Abstract
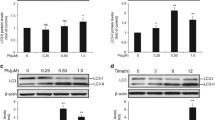
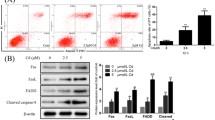
The renal proximal tubule (PT) is the major target of cadmium (Cd2+) toxicity where Cd2+ causes stress and apoptosis. Autophagy is induced by cell stress, e.g., endoplasmic reticulum (ER) stress, and may contribute to cell survival or death. The role of autophagy in Cd2+-induced nephrotoxicity remains unsettled due to contradictory results and lack of evidence for autophagic machinery damage by Cd2+. Cd2+-induced autophagy in rat kidney PT cell line NRK-52E and its role in cell death was investigated. Increased LC3-II and decreased p62 as autophagy markers indicate rapid induction of autophagic flux by Cd2+ (5–10 µM) after 1 h, accompanied by ER stress (increased p-PERK, p-eIF2α, CHOP). Cd2+ exposure exceeding 3 h results in p62/LC3-II accumulation, but diminished effect of lysosomal inhibitors (bafilomycin A1, pepstatin A +E-64d) on p62/LC3-II levels, indicating decreased autophagic flux and cargo degradation. At 24 h exposure, Cd2+ (5–25 µM) activates intrinsic apoptotic pathways (Bax/Bcl-2, PARP-1), which is not evident earlier (≤6 h) although cell viability by MTT assay is decreased. Autophagy inducer rapamycin (100 nM) does not overcome autophagy inhibition or Cd2+-induced cell viability loss. The autophagosome–lysosome fusion inhibitor liensinine (5 μM) increases CHOP and Bax/Bcl-2-dependent apoptosis by low Cd2+ stress, but not by high Cd2+. Lysosomal instability by Cd2+ (5 μM; 6 h) is indicated by increases in cellular sphingomyelin and membrane fluidity and decreases in cathepsins and LAMP1. The data suggest dual and temporal impact of Cd2+ on autophagy: Low Cd2+ stress rapidly activates autophagy counteracting damage but Cd2+ stress accrual disrupts autophagic flux and lysosomal stability, possibly resulting in lysosomal cell death.









Similar content being viewed by others
References
Abouhamed M, Gburek J, Liu W et al (2006) Divalent metal transporter 1 in the kidney proximal tubule is expressed in late endosomes/lysosomal membranes: implications for renal handling of protein-metal complexes. Am J Physiol Renal Physiol 290(6):F1525–F1533
Aits S, Jaattela M (2013) Lysosomal cell death at a glance. J Cell Sci 126(Pt 9):1905–1912. doi:10.1242/jcs.091181
Akesson A, Lundh T, Vahter M et al (2005) Tubular and glomerular kidney effects in Swedish women with low environmental cadmium exposure. Environ Health Perspect 113(11):1627–1631
Aki T, Funakoshi T, Unuma K, Uemura K (2013) Impairment of autophagy: from hereditary disorder to drug intoxication. Toxicology 311(3):205–215. doi:10.1016/j.tox.2013.07.001
Amoruso MA, Witz G, Goldstein BD (1987) Alteration of erythrocyte membrane fluidity by heavy metal cations. Toxicol Ind Health 3(1):135–144
Biagioli M, Pifferi S, Ragghianti M, Bucci S, Rizzuto R, Pinton P (2008) Endoplasmic reticulum stress and alteration in calcium homeostasis are involved in cadmium-induced apoptosis. Cell Calcium 43(2):184–195
Boukh-Viner T, Guo T, Alexandrian A et al (2005) Dynamic ergosterol- and ceramide-rich domains in the peroxisomal membrane serve as an organizing platform for peroxisome fusion. J Cell Biol 168(5):761–773. doi:10.1083/jcb.200409045
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chargui A, Zekri S, Jacquillet G et al (2011) Cadmium-induced autophagy in rat kidney: an early biomarker of subtoxic exposure. Toxicol Sci 121(1):31–42 doi:10.1093/toxsci/kfr031
Deegan S, Saveljeva S, Gorman AM, Samali A (2013) Stress-induced self-cannibalism: on the regulation of autophagy by endoplasmic reticulum stress. Cell Mol Life Sci 70(14):2425–2441. doi:10.1007/s00018-012-1173-4
