, Nishimura H et al (1996) Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol 8(5):765–772
Rosenberg SA, Yang JC, Restifo NP (2004) Cancer immunotherapy: moving beyond current vaccines. Nat Med 10:909–915
Jacquelot N, Yamazaki T, Roberti MP et al (2019) Sustained Type I interferon signaling as a mechanism of resistance to PD-1 blockade. Cell Res 29(10):846–861
Hailemichael Y, Dai Z, Jaffarzad N et al (2013) Persistent antigen at vaccination sites induces tumor-specific CD8(+) T cell sequestration, dysfunction and deletion. Nat Med 19:465–472
Danilova L, Wang H, Sunshine J et al (2016) Association of PD-1/PD-L axis expression with cytolytic activity, mutational load, and prognosis in melanoma and other solid tumors. Proc Natl Acad Sci U S A 113:E7769–E7777
Mariathasan S, Turley SJ, Nickles D et al (2018) TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554(7693):544–548
Sprinzl MF, Galle PR (2017) Current progress in immunotherapy of hepatocellular carcinoma. J Hepatol 66:482–484
Vibhakar R, Juan G, Traganos F et al (1997) Activation-induced expression of human programmed death-1 gene in T-lymphocytes. Exp Cell Res 232(1):25–28
Wu X, Zhang H, **ng Q et al (2014) PD-1(+) CD8(+) T cells are exhausted in tumours and functional in draining lymph nodes of colorectal cancer patients. Br J Cancer 111:1391–1399
Fourcade J, Sun Z, Pagliano O et al (2014) PD-1 and Tim-3 regulate the expansion of tumor antigen-specific CD8+ T cells induced by melanoma vaccines. Cancer Res 74:1045–1055
Sierro SR, Donda A, Perret R et al (2011) Combination of lentivector immunization and low-dose chemotherapy or PD-1/PD-L1 blocking primes self-reactive T cells and induces antitumor immunity. Eur J Immunol 41:2217–2228
Inada Y, Mizukoshi E, Seike T et al (2019) Characteristics of immune response to tumor-associated antigens and immune cell profile in patients with hepatocellular carcinoma. Hepatology 69(2):653–665
Finck A, Gill SI, June CH (2020) Cancer immunotherapy comes of age and looks for maturity. Nat Commun 11(1):3325
Acknowledgements
This work was supported by the Peking Union Medical College Innovation Research Team Fund, the Union Young Fund of Peking Union Medical College (3332016105) and the Sci-tech Development Project of Shandong Medicine and Health (2016WS0552) and the Natural Science Foundation of Shandong (ZR2017PH024) and the National Natural Science Foundation of China (81702821).
Author information
Authors and Affiliations
Contributions
CC and YP performed the experiments, analyzed the data and prepared the manuscript. CZ and WZ aided the data analysis and manuscript preparation. YQ contributed data analysis. SS designed and supervised the study, analyzed the data, prepared and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, C., Pei, Y., Zhang, C. et al. Combination therapy with dendritic cell loaded-exosomes supplemented with PD-1 inhibition at different time points have superior antitumor effect in hepatocellular carcinoma. Cancer Immunol Immunother 72, 3727–3738 (2023). https://doi.org/10.1007/s00262-023-03525-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03525-0