Abstract
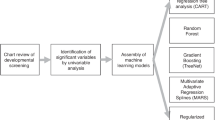
Prediction of outcomes following a prenatal diagnosis of congenital heart disease (CHD) is challenging. Machine learning (ML) algorithms may be used to reduce clinical uncertainty and improve prognostic accuracy. We performed a pilot study to train ML algorithms to predict postnatal outcomes based on clinical data. Specific objectives were to predict (1) in utero or neonatal death, (2) high-acuity neonatal care and (3) favorable outcomes. We included all fetuses with cardiac disease at Sunnybrook Health Sciences Centre, Toronto, Canada, from 2012 to 2021. Prediction models were created using the XgBoost algorithm (tree-based) with fivefold cross-validation. Among 211 cases of fetal cardiac disease, 61 were excluded (39 terminations, 21 lost to follow-up, 1 isolated arrhythmia), leaving a cohort of 150 fetuses. Fifteen (10%) demised (10 neonates) and 65 (48%) of live births required high acuity neonatal care. Of those with clinical follow-up, 60/87 (69%) had a favorable outcome. Prediction models for fetal or neonatal death, high acuity neonatal care and favorable outcome had AUCs of 0.76, 0.84 and 0.73, respectively. The most important predictors for death were the presence of non-cardiac abnormalities combined with more severe CHD. High acuity of postnatal care was predicted by anti Ro antibody and more severe CHD. Favorable outcome was most predicted by no right heart disease combined with genetic abnormalities, and maternal medications. Prediction models using ML provide good discrimination of key prenatal and postnatal outcomes among fetuses with congenital heart disease.


Similar content being viewed by others
Abbreviations
- CHD:
-
Congenital heart disease
- ML:
-
Machine learning
- CV:
-
Cross-validation
References
Oberije C et al (2014) A prospective study comparing the predictions of doctors versus models for treatment outcome of lung cancer patients: a step toward individualized care and shared decision making. Radiother Oncol 112(1):37–43
Hatch S (2017) Uncertainty in medicine. BMJ 357:j2180
Donofrio MT et al (2014) Diagnosis and treatment of fetal cardiac disease: a scientific statement from the American Heart Association. Circulation 129(21):2183–2242
Pinto NM et al (2020) Prenatal cardiac care: goals, priorities & gaps in knowledge in fetal cardiovascular disease: perspectives of the Fetal Heart Society. Prog Pediatr Cardiol 59:101312
Carvalho JS et al (2004) Clinical impact of first and early second trimester fetal echocardiography on high risk pregnancies. Heart 90(8):921–926
Yu D, Sui L, Zhang N (2020) Performance of first-trimester fetal echocardiography in diagnosing fetal heart defects: meta-analysis and systematic review. J Ultrasound Med 39(3):471–480
Morgan CT et al (2019) Improving prenatal diagnosis of coarctation of the aorta. Can J Cardiol 35(4):453–461
Freud LR et al (2014) Fetal aortic valvuloplasty for evolving hypoplastic left heart syndrome: postnatal outcomes of the first 100 patients. Circulation 130(8):638–645
MacColl CE et al (2012) Risk factors associated with in utero demise in fetuses with congenital heart disease: a case/control study. Cardiol Young 22:S74
Allan LD, Apfel HD, Printz BF (1998) Outcome after prenatal diagnosis of the hypoplastic left heart syndrome. Heart 79(4):371–373
Sharland GK, Chan KY, Allan LD (1994) Coarctation of the aorta: difficulties in prenatal diagnosis. Br Heart J 71(1):70–75
Weber RW et al (2013) Foetal echocardiographic assessment of borderline small left ventricles can predict the need for postnatal intervention. Cardiol Young 23(1):99–107
O’Brien SM et al (2009) An empirically based tool for analyzing mortality associated with congenital heart surgery. J Thorac Cardiovasc Surg 138(5):1139–1153
Brown DW et al (2011) Prenatal diagnosis of congenital heart disease and association with morbidity among survivors of initial palliation for single ventricle heart disease: analysis of the national pediatric cardiology quality improvement collaborative database. Circulation. https://doi.org/10.1161/circ.124.suppl_21.A16774
Bernard O et al (2018) Deep learning techniques for automatic MRI cardiac multi-structures segmentation and diagnosis: is the problem solved? IEEE Trans Med Imaging 37(11):2514–2525
LeCun Y, Bengio Y, Hinton G (2015) Deep learning. Nature 521(7553):436–444
Ouyang D et al (2020) Video-based AI for beat-to-beat assessment of cardiac function. Nature 580(7802):252–256
Arnaout R et al (2021) An ensemble of neural networks provides expert-level prenatal detection of complex congenital heart disease. Nat Med 27(5):882–891
