Abstract
Objective
To evaluate bone density changes at the level of normal trabecular bone and bone metastases (BMs) after denosumab (DM) treatment in oncologic patients.
Materials and methods
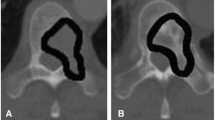
We retrospectively evaluated 31 consecutive adult patients with histologically confirmed solid tumors with at least one newly diagnosed bone metastatic lesion detected at CT. Patients received treatment with DM, 120 mg subcutaneous every 28 days for at least 6 months. Bone density was determined at the level of BMs and at the level of normal trabecular bone of lumbar vertebrae using a region of interest (ROI)–based approach.
Results
A progressive increase in CT bone density was demonstrated at the level of normal trabecular bone at 6 months (18% ± 5%) and 12 months (23% ± 7%) after the treatment begins. BMs showed a significant increase in CT bone density (p < 0.05) as compared to baseline after 6 months (57% ± 15%) and 12 months (1.06 ± 0.25 times higher) after treatment.
Conclusion
We have found that long-term treatment with DM increases bone density progressively in oncologic patients. This effect can be observed not only at the level of secondary lesions but also at the level of apparently normal trabecular bone and is more pronounced for osteolytic metastases.





Similar content being viewed by others
References
Tubiana-Hulin M. Incidence, prevalence and distribution of bone metastases. Bone. 1991;12(Suppl 1):S9–10.
Quattrocchi CC, Errante Y, Mallio CA, Santini D, Tonini G, Zobel BB. Brain metastatic volume and white matter lesions in advanced cancer patients. J Neurooncol. 2013;113(3):451–8.
Mundy GR. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer. 2002;2(8):584–93.
Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20 Pt 2):6243s–9s.
Mirels H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res. 1989;249:256–64.
Hill ME, Richards MA, Gregory WM, Smith P, Rubens RD. Spinal cord compression in breast cancer: a review of 70 cases. Br J Cancer. 1993;68(5):969–73.
Grill V, Ho P, Body JJ, Johanson N, Lee SC, Kukreja SC, et al. Parathyroid hormone-related protein: elevated levels in both humoral hypercalcemia of malignancy and hypercalcemia complicating metastatic breast cancer. J Clin Endocrinol Metab. 1991;73(6):1309–15.
Hamaoka T, Madewell JE, Podoloff DA, Hortobagyi GN, Ueno NT. Bone imaging in metastatic breast cancer. J Clin Oncol. 2004;22(14):2942–53.
Roodman GD. Mechanisms of bone metastasis. N Engl J Med. 2004;350(16):1655–64.
Mallio CA, Napolitano A, Castiello G, Giordano FM, D'Alessio P, Iozzino M, et al. Deep learning algorithm trained with COVID-19 pneumonia also identifies immune checkpoint inhibitor therapy-related pneumonitis. Cancers (Basel). 2021;13(4):652.
Quattrocchi CC, Mallio CA, Presti G, Beomonte Zobel B, Cardinale J, Iozzino M, et al. The challenge of COVID-19 low disease prevalence for artificial intelligence models: report of 1,610 patients. Quant Imaging Med Surg. 2020;10(9):1891–3.
Hayward JL, Carbone PP, Heusen JC, Kumaoka S, Segaloff A, Rubens RD. Assessment of response to therapy in advanced breast cancer. Br J Cancer. 1977;35(3):292–8.
Mundy GR. Mechanisms of bone metastasis. Cancer. 1997;80(Suppl. 8):1546–56.
Shih LY, Shih HN, Chen TH. Bone resorption activity of osteolytic metastatic lung and breast cancers. J Orthop Res. 2004;22(6):1161–7.
Body JJ. Clinical trials in metastatic breast cancer to bone: past–present–future. Can J Oncol. 1995;5(Suppl.1):16–27.
Hortobagyi GN, Theriault RL, Porter L, Blayney D, Lipton A, Sinoff C, et al. Efficacy of pamidronate in reducing skeletal complications in patients with breast cancer and lytic bone metastases. N Engl J Med. 1996;335(24):1785–91.
Purohit OP, Anthony C, Radstone CR, Owen J, Coleman RE. High-dose intravenous pamidronate for metastatic bone pain. Br J Cancer. 1994;70(3):554–8.
Berenson JR, Lichtenstein A, Porter L, Dimopoulos MA, Bordoni R, George S, et al. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. N Engl J Med. 1996;334(8):488–93.
Paterson AH, Powles TJ, Kanis JA, McCloskey E, Hanson J, Ashley S. Double-blind controlled trial of oral clodronate in patients with bone metastases from breast cancer. J Clin Oncol. 1993;11(1):59–65.