Ding WX, Ni HM, Gao W et al (2007) Linking of autophagy to ubiquitin-proteasome system is important for the regulation of endoplasmic reticulum stress and cell viability. Am J Pathol 171(2):513–524. doi:10.2353/ajpath.2007.070188
Dong T, Liao D, Liu X, Lei X (2015) Using small molecules to dissect non-apoptotic programmed cell death: necroptosis, ferroptosis, and pyroptosis. Chembiochem 16(18):2557–2561 doi:10.1002/cbic.201500422
Freisinger E, Vasak M (2013) Cadmium in metallothioneins. Met Ions Life Sci 11:339–371. doi:10.1007/978-94-007-5179-8_11
Fuchs Y, Steller H (2015) Live to die another way: modes of programmed cell death and the signals emanating from dying cells. Nat Rev Mol Cell Biol 16(6):329–344. doi:10.1038/nrm3999
Fujiki K, Inamura H, Matsuoka M (2014) PI3K signaling mediates diverse regulation of ATF4 expression for the survival of HK-2 cells exposed to cadmium. Arch Toxicol 88(2):403–414. doi:10.1007/s00204-013-1129-y
Galluzzi L, Bravo-San Pedro JM, Kepp O, Kroemer G (2016) Regulated cell death and adaptive stress responses. Cell Mol Life Sci 73(11–12):2405–2410. doi:10.1007/s00018-016-2209-y
Haswell-Elkins M, Satarug S, O’Rourke P et al (2008) Striking association between urinary cadmium level and albuminuria among Torres Strait Islander people with diabetes. Environ Res 106(3):379–383. doi:10.1016/j.envres.2007.10.004
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13(2):89–102. doi:10.1038/nrm3270
Hiramatsu N, Kasai A, Du S et al (2007) Rapid, transient induction of ER stress in the liver and kidney after acute exposure to heavy metal: evidence from transgenic sensor mice. FEBS Lett 581(10):2055–2059. doi:10.1016/j.febslet.2007.04.040
Holtzman E (2013) Lysosomes: a survey. Springer, New York
Hurwitz R, Ferlinz K, Sandhoff K (1994) The tricyclic antidepressant desipramine causes proteolytic degradation of lysosomal sphingomyelinase in human fibroblasts. Biol Chem Hoppe Seyler 375(7):447–450
Ishitsuka R, Yamaji-Hasegawa A, Makino A, Hirabayashi Y, Kobayashi T (2004) A lipid-specific toxin reveals heterogeneity of sphingomyelin-containing membranes. Biophys J 86(1 Pt 1):296–307 doi:10.1016/S0006-3495(04)74105-3
Jarup L, Berglund M, Elinder CG, Nordberg G, Vahter M (1998) Health effects of cadmium exposure–a review of the literature and a risk estimate. Scand J Work Environ Health 24(Suppl 1):1–51
Jung CH, Ro SH, Cao J, Otto NM, Kim DH (2010) mTOR regulation of autophagy. FEBS Lett 584(7):1287–1295. doi:10.1016/j.febslet.2010.01.017
Kabeya Y, Mizushima N, Yamamoto A, Oshitani-Okamoto S, Ohsumi Y, Yoshimori T (2004) LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci 117(Pt 13):2805–2812. doi:10.1242/jcs.01131
Kato H, Katoh R, Kitamura M (2013) Dual regulation of cadmium-induced apoptosis by mTORC1 through selective induction of IRE1 branches in unfolded protein response. PLoS One 8(5):e64344. doi:10.1371/journal.pone.0064344
Klionsky DJ, Elazar Z, Seglen PO, Rubinsztein DC (2008) Does bafilomycin A1 block the fusion of autophagosomes with lysosomes? Autophagy 4(7):849–850
Klionsky DJ, Abdalla FC, Abeliovich H et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4):445–544
Kolzer M, Werth N, Sandhoff K (2004) Interactions of acid sphingomyelinase and lipid bilayers in the presence of the tricyclic antidepressant desipramine. FEBS Lett 559(1–3):96–98. doi:10.1016/S0014-5793(04)00033-X
Komoike Y, Inamura H, Matsuoka M (2012) Effects of salubrinal on cadmium-induced apoptosis in HK-2 human renal proximal tubular cells. Arch Toxicol 86(1):37–44. doi:10.1007/s00204-011-0742-x
