Abstract
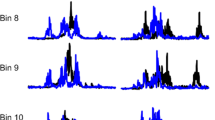
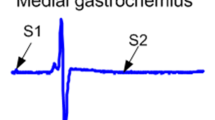
Neurorecovery from locomotor training is well established in human spinal cord injury (SCI). However, neurorecovery resulting from combined interventions has not been widely studied. In this randomized clinical trial, we established the tibialis anterior (TA) flexion reflex modulation pattern when transcranial magnetic stimulation (TMS) of the primary motor cortex was paired with transcutaneous spinal cord (transspinal) stimulation over the thoracolumbar region during assisted step training. Single pulses of TMS were delivered either before (TMS-transspinal) or after (transspinal-TMS) transspinal stimulation during the stance phase of the less impaired leg. Eight individuals with chronic incomplete or complete SCI received at least 20 sessions of paired stimulation during assisted step training. Each session consisted of 240 paired stimuli delivered over 10-min blocks for 1 h during robotic-assisted step training with the Lokomat6 Pro®. Body weight support, leg guidance force and treadmill speed were adjusted based on each participant’s ability to step without knee buckling or toe dragging. Both the early and late TA flexion reflex remained unaltered after TMS-transspinal and locomotor training. In contrast, the early and late TA flexion reflexes were significantly depressed during step** after transspinal-TMS and locomotor training. Reflex changes occurred at similar slopes and intercepts before and after training. Our findings support that targeted brain and spinal cord stimulation coupled with locomotor training reorganizes the function of flexion reflex pathways, which are a part of locomotor networks, in humans with varying levels of sensorimotor function after SCI.
Trial registration number NCT04624607; Registered on November 12, 2020.




Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
References
Andén N-E, Jukes MGM, Lundberg A, Vyklicky L (1966) The effect of DOPA on the spinal cord. 1. Influence on transmission from primary aflerents. Acta Physiol Scand 67:373–386. https://doi.org/10.1111/j.1748-1716.1966.tb03324.x
Barbeau H (2003) Locomotor training in neurorehabilitation: emerging rehabilitation concepts. Neurorehabil Neural Repair 17:3–11. https://doi.org/10.1177/0888439002250442
Berardelli A, Inghilleri M, Cruccu G, Manfredi M (1990) Descending volley after electrical and magnetic transcranial stimulation in man. Neurosci Lett 112:54–58. https://doi.org/10.1016/0304-3940(90)90321-Y
Berić A, Dimitrijević MR, Sharkey PC, Sherwood AM (1986) Cortical potentials evoked by epidural stimulation of the cervical and thoracic spinal cord in man. Electroencephalogr Clin Neurophysiol 65:102–110. https://doi.org/10.1016/0168-5597(86)90042-0
Brunhölzl C, Claus D (1994) Central motor conduction time to upper and lower limbs in cervical cord lesions. Arch Neurol 51:245–249. https://doi.org/10.1001/ARCHNEUR.1994.00540150039013
Bunday KL, Perez MA (2012) Motor recovery after spinal cord injury enhanced by strengthening corticospinal synaptic transmission. Curr Biol 22:2355–2361. https://doi.org/10.1016/J.CUB.2012.10.046
Bunday KL, Urbin MA, Perez MA (2018) Potentiating paired corticospinal-motoneuronal plasticity after spinal cord injury. Brain Stimul. https://doi.org/10.1016/J.BRS.2018.05.006
Buonomano DV, Merzenich MM (1998) Cortical plasticity: from synapses to maps. Annu Rev Neurosci 21:149–186. https://doi.org/10.1146/annurev.neuro.21.1.149
Cortes M, Thickbroom GW, Valls-Sole J et al (2011) Spinal associative stimulation: a non-invasive stimulation paradigm to modulate spinal excitability. Clin Neurophysiol 122:2254–2259. https://doi.org/10.1016/j.clinph.2011.02.038
