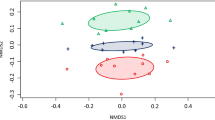
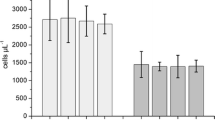
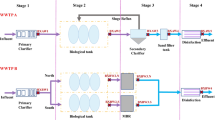
Abstract
This study found that the ratio of Thiothrix eikelboomii to total bacterial concentrations (TH/TB) (%) was a better indicator of bulking incidents affecting effluent quality compared to absolute T. eikelboomii abundance alone. This was determined using a genus-specific Thiothrix quantitative PCR primer and probe set, which was developed in this study to monitor specific Thiothrix populations over a 1-year period. T. eikelboomii was identified as the source of bulking incidents based on sequencing of the 16S rRNA gene at a nitrifying–denitrifying wastewater treatment plant. Peak T. eikelboomii concentrations observed in March, April, and July 2009 were 2.32 × 1010, 2.64 × 1010, and 1.84 × 1010 cells/l, respectively. The highest fraction of T. eikelboomii to total bacterial population was measured at 0.24% in March, and a ratio >0.19% caused increases of suspended solids and biochemical oxygen demand in the secondary effluent. Additionally, food/mass ratios, dissolved oxygen concentrations in the anoxic selector, and ammonium ion concentrations in the primary effluent were three parameters displaying statistically significant correlations (r = 0.40, r = 0.50, and r = 0.32, respectively) to Thiothrix spp. abundance in an aeration tank. No bulking events caused by T. eikelboomii occurred when the dissolved oxygen concentrations in the anoxic selector was maintained at lower than 0.12 mg/l and the TH/TB ratios were <0.10%.




Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
American Public Health Association/American Water Works Association/Water Environment Federation (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association/American Water Works Association/Water Environment Federation, Washington, DC
Aruga S, Kamagata Y, Kohno T, Hanada S, Nakamura K, Kanagawa T (2002) Characterization of filamentous Eikelboom type 021N bacteria and description of Thiothrix disciformis sp. nov. and Thiothrix flexilis sp. nov. Inter J Syst Evol Micr 52:1309–1316
Asvapathanagul P, Huang ZH, Gedalanga PB, Baylor AM, Olson BH (2012) Interaction of operational and physicochemical factors leading to Gordonia amarae-like foaming in an incompletely nitrifying activated sludge plant. Appl Environ Microbiol 78(23):8165–8175
Bikandi J, San Millan R, Rementeria A, Garaizar J (2004) In silico analysis of complete bacterial genomes: PCR, AFLP-PCR and endonuclease restriction. Bioinformatics 20(5):798–799
Bitton G (2011) Wastewater microbiology, New Jersey, US
Blackall LL, Tandoi V, Jenkins D (1991) Continuous culture studies with Nocardia-amarae from activated-sludge and their implications for Nocardia foaming control. J Water Pollut Con F 63(1):44–49
Chudoba J, Grau P, Ottova V (1973) Control of activated-sludge filamentous bulking—II. Selection of microorganisms by means of a selector. Water Res 7(10):1389–1398
Dumonceaux TJ, Hill JE, Pelletier CP, Paice MG, Van Kessel AG, Hemmingsen SM (2006) Molecular characterization of microbial communities in Canadian pulp and paper activated sludge and quantification of a novel Thiothrix eikelboomii-like bulking filament. Can J Microbiol 52(5):494–500
Eikelboom DH (1975) Filamentous organisms observed in activated-sludge. Water Res 9(4):365–388
Eikelboom DH (1977) Identification of filamentous organisms in bulking activated-sludge. Prog Water Technol 8(6):153–161
Ferris M, Muyzer G, Ward DM (1996) Denaturing gradient gel electrophoresis profiles of 16S rRNA-defined populations inhabiting a hot spring microbial mat community. Appl Environ Microbiol 62(2):340–346
Gedalanga PB (2010) Molecular-based analysis and monitoring of microbial groups in activated sludge to advance the knowledge of biological processes in wastewater treatment. Ph.D.dissertation, University of California at Irvine, CA