Lee WK, Bork U, Gholamrezaei F, Thévenod F (2005) Cd2+-induced cytochrome c release in apoptotic proximal tubule cells: role of mitochondrial permeability transition pore and Ca2+ uniporter. Am J Physiol Renal Physiol 288(1):F27–F39
Lee WK, Abouhamed M, Thévenod F (2006) Caspase-dependent and -independent pathways for cadmium-induced apoptosis in cultured kidney proximal tubule cells. Am J Physiol Renal Physiol 291:F823–F832
Lee WK, Torchalski B, Thévenod F (2007) Cadmium-induced ceramide formation triggers calpain-dependent apoptosis in cultured kidney proximal tubule cells. Am J Physiol Cell Physiol 293(3):C839–C847
Lee WK, Braun M, Langeluddecke C, Thévenod F (2012a) Cyclosporin a, but not FK506, induces osmotic lysis of pancreas zymogen granules, intra-acinar enzyme release, and lysosome instability by activating K+ channel. Pancreas 41(4):596–604. doi:10.1097/MPA.0b013e318239c6e5
Lee WK, Chakraborty PK, Roussa E, Wolff NA, Thévenod F (2012b) ERK1/2-dependent bestrophin-3 expression prevents ER-stress-induced cell death in renal epithelial cells by reducing CHOP. Biochim Biophys Acta 1823(10):1864–1876. doi:10.1016/j.bbamcr.2012.06.003
Li J, Kim SG, Blenis J (2014) Rapamycin: one drug, many effects. Cell Metab 19(3):373–379. doi:10.1016/j.cmet.2014.01.001
Li M, Pi H, Yang Z et al (2016) Melatonin antagonizes cadmium-induced neurotoxicity by activating the transcription factor EB -dependent autophagy–lysosome machinery in mouse neuroblastoma cells. J Pineal Res 61(3):353–369. doi:10.1111/jpi.12353
Lieberthal W, Levine JS (2012) Mammalian target of rapamycin and the kidney. I. The signaling pathway. Am J Physiol Renal Physiol 303(1):F1–F10. doi:10.1152/ajprenal.00014.2012
Linke T, Wilkening G, Lansmann S et al (2001) Stimulation of acid sphingomyelinase activity by lysosomal lipids and sphingolipid activator proteins. Biol Chem 382(2):283–290. doi:10.1515/BC.2001.035
Liu F, Inageda K, Nishitai G, Matsuoka M (2006) Cadmium induces the expression of Grp78, an endoplasmic reticulum molecular chaperone, in LLC-PK1 renal epithelial cells. Environ Health Perspect 114(6):859–864
Loos B, du Toit A, Hofmeyr JH (2014) Defining and measuring autophagosome flux-concept and reality. Autophagy 10(11):2087–2096. doi:10.4161/15548627.2014.973338
Lowry O, Rosebrough N, Farr A, Randall RJ (1951) Protein measurements with the folin phenol reagent. J Biol Chem 194:265–275
Luo B, Lin Y, Jiang S et al (2016) Endoplasmic reticulum stress eIF2alpha-ATF4 pathway-mediated cyclooxygenase-2 induction regulates cadmium-induced autophagy in kidney. Cell Death Dis 7(6):e2251. doi:10.1038/cddis.2016.78
Messner B, Turkcan A, Ploner C, Laufer G, Bernhard D (2016) Cadmium overkill: autophagy, apoptosis and necrosis signalling in endothelial cells exposed to cadmium. Cell Mol Life Sci 73(8):1699–1713. doi:10.1007/s00018-015-2094-9
Mizushima N (2007) Autophagy: process and function. Genes Dev 21(22):2861–2873. doi:10.1101/gad.1599207
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147(4):728–741. doi:10.1016/j.cell.2011.10.026
Mizushima N, Yoshimori T (2007) How to interpret LC3 immunoblotting. Autophagy 3(6):542–545
Nair AR, Lee WK, Smeets K et al (2015) Glutathione and mitochondria determine acute defense responses and adaptive processes in cadmium-induced oxidative stress and toxicity of the kidney. Arch Toxicol 89(12):2273–2289. doi:10.1007/s00204-014-1401-9
Nawrot TS, Staessen JA, Roels HA et al (2010) Cadmium exposure in the population: from health risks to strategies of prevention. Biometals 23(5):769–782. doi:10.1007/s10534-010-9343-z
Pankiv S, Clausen TH, Lamark T et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282(33):24131–24145. doi:10.1074/jbc.M702824200