Côté MP, Murray LM, Knikou M (2018) Spinal control of locomotion: individual neurons, their circuits and functions. Front Physiol 9:784. https://doi.org/10.3389/fphys.2018.00784
Courtine G, Harkema SJ, Dy CJ et al (2007) Modulation of multisegmental monosynaptic responses in a variety of leg muscles during walking and running in humans. J Physiol 582:1125–1139. https://doi.org/10.1113/jphysiol.2007.128447
Dan Y, Poo M-M (2006) Spike timing-dependent plasticity: from synapse to perception. Physiol Rev 86:1033–1048. https://doi.org/10.1152/physrev.00030.2005
Dietz V, Grillner S, Trepp A et al (2009) Changes in spinal reflex and locomotor activity after a complete spinal cord injury: a common mechanism? Brain 132:2196–2205. https://doi.org/10.1093/BRAIN/AWP124
Dixon L, Ibrahim MM, Santora D, Knikou M (2016) Paired associative transspinal and transcortical stimulation produces plasticity in human cortical and spinal neuronal circuits. J Neurophysiol 116:904–916. https://doi.org/10.1152/jn.00259.2016
Duysens J, Tax AAM, Trippel M, Dietz V (1993) Increased amplitude of cutaneous reflexes during human running as compared to standing. Brain Res 613:230–238. https://doi.org/10.1016/0006-8993(93)90903-Z
Eccles JC, Kostyuk PG, Schmidt RF (1962) Presynaptic inhibition of the central actions of flexor reflex afferents. J Physiol 161:258–281. https://doi.org/10.1113/jphysiol.1962.sp006885
Edgerton VR, Tillakaratne NJK, Bigbee AJ et al (2004) Plasticity of the spinal neural circuitry after injury. Annu Rev Neurosci 27:145–167. https://doi.org/10.1146/annurev.neuro.27.070203.144308
Edgley SA, Eyre JA, Lemon RN, Miller S (1990) Excitation of the corticospinal tract by electromagnetic and electrical stimulation of the scalp in the macaque monkey. J Physiol 425:301. https://doi.org/10.1113/JPHYSIOL.1990.SP018104
Ertekin C, Sarica Y, Üçkardeşler L (1984) Somatosensory cerebral potentials evoked by stimulation of the lumbo-sacral spinal cord in normal subjects and in patients with conus medullaris and cauda equina lesions. Electroencephalogr Clin Neurophysiol 59:57–66. https://doi.org/10.1016/0168-5597(84)90020-0
Hebb DO (1949) The organization of behavior: a neuropsychological theory. Wiley, New York
Jankowska E, Jukes MGM, Lund S, Lundberg A (1967a) The effect of DOPA on the spinal cord 6. Half-centre organization of interneurones transmitting effects from the flexor reflex afferents. Acta Physiol Scand 70:389–402. https://doi.org/10.1111/j.1748-1716.1967.tb03637.x
Jankowska E, Jukes MGM, Lund S, Lundberg A (1967b) The effect of DOPA on the spinal cord 5. Reciprocal organization of pathways transmitting excitatory action to alpha motoneurones of flexors and extensors. Acta Physiol Scand 70:369–388. https://doi.org/10.1111/j.1748-1716.1967.tb03636.x
Jayaram G, Stinear JW (2007) Contralesional paired associative stimulation increases paretic lower limb motor excitability post-stroke. Exp Brain Res 185:563–570. https://doi.org/10.1007/S00221-007-1183-X
Jo HJ, Perez MA (2020) Corticospinal-motor neuronal plasticity promotes exercise-mediated recovery in humans with spinal cord injury—pubMed. Brain 143:1356–1382
Knikou M (2010a) Neural control of locomotion and training-induced plasticity after spinal and cerebral lesions. Clin Neurophysiol 121:1655–1668. https://doi.org/10.1016/j.clinph.2010.01.039
Knikou M (2010b) Plantar cutaneous afferents normalize the reflex modulation patterns during step** in chronic human spinal cord injury. J Neurophysiol 103:1304–1314. https://doi.org/10.1152/jn.00880.2009
Knikou M (2013a) Neurophysiological characterization of transpinal evoked potentials in human leg muscles. Bioelectromagnetics 34:630–640. https://doi.org/10.1002/bem.21808