Gedalanga PB, Olson BH (2009) Development of a quantitative PCR method to differentiate between viable and nonviable bacteria in environmental water samples. Appl Microbiol Biotechnol 82(3):587–596
Guo JH, Peng YZ, Wang SY, Yang X, Wang ZW, Zhu A (2012) Stable limited filamentous bulking through kee** the competition between floc-formers and filaments in balance. Bioresour Technol 103(1):7–15
Harms G, Layton AC, Dionisi HM, Gregory IR, Garrett VM, Hawkins SA, Robinson KG, Sayler GS (2003) Real-time PCR quantification of nitrifying bacteria in a municipal wastewater treatment plant. Environ Sci Technol 37(2):343–351
Howarth R, Unz RF, Seviour EM, Seviour RJ, Blackall LL, Pickup RW, Head IM (1999) Phylogenetic relationships of filamentous sulfur bacteria (Thiothrix spp. and Eikelboom type 021N bacteria) isolated from wastewater-treatment plants and description of Thiothrix eikelboomii sp. nov., Thiothrix unzii sp. nov., Thiothrix fructosivorans sp. nov. and Thiothrix defluvii sp. nov. Inter J Syst Bacteriol 49:1817–1827
Jenkins D, Richard MG, Neethling JB (1984) Causes and control of activated-sludge bulking. J Water Pollut Con F 83(4):455–472
Jenkins D, Richard MG, Daigger GT (2003) Manual on causes and control of activated sludge bulking, foaming, and other solids separation problems. CRC Press; 3 edition, London
Kanagawa T, Kamagata Y, Aruga S, Kohno T, Horn M, Wagner M (2000) Phylogenetic analysis of and oligonucleotide probe development for Eikelboom type 021N filamentous bacteria isolated from bulking activated sludge. Appl Environ Microbiol 66(11):5043–5052
Klappenbach JA, Saxman PR, Cole JR, Schmidt TM (2001) rrndb: the ribosomal RNA operon copy number database. Nucleic Acids Res 29(1):181–184
Kristensen GH, Jorgensen PE, Nielsen PH (1994) Settling characteristics of activated-sludge in Danish treatment plants with biological nutrient removal. Water Sci Technol 29(7):157–165
Lane DJ (1991) Nucleic acid techniques in bacterial systematics. John Wiley & Sons, Chichester, UK
Lapidus A, Nolan M, Lucas M, Glavina Del Rio T, Tice H, Cheng JF, Tapia R, Han C, Goodwin L, Pitluck S, Liolios K, Pagani I, Ivanova N, Huntemann M, Mavromatis K, Mikhailova N, Pati A, Chen A, Palaniappan K, Land M, Brambilla EM, Rohde M, Abt B, Verbarg S, Göker M, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk H-P, Woyke T (2011) Genome sequence of the filamentous, gliding Thiothrix nivea neotype strain (JP2 (T)). Stand Genomic Sci 5(3):398–406
Lechevallier MW, Cawthon CD, Lee RG (1988) Inactivation of biofilm bacteria. Appl Environ Microbiol 54(10):2492–2499
Lee SE, Koopman B, Bode H, Jenkins D (1983) Evaluation of alternative sludge settleability indexes. Water Res 17(10):1421–1426
Madoni P, Davoli D, Gibin G (2000) Survey of filamentous microorganisms from bulking and foaming activated-sludge plants in Italy. Water Res 34(6):1767–1772
Marmur J, Doty P (1962) Determination of the base composition of deoxyribonucleic acid from its thermal denaturation temperature. J Mol Biol 5(1):109–118
Martins AMP, Heijnen JJ, van Loosdrecht MCM (2004) Bulking sludge in biological nutrient removal systems. Biotechnol Bioeng 86(2):125–135
Microsoft (2007) Microsoft Excel. Microsoft, Redmond, WA
Nielsen PH, de Muro MA, Nielsen JL (2000) Studies on the in situ physiology of Thiothrix spp. present in activated sludge. Environ Microbiol 2(4):389–398
Pacheco SVF, Jauregui RB, Pavon STB, Mejia PGV (2003) Control of growing of filamentous microorganisms in an industrial sewage treatment plant. Rev Int Contaminacion Ambient 19:47–53
Palm JC, Jenkins D, Parker DS (1980) Relationship between organic loading, dissolved-oxygen concentration and sludge settleability in the completely-mixed activated-sludge process. J Water Pollut Con F 52(10):2484–2506