Lahtinen R, Laakso M, Palva I, Virkkunen P, Elomaa I. Randomised, placebo-controlled multicentre trial of clodronate in multiple myeloma. Lancet. 1992;340(8827):1049–52.
Saad F, Lipton A. Zoledronic acid is effective in preventing and delaying skeletal events in patients with bone metastases secondary to genitourinary cancers. BJU Int. 2005;96(7):964–9.
Brown JE, Cook RJ, Major P, Lipton A, Saad F, Smith M, et al. Bone turnover markers as predictors of skeletal complications in prostate cancer, lung cancer, and other solid tumors. J Natl Cancer Inst. 2005;97(1):59–69.
Silverman SL. Paget disease of bone: therapeutic options. J Clin Rheumatol. 2008;14(5):299–305.
Kawada K, Minami H, Okabe K, Watanabe T, Inoue K, Sawamura M, et al. A multicenter and open label clinical trial of zoledronic acid 4 mg in patients with hypercalcemia of malignancy. Jpn J Clin Oncol. 2005;35(1):28–33.
Reed SD, Radeva JI, Glendenning GA, Coleman RE, Schulman KA. Economic evaluation of zoledronic acid versus pamidronate for the prevention of skeletal-related events in metastatic breast cancer and multiple myeloma. Am J Clin Oncol. 2005;28(1):8–16.
James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP, Anderson J, et al. Systemic therapy for advancing or metastatic prostate cancer: a multi-arm, multistage randomized controlled trial. BJU Int. 2009;103(4):464–9.
Langer CJ. Selected clinical trials in advanced non-small-cell lung cancer. Clin Lung Cancer. 2010;11(5):358–9.
Rosen LS, Gordon DH, Dugan W Jr, Major P, Eisenberg PD, Provencher L. Zoledronic acid is superior to pamidronate for the treatment of bone metastases in breast carcinoma patients with at least one osteolytic lesion. Cancer. 2004;100(1):36–43.
Body JJ. Rationale for the use of bisphosphonates in osteoblastic and osteolytic bone lesions. Breast. 2003;12(Suppl. 2):37–44.
Santini D, Fratto ME, Vincenzi B, Galluzzo S, Tonini G. Zoledronic acid in the management of metastatic bone disease. Expert Opin Biol Ther. 2006;6(12):1333–48.
McClung MR, Lewiecki EM, Cohen SB, Bolognese MA, Woodson GC, Moffett AH, et al. Denosumab in postmenopausal women with low bone mineral density. N Engl J Med. 2006;354(8):821–31.
Wan Y, Zeng F, Tan H, Lu Y, Zhang Y, Zhao L, You R. Cost-effectiveness analyses of denosumab for osteoporosis: a systematic review. Osteopor Int. 2022;33(5):979–1015.
Ko HW, Chiu CT, Wang CL, Yang TY, Liu CY, Yu CT, et al. Overall survival improvement in patients with epidermal growth factor receptor-mutated non-small cell lung cancer and bone metastasis treated with denosumab. Cancers (Basel). 2022;14((14):3470.
Quattrocchi CC, Dell'aia P, Errante Y, Occhicone F, Longo D, Virzì V, et al. Differential effect of zoledronic acid on normal trabecular and cortical bone density in oncologic patients with bone metastases. J Bone Oncol. 2012;1(1):24–9.
Quattrocchi CC, Santini D, Dell'aia P, Piciucchi S, Leoncini E, Vincenzi B, et al. A prospective analysis of CT density measurements of bone metastases after treatment with zoledronic acid. Skeletal Radiol. 2007;36(12):1121–7.
Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361(8):756–65.
Kutleša Z, Jerković K, Ordulj I, Budimir MD. The effect of contrast media on CT measures of bone mineral density: a systematic review. Skeletal Radiol. 2023;52(4):687–94.
Zebaze RM, Libanati C, Austin M, Ghasem-Zadeh A, Hanley DA, Zanchetta JR, et al. Differing effects of denosumab and alendronate on cortical and trabecular bone. Bone. 2014;59:173–9.
Hanley DA, Adachi JD, Bell A, Brown V. Denosumab: mechanism of action and clinical outcomes. Int J Clin Pract. 2012;66(12):1139–46.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mallio, C.A., Greco, F., Gaudino, F. et al. Computed tomography density changes of bone metastases after concomitant denosumab. Skeletal Radiol 52, 1567–1575 (2023). https://doi.org/10.1007/s00256-023-04326-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-023-04326-3