Knikou M (2013b) Neurophysiological characteristics of human leg muscle action potentials evoked by transcutaneous magnetic stimulation of the spine. Bioelectromagnetics 34:200–210. https://doi.org/10.1002/bem.21768
Knikou M (2013c) Functional reorganization of soleus H-reflex modulation during step** after robotic-assisted step training in people with complete and incomplete spinal cord injury. Exp Brain Res 228:279–296. https://doi.org/10.1007/s00221-013-3560-y
Knikou M (2014) Transpinal and transcortical stimulation alter corticospinal excitability and increase spinal output. PLoS One. https://doi.org/10.1371/journal.pone.0102313
Knikou M (2017) Spinal excitability changes after transspinal and transcortical paired associative stimulation in humans. Neural Plast. https://doi.org/10.1155/2017/6751810
Knikou M, Conway BA (2005) Effects of electrically induced muscle contraction on flexion reflex in human spinal cord injury. Spinal Cord 43:640–648. https://doi.org/10.1038/sj.sc.3101772
Knikou M, Murray LM (2019) Repeated transspinal stimulation decreases soleus H-reflex excitability and restores spinal inhibition in human spinal cord injury. PLoS One 14:e0223135. https://doi.org/10.1371/journal.pone.0223135
Knikou M, Angeli CA, Ferreira CK, Harkema SJ (2009) Flexion reflex modulation during step** in human spinal cord injury. Exp Brain Res 196:341–351. https://doi.org/10.1007/s00221-009-1854-x
Lamy JC, Russmann H, Shamim EA et al (2010) Paired associative stimulation induces change in presynaptic inhibition of Ia terminals in wrist flexors in humans. J Neurophysiol 104:755–764. https://doi.org/10.1152/jn.00761.2009
Mackey AS, Uttaro D, McDonough MP et al (2016) Convergence of flexor reflex and corticospinal inputs on tibialis anterior network in humans. Clin Neurophysiol 127:706–715. https://doi.org/10.1016/J.CLINPH.2015.06.011
Markram H, Lübke J, Frotscher M, Sakmann B (1997) Regulation of synaptic efficacy by coincidence of postsynaptic APs and EPSPs the increase in. Science 80(275):213–214. https://doi.org/10.1126/science.275.5297.213
Markram H, Gerstner W, Sjöström PJ (2011) A history of spike-timing-dependent plasticity. Front Synaptic Neurosci 3:1–24
McPherson JG, Miller RR, Perlmutter SI (2015) Targeted, activity-dependent spinal stimulation produces long-lasting motor recovery in chronic cervical spinal cord injury. Proc Natl Acad Sci USA 112:12193–12198. https://doi.org/10.1073/pnas.1505383112
Ménard A, Leblond H, Gossard J-P (2002) Sensory integration in presynaptic inhibitory pathways during fictive locomotion in the cat. J Neurophysiol 88:163–171. https://doi.org/10.1152/jn.2002.88.1.163
Ménard A, Leblond H, Gossard JP (2003) Modulation of monosynaptic transmission by presynaptic inhibition during fictive locomotion in the cat. Brain Res 964:67–82. https://doi.org/10.1016/S0006-8993(02)04067-2
Meunier S, Russmann H, Simonetta-Moreau M, Hallett M (2007) Changes in spinal excitability after PAS. J Neurophysiol 97:3131–3135. https://doi.org/10.1152/jn.01086.2006
Minassian K, Persy I, Rattay F et al (2007) Posterior root–muscle reflexes elicited by transcutaneous stimulation of the human lumbosacral cord. Muscle Nerve 35:327–336. https://doi.org/10.1002/mus.20700
Müller-Dahlhaus F, Ziemann U, Classen J (2010) Plasticity resembling spike-timing dependent synaptic plasticity: the evidence in human cortex. Front Synaptic Neurosci. https://doi.org/10.3389/fnsyn.2010.00034
Murray LM, Knikou M (2019a) Transspinal stimulation increases motoneuron output of multiple segments in human spinal cord injury. PLoS One. https://doi.org/10.1371/journal.pone.0213696