Parasassi T, Krasnowska EK, Bagatolli L, Gratton E (1998) Laurdan and prodan as polarity sensitive fluorescent membrane probes. J Fluoresc 8:365–373
Payliss BJ, Hassanin M, Prenner EJ (2015) The structural and functional effects of Hg(II) and Cd(II) on lipid model systems and human erythrocytes: a review. Chem Phys Lipids 193:36–51. doi:10.1016/j.chemphyslip.2015.09.009
Periasamy N, Winter R (2006) The effects of temperature, pressure and peptide incorporation on ternary model raft mixtures—a laurdan fluorescence spectroscopy study. Biochim Biophys Acta 1764(3):398–404. doi:10.1016/j.bbapap.2005.10.013
Perrotta C, Cervia D, De Palma C et al (2015) The emerging role of acid sphingomyelinase in autophagy. Apoptosis 20(5):635–644. doi:10.1007/s10495-015-1101-9
Petersen NH, Olsen OD, Groth-Pedersen L et al (2013) Transformation-associated changes in sphingolipid metabolism sensitize cells to lysosomal cell death induced by inhibitors of acid sphingomyelinase. Cancer Cell 24(3):379–393. doi:10.1016/j.ccr.2013.08.003
Prenner E, Honsek G, Honig D, Mobius D, Lohner K (2007) Imaging of the domain organization in sphingomyelin and phosphatidylcholine monolayers. Chem Phys Lipids 145(2):106–118. doi:10.1016/j.chemphyslip.2006.11.002
Prozialeck WC, Edwards JR (2012) Mechanisms of cadmium-induced proximal tubule injury: new insights with implications for biomonitoring and therapeutic interventions. J Pharmacol Exp Ther 343(1):2–12. doi:10.1124/jpet.110.166769
Ramstedt B, Slotte JP (2006) Sphingolipids and the formation of sterol-enriched ordered membrane domains. Biochim Biophys Acta 1758(12):1945–1956. doi:10.1016/j.bbamem.2006.05.020
Rogasevskaia T, Coorssen JR (2006) Sphingomyelin-enriched microdomains define the efficiency of native Ca(2+)-triggered membrane fusion. J Cell Sci 119(Pt 13):2688–2694. doi:10.1242/jcs.03007
Samali A, Fitzgerald U, Deegan S, Gupta S (2010) Methods for monitoring endoplasmic reticulum stress and the unfolded protein response. Int J Cell Biol 2010:830307. doi:10.1155/2010/830307
Suwalsky M, Villena F, Norris B, Cuevas F, Sotomayor CP (2004) Cadmium-induced changes in the membrane of human erythrocytes and molecular models. J Inorg Biochem 98(6):1061–1066. doi:10.1016/j.**orgbio.2004.02.027
Tait SW, Ichim G, Green DR (2014) Die another way–non-apoptotic mechanisms of cell death. J Cell Sci 127(Pt 10):2135–2144. doi:10.1242/jcs.093575
Templeton DM, Liu Y (2010) Multiple roles of cadmium in cell death and survival. Chem Biol Interact 188(2):267–275. doi:10.1016/j.cbi.2010.03.040
Thévenod F (2009) Cadmium and cellular signaling cascades: to be or not to be? Toxicol Appl Pharmacol 238:221–239
Thévenod F, Lee WK (2013a) Cadmium and cellular signaling cascades: interactions between cell death and survival pathways. Arch Toxicol 87(10):1743–1786. doi:10.1007/s00204-013-1110-9
Thévenod F, Lee WK (2013b) Toxicology of cadmium and its damage to mammalian organs. Met Ions Life Sci 11:415–490. doi:10.1007/978-94-007-5179-8_14
Thévenod F, Lee WK (2015) Live and let die: roles of autophagy in cadmium nephrotoxicity. Toxics 3:130–151
Thévenod F, Wolff NA, Bork U, Lee WK, Abouhamed M (2007) Cadmium induces nuclear translocation of beta-catenin and increases expression of c-myc and Abcb1a in kidney proximal tubule cells. Biometals 20(5):807–820
Thévenod F, Lee WK, Wolff NA (2015) Rapamycin: a therapy of choice for endoplasmic reticulum stress-induced renal proximal tubule toxicity? Toxicology 330:41–43. doi:10.1016/j.tox.2015.02.001
Tokumoto M, Fujiwara Y, Shimada A et al (2011) Cadmium toxicity is caused by accumulation of p53 through the down-regulation of Ube2d family genes in vitro and in vivo. J Toxicol Sci 36(2):191–200
