Abstract
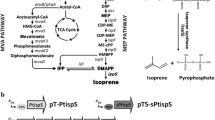
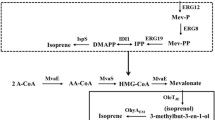
Isoprene is a useful phytochemical with high commercial values in many industrial applications including synthetic rubber, elastomers, isoprenoid medicines, and fossil fuel. Currently, isoprene is on large scale produced from petrochemical sources. An efficient biological process for isoprene production utilizing renewable feedstocks would be an important direction of research due to the fossil raw material depletion and air pollution. In this study, we introduced the mevalonate (MVA) pathway genes/acetoacetyl-coenzyme A thiolase (mvaE) and MVA synthase (mvaS) from Enterococcus faecalis (E. faecalis); MVA kinase (mvk) derived from Methanosarcina mazei (M. mazei); and phosphomevalonate kinase (pmk), diphosphomevalonate decarboxylase (mvaD), and isopentenyl diphosphate isomerase (idi) from Streptococcus pneumoniae (S. pneumoniae) to accelerate dimethylallyl diphosphate (DMAPP) accumulation in Escherichia coli (E. coli). Together with a codon-optimized isoprene synthase (ispS) from Populus alba (P. alba), E. coli strain succeeded in formation of isoprene. We then manipulated the heterologous MVA pathway for high-level production of isoprene, by controlling the gene expression levels of the MVA pathway genes. We engineered four E. coli strains which showed different gene expression levels and different isoprene productivities, and we also characterized them with quantitative real-time PCR and metabolite analysis. To further improve the isoprene titers and release the toxicity to cells, we developed the extraction fermentation by adding dodecane in cultures. Finally, strain BL2T7P1TrcP harboring balanced gene expression system produced 587 ± 47 mg/L isoprene, with a 5.2-fold titer improvement in comparison with strain BL7CT7P. This work indicated that a balanced metabolic flux played a significant role to improve the isoprene production via MVA pathway.







Similar content being viewed by others
References
Alonso-Gutierrez J, Kim EM, Batth TS, Cho N, Hu Q, Chan LJ, Petzold CJ, Hillson NJ, Adams PD, Keasling JD, Martin GH, Lee TS (2015) Principal component analysis of proteomics (PCAP) as a tool to direct metabolic engineering. Metab Eng 28:123–133
Dı’az EE, Stams JMA, Amils R, Sanz LJ (2006) Phenotypic properties and microbial diversity of methanogenic granules from a full-scale upflow anaerobic sludge bed reactor treating brewery wastewater. Appl Environ Microbiol 72(7):4942–4949
Eisenreich W, Bacher A, Arigoni D, Rohdich F (2004) Biosynthesis of isoprenoids via the non-mevalonate pathway. Cell Mol Life Sci 61:1401–1426
Henneman L, van Cruchten AG, Denis SW, Amolins MW, Placzek AT, Gibbs RA, Kulik W, Waterham HR (2008) Detection of nonsterol isoprenoids by HPLC-MS/MS. Anal Biochem 383:18–24
Julsing MK, Rijpkema M, Woerdenbag HJ, Quax WJ, Kayser O (2007) Functional analysis of genes involved in the biosynthesis of isoprene in Bacillus subtilis. Appl Microbiol Biotechnol 75:1377–1384
Kevin W, George AC, Jain A, Batth TS, Baidoo EEK, Wang G, Adams PD, Petzold CJ, Keasling JD, Lee TS (2014) Correlation analysis of targeted proteins and metabolites to assess and engineer microbial isopentenol production. Biotechnol Bioeng 111:11
Kim J, Wang C, Jang H-J, Cha M-S, Park J-E, Jo S-Y, Choi E-S, Kim S-W (2016) Isoprene production by Escherichia coli through the exogenous mevalonate pathway with reduced formation of fermentation byproducts. Microb Cell Factories 15(1):214
Kuzma J, Nemecek-Marshall M, Pollock WH, Fall R (1995) Bacteria produce the volatile hydrocarbon isoprene. Curr Microbiol 30:97–103
Liu C-L, Fan L-H, Liu L, Tan T-W (2014) Combinational biosynthesis of isoprene by engineering the MEP pathway in Escherichia coli. Process Biochem 49:2078–2085
Liu C-L, Lv Q, Tan T-W (2015) Joint antisense RNA strategies for regulating isoprene production in Escherichia coli. RSC Adv 5:74892–74898