Hoffman JI, Kaplan S (2002) The incidence of congenital heart disease. J Am Coll Cardiol 39(12):1890–1900
Fouad KM et al (2021) Advanced methods for missing values imputation based on similarity learning. PeerJ Comput Sci 7:e619
Chen T, Guestrin C (2016) XGBoost: a scalable tree boosting system. Proceedings of the 22nd ACM SIGKDD international conference on knowledge discovery and data mining. Association for Computing Machinery, San Francisco p, pp 785–794
Bergstra J, Bengio Y (2012) Random search for hyper-parameter optimization. J Mach Learn Res 13:281–305
Snoek J, Larochelle H, Adams RP (2012) Practical Bayesian optimization of machine learning algorithms. Adv Neural Info Process Syst 2012:2951–2959
Lundberg SM et al (2020) From local explanations to global understanding with explainable AI for trees. Nat Mach Intell 2(1):56–67
Athalye C, Arnaout R (2023) Domain-guided data augmentation for deep learning on medical imaging. PLoS ONE 18(3):e0282532
Athalye C et al (2023) Deep learning model for prenatal congenital heart disease (CHD) screening can be applied to retrospective imaging from the community setting, outperforming initial clinical detection in a well-annotated cohort. Ultrasound Obstet Gynecol 2023:10
Truong VT et al (2022) Application of machine learning in screening for congenital heart diseases using fetal echocardiography. Int J Cardiovasc Imaging 38:1007
Gardiner HM (2013) First-trimester fetal echocardiography: routine practice or research tool? Ultrasound Obstet Gynecol 42(6):611–612
Zidere V et al (2013) Comparison of echocardiographic findings in fetuses at less than 15 weeks’ gestation with later cardiac evaluation. Ultrasound Obstet Gynecol 42(6):679–686
MacColl CE et al (2014) Factors associated with in utero demise of fetuses that have underlying cardiac pathologies. Pediatr Cardiol 35(8):1403–1414
Jepson BM et al (2023) Pregnancy loss in major fetal congenital heart disease: incidence, risk factors and timing. Ultrasound Obstet Gynecol 62(1):75–87
Rudolph AM (2018) Circulatory changes during gestational development of the sheep and human fetus. Pediatr Res 84(3):348–351
Rudolph AM, Heymann MA (1968) The fetal circulation. Annu Rev Med 19:195–206
Sun L et al (2020) Understanding fetal hemodynamics using cardiovascular magnetic resonance imaging. Fetal Diagn Ther 47(5):354–362
Funding
This research study was not funded.
Author information
Authors and Affiliations
Contributions
LN, SR and CM wrote the main manuscript text. CM, BC and AI performed the ML analysis and prepared the Tables and Figures. KM performed the data collection with the aid of LF, TM, ON, NM, DW helped write the manuscript and were instrumental in the original REB study proposal. All authors have reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A
Appendix A
Hoffman Criteria for defining severity of congenital heart disease [19].
Severe Congenital Heart Disease
-
A.
All those with cyanotic heart disease
-
a.
d-transposition of the great arteries
-
b.
Tetralogy of Fallot, including pulmonary atresia and absent pulmonary valve
-
c.
Hypoplastic right heart
-
i.
Tricuspid atresia
-
ii.
Pulmonary atresia with an intact ventricular septum
-
iii.
Ebstein anomaly
-
i.
-
d.
Hypoplastic left heart
-
i.
Aortic atresia
-
ii.
Mitral atresia
-
i.
-
e.
SV (single ventricle)
-
f.
DORV (double outlet right ventricle)
-
g.
Truncus arteriosus
-
h.
Total anomalous pulmonary venous connection
-
i.
Critical pulmonary stenosis
-
j.
Miscellaneous uncommon lesions such as double outlet left ventricle, malpositions and some forms of l-transposition of the great arteries (congenitally corrected transposition)
-
a.
-
B.
Acyanotic lesions
-
a.
Atrioventricular septal defect (AVSD)
-
b.
Large ventricular septal defect (VSD)
-
c.
Large patent ductus arteriosus (PDA)
-
d.
Critical or severe aortic stenosis
-
e.
Severe pulmonary stenosis
-
f.
Critical coarctation of the aorta
-
a.
Moderate Congenital Heart Disease
-
A.
Mild or moderate aortic stenosis or aortic incompetence
-
B.
Moderate pulmonary stenosis or incompetence
-
C.
Non critical coarctation of the aorta
-
D.
Large ASD
-
E.
Complex forms of VSD
Mild Congenital Heart Disease
-
A.
Small VSD
-
B.
Small PDA
-
C.
Mild pulmonary stenosis
-
D.
Bicuspid aortic valve without aortic stenosis or incompetence
-
E.
Small or spontaneously closed ASD
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nield, L.E., Manlhiot, C., Magor, K. et al. Machine Learning to Predict Outcomes of Fetal Cardiac Disease: A Pilot Study. Pediatr Cardiol (2024). https://doi.org/10.1007/s00246-024-03512-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00246-024-03512-x