Urra H, Dufey E, Lisbona F, Rojas-Rivera D, Hetz C (2013) When ER stress reaches a dead end. Biochim Biophys Acta 1833(12):3507–3517. doi:10.1016/j.bbamcr.2013.07.024
Vaseva AV, Moll UM (2009) The mitochondrial p53 pathway. Biochim Biophys Acta 1787(5):414–420. doi:10.1016/j.bbabio.2008.10.005
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334(6059):1081–1086. doi:10.1126/science.1209038
Wang SH, Shih YL, Ko WC, Wei YH, Shih CM (2008) Cadmium-induced autophagy and apoptosis are mediated by a calcium signaling pathway. Cell Mol Life Sci 65(22):3640–3652. doi:10.1007/s00018-008-8383-9
Wang T, Wang Q, Song R et al (2016) Cadmium induced inhibition of autophagy is associated with microtubule disruption and mitochondrial dysfunction in primary rat cerebral cortical neurons. Neurotoxicol Teratol 53:11–18. doi:10.1016/j.ntt.2015.11.007
Wei X, Qi Y, Zhang X et al (2014) Cadmium induces mitophagy through ROS-mediated PINK1/Parkin pathway. Toxicol Mech Methods 24(7):504–511. doi:10.3109/15376516.2014.943444
Woehlbier U, Hetz C (2011) Modulating stress responses by the UPRosome: a matter of life and death. Trends Biochem Sci 36(6):329–337. doi:10.1016/j.tibs.2011.03.001
Wolff NA, Abouhamed M, Verroust PJ, Thévenod F (2006) Megalin-dependent internalization of cadmium-metallothionein and cytotoxicity in cultured renal proximal tubule cells. J Pharmacol Exp Ther 318(2):782–791
**e J, Shaikh ZA (2006) Cadmium induces cell cycle arrest in rat kidney epithelial cells in G2/M phase. Toxicology 224(1–2):56–65. doi:10.1016/j.tox.2006.04.026
Yamaji A, Sekizawa Y, Emoto K et al (1998) Lysenin, a novel sphingomyelin-specific binding protein. J Biol Chem 273(9):5300–5306
Yamamoto A, Tagawa Y, Yoshimori T, Moriyama Y, Masaki R, Tashiro Y (1998) Bafilomycin A1 prevents maturation of autophagic vacuoles by inhibiting fusion between autophagosomes and lysosomes in rat hepatoma cell line, H-4-II-E cells. Cell Struct Funct 23(1):33–42
Yokouchi M, Hiramatsu N, Hayakawa K et al (2007) Atypical, bidirectional regulation of cadmium-induced apoptosis via distinct signaling of unfolded protein response. Cell Death Differ 14(8):1467–1474. doi:10.1038/sj.cdd.4402154
Yokouchi M, Hiramatsu N, Hayakawa K et al (2008) Involvement of selective reactive oxygen species upstream of proapoptotic branches of unfolded protein response. J Biol Chem 283(7):4252–4260. doi:10.1074/jbc.M705951200
Zhou J, Li G, Zheng Y et al (2015) A novel autophagy/mitophagy inhibitor liensinine sensitizes breast cancer cells to chemotherapy through DNM1L-mediated mitochondrial fission. Autophagy 11(8):1259–1279. doi:10.1080/15548627.2015.1056970
Acknowledgements
Funding was obtained from the Center for Biomedical Training and Research (ZBAF) of the University of Witten/Herdecke (F.T.), Deutsche Forschungsgemeinschaft (TH345/11-1 to F.T.), BMBF 01DN16039 (F.T.) Max Kade Foundation (W.-K.L.), CONACYT (Consejo Nacional de Ciencia y Tecnología, Mexico) (235537 to M.P.S.-S.), NSERC Discovery grant (E.J.P.) and a fellowship from PhD program Biomedicine, Witten/Herdecke University (W. A.-H.).
Author information
Authors and Affiliations
Corresponding authors
Additional information
W.-K. Lee, S. Probst and M. P. Santoyo-Sánchez contributed equally.
Rights and permissions
About this article
Cite this article
Lee, WK., Probst, S., Santoyo-Sánchez, M.P. et al. Initial autophagic protection switches to disruption of autophagic flux by lysosomal instability during cadmium stress accrual in renal NRK-52E cells. Arch Toxicol 91, 3225–3245 (2017). https://doi.org/10.1007/s00204-017-1942-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-017-1942-9