Abstract
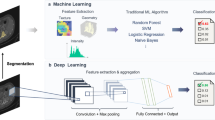
Artificial intelligence and deep learning (DL) offer musculoskeletal radiology exciting possibilities in multiple areas, including image reconstruction and transformation, tissue segmentation, workflow support, and disease detection. Novel DL-based image reconstruction algorithms correcting aliasing artifacts, signal loss, and noise amplification with previously unobtainable effectiveness are prime examples of how DL algorithms deliver promised value propositions in musculoskeletal radiology. The speed of DL-based tissue segmentation promises great efficiency gains that may permit the inclusion of tissue compositional-based information routinely into radiology reports. Similarly, DL algorithms give rise to a myriad of opportunities for workflow improvements, including intelligent and adaptive hanging protocols, speech recognition, report generation, scheduling, precertification, and billing. The value propositions of disease-detecting DL algorithms include reduced error rates and increased productivity. However, more studies using authentic clinical workflow settings are necessary to fully understand the value of DL algorithms for disease detection in clinical practice. Successful workflow integration and management of multiple algorithms are critical for translating the value propositions of DL algorithms into clinical practice but represent a major roadblock for which solutions are critically needed. While there is no consensus about the most sustainable business model, radiology departments will need to carefully weigh the benefits and disadvantages of each commercially available DL algorithm. Although more studies are needed to understand the value and impact of DL algorithms on clinical practice, DL technology will likely play an important role in the future of musculoskeletal imaging.
Similar content being viewed by others
References
LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521(7553):436–44.
Kijowski R, Liu F, Caliva F, Pedoia V. Deep learning for lesion detection, progression, and prediction of musculoskeletal disease. J Magn Reson Imaging. 2020;52(6):1607–19.
Gyftopoulos S, Lin D, Knoll F, Doshi AM, Rodrigues TC, Recht MP. Artificial intelligence in musculoskeletal imaging: current status and future directions. AJR Am J Roentgenol. 2019;213(3):506–13.
Chea P, Mandell JC. Current applications and future directions of deep learning in musculoskeletal radiology. Skelet Radiol. 2020;49(2):183–97.
Grauhan NF, Niehues SM, Gaudin RA, Keller S, Vahldiek JL, Adams LC, et al. Deep learning for accurately recognizing common causes of shoulder pain on radiographs. Skeletal Radiol. 2021.
Guan B, Liu F, Mizaian AH, Demehri S, Samsonov A, Guermazi A, et al. Deep learning approach to predict pain progression in knee osteoarthritis. Skeletal Radiol. 2021.
Del Grande F, Guggenberger R, Fritz J. Rapid musculoskeletal MRI in 2021: value and optimized use of widely accessible techniques. AJR Am J Roentgenol. 2021;216(3):704–17.
Fritz J, Guggenberger R, Del Grande F. Rapid musculoskeletal MRI in 2021: clinical application of advanced accelerated techniques. AJR Am J Roentgenol. 2021;216(3):718–33.
Recht MP, Zbontar J, Sodickson DK, Knoll F, Yakubova N, Sriram A, et al. Using deep learning to accelerate knee MRI at 3 T: results of an interchangeability study. AJR Am J Roentgenol. 2020;215(6):1421–9.
Del Grande F, Rashidi A, Luna R, Delcogliano M, Stern SE, Dalili D, et al. Five-minute five-sequence knee MRI using combined simultaneous multislice and parallel imaging acceleration: comparison with 10-minute parallel imaging knee MRI. Radiology. 2021;203655.
Nakamura Y, Higaki T, Tatsugami F, Honda Y, Narita K, Akagi M, et al. Possibility of deep learning in medical imaging focusing improvement of computed tomography image quality. J Comput Assist Tomogr. 2020;44(2):161–7.
Jans LBO, Chen M, Elewaut D, Van den Bosch F, Carron P, Jacques P, et al. MRI-based synthetic CT in the detection of structural lesions in patients with suspected sacroiliitis: comparison with MRI. Radiology. 2021;298(2):343–9.