Pernelle JJ, Gaval G, Cotteux E, Duchene P (2001) Influence of transient substrate overloads on the proliferation of filamentous bacterial populations in an activated sludge pilot plant. Water Res 35(1):129–134
Richard MG, Jenkins D, Hao O, Shimizu G (1982) The isolation and characterization of filamentous microorganisms form activated sludge bulking. Sanitary Engineering Health Research Laboratory, University of California, Berkeley
Richard MG, Shimizu GP, Jenkins D (1985) The growth physiology of the filamentous organism type-021N and its significance to activated-sludge bulking. J Water Pollut Con F 57(12):1152–1162
Ridgway HF, Olson BH (1982) Chlorine resistance patterns of bacteria from 2 drinking-water distribution-systems. Appl Environ Microbiol 44(4):972–987
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Schuler AJ, Jassby D (2007) Filament content threshold for activated sludge bulking: artifact or reality? Water Res 41(19):4349–4356
Seka MA, Kalogo S, Hammes F, Kielemoes J, Verstraete W (2001) Chlorine-susceptible and chlorine-resistant type 021N bacteria occurring in bulking activated sludges. Appl Environ Microbiol 67(11):5303–5307
Sezgin M, Jenkins D, Parker DS (1978) Unified theory of filamentous activated-sludge bulking. J Water Pollut Con F 50(2):362–381
Skow A, Mangold KA, Tajuddin M, Huntington A, Fritz B, Thomson RB, Kual KL (2005) Species-level identification of staphylococcal isolates by real-time PCR and melt curve analysis. J Clin Microbiol 43(6):2876–2880
Stewart MH, Olson BH (1992) Impact of growth-conditions on resistance of Klebsiella-Pneumoniae to chloramines. Appl Environ Microbiol 58(8):2649–2653
Subramaniam S (1998) The biology workbench—a seamless database and analysis environment for the biologist. Proteins 32(1):1–2
Thompson G, Forster C (2003) Bulking in activated sludge plants treating paper mill wastewaters. Water Res 37(11):2636–2644
Thompson G, Swain J, Kay M, Forster CF (2001) The treatment of pulp and paper mill effluent: a review. Bioresour Technol 77(3):275–286
Vervaeren H, De Wilde K, Matthys J, Boon N, Raskin L, Verstraete W (2005) Quantification of an Eikelboom type 021N bulking event with fluorescence in situ hybridization and real-time PCR. Appl Microbiol Biotechnol 68(5):695–704
Williams TM, Unz RF (1989) The nutrition of Thiothrix, type-021N, Beggiatoa and Leucothrix strains. Water Res 23(1):15–22
**ao M, Kong F, Sorrell TC, Cao Y, Lee OC, Liu Y, Sintchenko V, Chen SCA (2010) Identification of pathogenic Nocardia species by reverse line blot hybridization targeting the 16S rRNA and 16-23S rRNA gene spacer regions. J Clin Microbiol 48:503–511
Yu ZT, Mohn WW (1999) Killing two birds with one stone: simultaneous extraction of DNA and RNA from activated sludge biomass. Can J Microbiol 45(3):269–272
Acknowledgments
We are thankful to Irvine Ranch Water District for laboratory personnel and operators at Michelson Water Reclamation Plant for their assistance with data collection, sample processing, and chemical analysis. This research was supported by the National Water Research Institute (Award number no. 08-TM.004), CA and US National Science Foundation (Fellowship Award 1015730). We acknowledge Dr. Linda Tseng for her assistance with MANOVA analysis and Dr. Tom Wang for cloning T. eikelboomii PCR amplicons. We would like to also acknowledge Fabricio Berger de Vargas and Andre Borin Venturini for performing the melt-curve analysis for all environmental samples.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 352 kb)
Rights and permissions
About this article
Cite this article
Asvapathanagul, P., Olson, B.H., Gedalanga, P.B. et al. Identification and quantification of Thiothrix eikelboomii using qPCR for early detection of bulking incidents in a full-scale water reclamation plant. Appl Microbiol Biotechnol 99, 4045–4057 (2015). https://doi.org/10.1007/s00253-014-6230-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6230-5