Pitera DJ, Paddon CJ, Newman JD, Keasling JD (2007) Balancing a heterologous mevalonate pathway for improved isoprenoid production in Escherichia coli. Metab Eng 9:193–207
Primak YA, Du M, Miller MC, Wells DH, Nielsen AT, Weyler W, Beck ZQ (2011) Characterization of a feedback-resistant mevalonate kinase from the archaeon Methanosarcina mazei. Appl Environ Microbiol 77:7772–7778
Redding-Johanson AM, Batth TS, Chan R, Krupa R, Szmidt HL, Adams PD, Keasling JD, Lee TS, Mukhopadhyay A, Petzold CJ (2011) Targeted proteomics for metabolic pathway optimization: application to terpene production. Metab Eng 13:194–203
Rohmer M (1999) A mevalonate-independent route to isopentenyl diphosphate. Compr Nat Products Chem 45–67
Sanchez JJ, Berenguer JA, Calderon V, Herce MD (1991) Spoilage of a bakery product by isoprene-producing molds. Rev Agroquim Technol Aliment 31:4
Sharkey TD, Yeh SS (2001) Isoprene emission from plants. Annu Rev Plant Physiol Plant Mol Biol 52:407–436
Whited MG, Feher JF, Benko AD, Cervin AM, Chotani KG, McAuliffe CJ, LaDuca JR, Ben-Shoshan AE, Sanford JK (2010) Development of a gas-phase bioprocess for isoprene-monomer production using metabolic pathway engineering. Ind Biotechnol 12:152–163
Xue J, Ahring BK (2011) Enhancing isoprene production by genetic modification of the 1-deoxy-d-xylulose-5-phosphate pathway in Bacillus subtilis. Appl Environ Microbiol 77:2399–2405
Yang J, Zhao G, Sun Y, Zheng Y, Jiang X, Liu W, **an M (2012) Bio-isoprene production using exogenous MVA pathway and isoprene synthase in Escherichia coli. Bioresour Technol 104:642–647
Yang C, Gao X, Jiang Y, Sun B, Gao F, Yang S (2016) Synergy between methylerythritol phosphate pathway and mevalonate pathway for isoprene production in Escherichia coli. Metab Eng 37:79–91
Ye L, Lv X, Yu H (2016) Engineering microbes for isoprene production. Metab Eng 38:125–138
Yoon SH, Lee YM, Kim JE, Lee SH, Lee JH, Kim JY, Jung KH, Shin YC, Keasling JD, Kim SW (2006) Enhanced lycopene production in Escherichia coli engineered to synthesize isopentenyl diphosphate and dimethylallyl diphosphate from mevalonate. Biotechnol Bioeng 94:1025–1032
Yoon SH, Lee SH, Das A, Ryu HK, Jang HJ, Kim JY, Oh DK, Keasling JD, Kim SW (2009) Combinatorial expression of bacterial whole mevalonate pathway for the production of beta-carotene in E. coli. J Biotechnol 140:218–226
Zahiri HS, Yoon SH, Keasling JD, Lee SH, Won Kim S, Yoon SC, Shin YC (2006) Coenzyme Q10 production in recombinant Escherichia coli strains engineered with a heterologous decaprenyl diphosphate synthase gene and foreign mevalonate pathway. Metab Eng 8:406–416
Zurbriggen A, Kirst H, Melis A (2012) Isoprene production via the mevalonic acid pathway in Escherichia coli (Bacteria). BioEnergy Res 5(4):814–828
Acknowledgments
We thank ** to revise this manuscript.
Funding
This work was supported by the State key laboratory of organic–inorganic composites, the National Basic Research Program of China (973 program) (2013CB733600), the National Nature Science Foundation of China (21390202, 21476017).
Author information
Authors and Affiliations
Contributions
C.-L. Liu and T.-W. Tan designed the study. C.-L. Liu and H.-R. Bi performed the experiments. C.-L. Liu, B. Hu, and L.-H. Fan are involved in the manuscript writing and editing. All the authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical statement
This article does not contain any studies with human participants performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 443 kb)
Rights and permissions
About this article
Cite this article
Liu, CL., Bi, HR., Bai, Z. et al. Engineering and manipulation of a mevalonate pathway in Escherichia coli for isoprene production. Appl Microbiol Biotechnol 103, 239–250 (2019). https://doi.org/10.1007/s00253-018-9472-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9472-9