Fayad LM, Parekh VS, de Castro Luna R, Ko CC, Tank D, Fritz J, et al. A deep learning system for synthetic knee magnetic resonance imaging: is artificial intelligence-based fat-suppressed imaging feasible? Invest Radiol. 2020; Publish Ahead of Print.
Breighner RE, Endo Y, Konin GP, Gulotta LV, Koff MF, Potter HG. Technical developments: zero echo time imaging of the shoulder: enhanced osseous detail by using MR imaging. Radiology. 2018;286(3):960–6.
Fritz J. Automated and radiation-free generation of synthetic CT from MRI data: does AI help to cross the finish line? Radiology. 2021;298(2):350–2.
Medina G, Buckless CG, Thomasson E, Oh LS, Torriani M. Deep learning method for segmentation of rotator cuff muscles on MR images. Skelet Radiol. 2021;50(4):683–92.
Liu F, Zhou Z, Samsonov A, Blankenbaker D, Larison W, Kanarek A, et al. Deep learning approach for evaluating knee MR images: achieving high diagnostic performance for cartilage lesion detection. Radiology. 2018;289(1):160–9.
Boutin RD, Lenchik L. Value-added opportunistic CT: insights into osteoporosis and sarcopenia. AJR Am J Roentgenol. 2020;215(3):582–94.
Kalra A, Chakraborty A, Fine B, Reicher J. Machine learning for automation of radiology protocols for quality and efficiency improvement. J Am Coll Radiol. 2020;17(9):1149–58.
Lee M, Kasmanoff N, Parente M, Razavian N, Lui Y. Using deep multi-task learning to classify brain MR images by sequence and orientation. American Society of Neuroradiology Annual Meeting. 2020.
Quint LE, Quint DJ, Myles JD. Frequency and spectrum of errors in final radiology reports generated with automatic speech recognition technology. J Am Coll Radiol. 2008;5(12):1196–9.
Chew FS, Mulcahy MJ, Porrino JA, Mulcahy H, Relyea-Chew A. Prevalence of burnout among musculoskeletal radiologists. Skeletal Radiol. 2017;46(4):497–506.
Chong LR, Tsai KT, Lee LL, Foo SG, Chang PC. Artificial intelligence predictive analytics in the management of outpatient MRI appointment no-shows. AJR Am J Roentgenol. 2020;215(5):1155–62.
Harvey HB, Liu C, Ai J, Jaworsky C, Guerrier CE, Flores E, et al. Predicting no-shows in radiology using regression modeling of data available in the electronic medical record. J Am Coll Radiol. 2017;14(10):1303–9.
Curtis C, Liu C, Bollerman TJ, Pianykh OS. Machine learning for predicting patient wait times and appointment delays. J Am Coll Radiol. 2018;15(9):1310–6.
Fritz B, Muller DA, Sutter R, Wurnig MC, Wagner MW, Pfirrmann CWA, et al. Magnetic resonance imaging-based grading of cartilaginous bone tumors: added value of quantitative texture analysis. Investig Radiol. 2018;53(11):663–72.
Gillies RJ, Kinahan PE, Hricak H. Radiomics: images are more than pictures, they are data. Radiology. 2016;278(2):563–77.
Harvey HB, Gowda V. Clinical applications of AI in MSK imaging: a liability perspective. Skelet Radiol. 2021.
Yang S, Yin B, Cao W, Feng C, Fan G, He S. Diagnostic accuracy of deep learning in orthopaedic fractures: a systematic review and meta-analysis. Clin Radiol. 2020;75(9):713 e717–28.
Chung SW, Han SS, Lee JW, Oh KS, Kim NR, Yoon JP, et al. Automated detection and classification of the proximal humerus fracture by using deep learning algorithm. Acta Orthop. 2018;89(4):468–73.
Urakawa T, Tanaka Y, Goto S, Matsuzawa H, Watanabe K, Endo N. Detecting intertrochanteric hip fractures with orthopedist-level accuracy using a deep convolutional neural network. Skelet Radiol. 2019;48(2):239–44.