Murray LM, Knikou M (2019b) Repeated cathodal transspinal pulse and direct current stimulation modulate cortical and corticospinal excitability differently in healthy humans. Exp Brain Res 237:1841–1852. https://doi.org/10.1007/s00221-019-05559-2
Murray LM, Islam MA, Knikou M (2019) Cortical and subcortical contributions to neuroplasticity after repetitive transspinal stimulation in humans. Neural Plast. https://doi.org/10.1155/2019/4750768
Paradiso C, De Vito L, Rossi S et al (1995) Cervical and scalp recorded short latency somatosensory evoked potentials in response to epidural spinal cord stimulation in patients with peripheral vascular disease. Electroencephalogr Clin Neurophysiol 96:105–113
Pulverenti TS, Anamul Islam M, Alsalman O et al (2019) Transspinal stimulation decreases corticospinal excitability and alters the function of spinal locomotor networks. J Neurophysiol 122:2331–2343. https://doi.org/10.1152/jn.00554.2019
Pulverenti TS, Zaaya M, Grabowski M et al (2021) Neurophysiological changes after paired brain and spinal cord stimulation coupled with locomotor training in human spinal cord injury. Front Neurol 12:627975. https://doi.org/10.3389/fneur.2021.627975
Roby-Brami A, Bussel B (1987) Long-latency spinal reflex in man after flexor reflex afferent stimulation. Brain 110:707–725. https://doi.org/10.1093/brain/110.3.707
Roby-Brami A, Bussel B (1990) Effects of flexor reflex afferent stimulation on the soleus H reflex in patients with a complete spinal cord lesion: evidence for presynaptic inhibition of Ia transmission. Exp Brain Res 81:593–601. https://doi.org/10.1007/BF02423509
Roby-Brami A, Bussel B (1992) Inhibitory effects on flexor reflexes in patients with a complete spinal cord lesion. Exp Brain Res 90:201–208. https://doi.org/10.1007/BF00229272
Rosenkranz K, Williamon A, Rothwell JC (2007) Motorcortical excitability and synaptic plasticity is enhanced in professional musicians. J Neurosci 27:5200–5206. https://doi.org/10.1523/JNEUROSCI.0836-07.2007
Rossignol S, Frigon A (2011) Recovery of locomotion after spinal cord injury: some facts and mechanisms. Annu Rev Neurosci 34:413–440. https://doi.org/10.1146/annurev-neuro-061010-113746
Rossini PM, Burke D, Chen R et al (2015) Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N Committee. Clin Neurophysiol 126:1071–1107. https://doi.org/10.1016/j.clinph.2015.02.001
Rothwell J, Thompson P, Day B et al (1991) Stimulation of the human motor cortex through the scalp. Exp Physiol 76:159–200. https://doi.org/10.1113/EXPPHYSIOL.1991.SP003485
Roy FD, Bosgra D, Stein RB (2014) Interaction of transcutaneous spinal stimulation and transcranial magnetic stimulation in human leg muscles. Exp Brain Res 232:1717–1728. https://doi.org/10.1007/s00221-014-3864-6
Schomburg ED, Petersen N, Barajon I, Hultborn H (1998) Flexor reflex afferents reset the step cycle during fictive locomotion in the cat. Exp Brain Res 122:339–350. https://doi.org/10.1007/s002210050522
Sjöström PJ, Rancz EA, Roth A, Hausser M (2008) Dendritic excitability and synaptic plasticity. Physiol Rev 88:769–840. https://doi.org/10.1152/physrev.00016.2007
Smith AC, Knikou M (2016) A review on locomotor training after spinal cord injury: reorganization of spinal neuronal circuits and recovery of motor function. Neural Plast. https://doi.org/10.1155/2016/1216258
Smith AC, Mummidisetty CK, Rymer WZ, Knikou M (2014) Locomotor training alters the behavior of flexor reflexes during walking in human spinal cord injury. J Neurophysiol 112:2164–2175. https://doi.org/10.1152/jn.00308.2014.-In
Smith AC, Rymer WZ, Knikou M (2015) Locomotor training modifies soleus monosynaptic motoneuron responses in human spinal cord injury. Exp Brain Res 233:89–103. https://doi.org/10.1007/s00221-014-4094-7