Choi JW, Cho YJ, Lee S, Lee J, Lee S, Choi YH, et al. Using a dual-input convolutional neural network for automated detection of pediatric supracondylar fracture on conventional radiography. Investig Radiol. 2020;55(2):101–10.
Gan K, Xu D, Lin Y, Shen Y, Zhang T, Hu K, et al. Artificial intelligence detection of distal radius fractures: a comparison between the convolutional neural network and professional assessments. Acta Orthop. 2019;90(4):394–400.
Yu JS, Yu SM, Erdal BS, Demirer M, Gupta V, Bigelow M, et al. Detection and localisation of hip fractures on anteroposterior radiographs with artificial intelligence: proof of concept. Clin Radiol. 2020;75(3):237 e231–9.
Fritz B, Marbach G, Civardi F, Fucentese SF, Pfirrmann CWA. Deep convolutional neural network-based detection of meniscus tears: comparison with radiologists and surgery as standard of reference. Skelet Radiol. 2020;49(8):1207–17.
Bien N, Rajpurkar P, Ball RL, Irvin J, Park A, Jones E, et al. Deep-learning-assisted diagnosis for knee magnetic resonance imaging: development and retrospective validation of MRNet. PLoS Med. 2018;15(11):e1002699.
Liu F, Guan B, Zhou Z, Samsonov A, Rosas H, Lian K, et al. Fully automated diagnosis of anterior cruciate ligament tears on knee MR images by using deep learning. Radiol Artif Intell. 2019;1(3):180091.
Germann C, Marbach G, Civardi F, Fucentese SF, Fritz J, Sutter R, et al. Deep convolutional neural network-based diagnosis of anterior cruciate ligament tears: performance comparison of homogenous versus heterogeneous knee MRI cohorts with different pulse sequence protocols and 1.5-T and 3-T magnetic field strengths. Investig Radiol. 2020;55(8):499–506.
Fritz J, Germann C, Sutter R, Fritz B. AI-augmented MRI diagnosis of ACL tears: which readers benefit? SSR 2021 Annual Meeting. 2021.
Zhang B, Jia C, Wu R, Lv B, Li B, Li F, et al. Improving rib fracture detection accuracy and reading efficiency with deep learning-based detection software: a clinical evaluation. Br J Radiol. 2021;94(1118):20200870.
Benjamens S, Dhunnoo P, Mesko B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit Med. 2020;3:118.
Leiner T, Bennink E, Mol CP, Kuijf HJ, Veldhuis WB. Bringing AI to the clinic: blueprint for a vendor-neutral AI deployment infrastructure. Insights Imaging. 2021;12(1):11.
Yi PH, Arun A, Hafezi-Nejad N, Choy G, Sair HI, Hui FK, et al. Garbage In, Garbage out? Performance of state-of-the-art deep learning-based bone age algorithm on inappropriate data inputs. SSR 2021 Annual Meeting 2021.
Yi PH, Kim TK, Wei J, Shin J, Hui FK, Sair HI, et al. Automated semantic labeling of pediatric musculoskeletal radiographs using deep learning. Pediatr Radiol. 2019;49(8):1066–70.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jan Fritz received institutional research support from Siemens AG, BTG International, Zimmer Biomed, DePuy Synthes, QED, and SyntheticMR; is a scientific advisor for Siemens AG, SyntheticMR, GE Healthcare, QED, BTG, ImageBiopsy Lab, Boston Scientific, and Mirata Pharma; and has shared patents with Siemens Healthcare and Johns Hopkins University. Richard Kijowski: none. Michael Recht: none.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fritz, J., Kijowski, R. & Recht, M.P. Artificial intelligence in musculoskeletal imaging: a perspective on value propositions, clinical use, and obstacles. Skeletal Radiol 51, 239–243 (2022). https://doi.org/10.1007/s00256-021-03802-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-021-03802-y