Song S, Miller KD, Abbott LF (2000) Competitive Hebbian learning through spike-timing-dependent synaptic plasticity. Nat Neurosci 3:919–926. https://doi.org/10.1038/78829
Stefan K, Kunesch E, Cohen LG et al (2000) Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123:572–584
Stefan K, Kunesch E, Benecke R et al (2002) Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J Physiol 543:699–708. https://doi.org/10.1113/jphysiol.2002.023317
Tax AAM, Van Wezel BMH, Dietz V (1995) Bipedal reflex coordination to tactile stimulation of the sural nerve during human running. J Neurophysiol 73:1947–1964. https://doi.org/10.1152/jn.1995.73.5.1947
Taylor JL, Martin PG (2009) Voluntary motor output is altered by spike-timing-dependent changes in the human corticospinal pathway. J Neurosci 29:11708–11716. https://doi.org/10.1523/JNEUROSCI.2217-09.2009
Van Wezel BMH, Ottenhoff FAM, Duysens J (1997) Dynamic control of location-specific information in tactile cutaneous reflexes from the foot during human walking. J Neurosci 17:3804–3814. https://doi.org/10.1523/jneurosci.17-10-03804.1997
Zaaya M, Pulverenti TS, Islam MA, Knikou M (2020) Transspinal stimulation downregulates activity of flexor locomotor networks during walking in humans. J Electromyogr Kinesiol 52:102420. https://doi.org/10.1016/j.jelekin.2020.102420
Zaaya M, Pulverenti TS, Knikou M (2021) Transspinal stimulation and step training alter function of spinal networks in complete spinal cord injury. Spinal Cord Ser Cases 71(7):1–8. https://doi.org/10.1038/s41394-021-00421-6
Zehr EP, Komiyama T, Stein RB (1997) Cutaneous reflexes during human gait: electromyographic and kinematic responses to electrical stimulation. J Neurophysiol 77:3311–3325. https://doi.org/10.1152/jn.1997.77.6.3311
Zehr EP, Stein RB, Komiyama T (1998) Function of sural nerve reflexes during human walking. J Physiol 507:305–314. https://doi.org/10.1111/j.1469-7793.1998.305bu.x
Ziemann U, Muellbacher W, Hallett M, Cohen LG (2001) Modulation of practice-dependent plasticity in human motor cortex. Brain 124:1171–1181. https://doi.org/10.1093/brain/124.6.1171
Acknowledgements
We thank all participants and their care givers for their commitment during participation in the research study.
Funding
This study was supported by the Spinal Cord Injury Research Board (SCIRB) of the New York State Department of Health (NYSDOH), Wadsworth Center (Grants C32095GG, C33276GG), and, in part, by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), National Institutes of Health (NIH), under grant number R01HD100544 awarded to Maria Knikou. Funding sources were not involved in study design, data collection, data analysis, data interpretation, or decision to publish.
Author information
Authors and Affiliations
Contributions
MK: conception and design of research, interpreted results of experiments and wrote first draft of manuscript. TP, MZ, MK: performed experiments. MZ: analyzed data, prepared figures. TP, MZ and MK: edited and revised manuscript. TP, MZ and MK: approved final version of manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Compliance with ethical standards
The study was approved by the Institutional Review Board of the City University of New York (IRB no. 2017-0261). All participants gave informed consent before study enrollment and participation.
Additional information
Communicated by Francesco Lacquaniti.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pulverenti, T.S., Zaaya, M. & Knikou, M. Brain and spinal cord paired stimulation coupled with locomotor training affects polysynaptic flexion reflex circuits in human spinal cord injury. Exp Brain Res 240, 1687–1699 (2022). https://doi.org/10.1007/s00221-022-06375-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-022-06375-x