Abstract
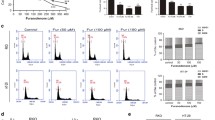
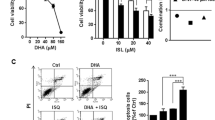
Alpha-lipoic acid (LA), which plays a pivotal role in mitochondrial energy metabolism, is an endogenous dithiol compound with an array of antioxidative functions. It has been shown that LA triggers cell death in tumor cell lines, whereas non-transformed cells are hardly affected. In the present study, we analyzed the cytotoxicity of LA on colorectal cancer (CRC) cells differing in their p53 status and investigated a putative synergistic effect with the anticancer drug 5-fluorouracil (5-FU). We show that LA induces a dose-dependent decrease in cell viability, which was independent of the p53 status as attested in isogenic p53-proficient and p53-deficient cell lines. This effect was largely attributable to cell death induction as revealed by Annexin-V/PI staining. LA-treated HCT116 cells underwent caspase-dependent and caspase-independent cell death, which was blocked by the pan-caspase inhibitor zVAD and the RIP-kinase inhibitor Necrostatin-1, respectively. In CaCO-2 and HT29 cells, LA induced caspase-dependent cell demise via activation of caspase-9, caspase-3 and caspase-7 with subsequent PARP-1 cleavage as demonstrated by immunoblot analysis, activity assays and pan-caspase inhibition. Interestingly, LA treatment did neither activate p53 nor induced genotoxic effects as shown by lack of DNA strand breaks and phosphorylation of histone 2AX. Finally, we provide evidence that LA increases the cytotoxic effect induced by the anticancer drug 5-FU as revealed by significantly enhanced cell death rates in HCT116 and CaCO-2 cells. Collectively, these findings demonstrate that LA induces CRC cell death independent of their p53 status and potentiates the cytotoxicity of 5-FU without causing DNA damage on its own, which makes it a candidate for tumor therapy.







Similar content being viewed by others
References
Abbas T, Dutta A (2009) p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer 9(6):400–414. doi:10.1038/nrc2657
Bao Q, Shi Y (2007) Apoptosome: a platform for the activation of initiator caspases. Cell Death Differ 14(1):56–65. doi:10.1038/sj.cdd.4402028
Bonnet S, Archer SL, Allalunis-Turner J et al (2007) A mitochondria-K + channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell 11(1):37–51. doi:10.1016/j.ccr.2006.10.020
Bunz F, Hwang PM, Torrance C et al (1999) Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J Clin Investig 104(3):263–269. doi:10.1172/JCI6863
Carlson DA, Smith AR, Fischer SJ, Young KL, Packer L (2007) The plasma pharmacokinetics of R-(+)-lipoic acid administered as sodium R-(+)-lipoate to healthy human subjects. Altern Med Rev 12(4):343–351
Chinery R, Brockman JA, Peeler MO, Shyr Y, Beauchamp RD, Coffey RJ (1997) Antioxidants enhance the cytotoxicity of chemotherapeutic agents in colorectal cancer: a p53-independent induction of p21WAF1/CIP1 via C/EBPbeta. Nat Med 3(11):1233–1241
Chowdhury D, Keogh MC, Ishii H, Peterson CL, Buratowski S, Lieberman J (2005) gamma-H2AX dephosphorylation by protein phosphatase 2A facilitates DNA double-strand break repair. Mol Cell 20(5):801–809. doi:10.1016/j.molcel.2005.10.003
Cunningham D, Atkin W, Lenz HJ et al (2010) Colorectal cancer. Lancet 375(9719):1030–1047. doi:10.1016/S0140-6736(10)60353-4
Degterev A, Hitomi J, Germscheid M et al (2008) Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol 4(5):313–321. doi:10.1038/nchembio.83
Dörsam B, Wu CF, Efferth T, Kaina B, Fahrer J (2014) The eucalyptus oil ingredient 1,8-cineol induces oxidative DNA damage. Arch Toxicol. doi:10.1007/s00204-014-1281-z
Dozio E, Ruscica M, Passafaro L et al (2010) The natural antioxidant alpha-lipoic acid induces p27(Kip1)-dependent cell cycle arrest and apoptosis in MCF-7 human breast cancer cells. Eur J Pharmacol 641(1):29–34. doi:10.1016/j.ejphar.2010.05.009
Fahrer J, Kaina B (2013) O6-methylguanine-DNA methyltransferase in the defense against N-nitroso compounds and colorectal cancer. Carcinogenesis 34(11):2435–2442. doi:10.1093/carcin/bgt275
Fahrer J, Huelsenbeck J, Jaurich H et al (2014) Cytolethal distending toxin (CDT) is a radiomimetic agent and induces persistent levels of DNA double-strand breaks in human fibroblasts. DNA Repair 18:31–43. doi:10.1016/j.dnarep.2014.03.002
Fava LL, Bock FJ, Geley S, Villunger A (2012) Caspase-2 at a glance. J Cell Sci 125(Pt 24):5911–5915. doi:10.1242/jcs.115105
Fulda S, Debatin KM (2006) Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 25(34):4798–4811. doi:10.1038/sj.onc.1209608
Gogvadze V, Orrenius S, Zhivotovsky B (2009) Mitochondria as targets for cancer chemotherapy. Semin Cancer Biol 19(1):57–66. doi:10.1016/j.semcancer.2008.11.007
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. doi:10.1016/j.cell.2011.02.013
Handelman GJ, Han D, Tritschler H, Packer L (1994) Alpha-lipoic acid reduction by mammalian cells to the dithiol form, and release into the culture medium. Biochem Pharmacol 47(10):1725–1730
Hiramatsu N, Kasai A, Hayakawa K, Yao J, Kitamura M (2006) Real-time detection and continuous monitoring of ER stress in vitro and in vivo by ES-TRAP: evidence for systemic, transient ER stress during endotoxemia. Nucleic Acids Res 34(13):e93. doi:10.1093/nar/gkl515
Hiratsuka T, Inomata M, Kono Y, Yokoyama S, Shiraishi N, Kitano S (2013) DHLTauZnNa, a newly synthesized alpha-lipoic acid derivative, induces autophagy in human colorectal cancer cells. Oncol Rep 29(6):2140–2146. doi:10.3892/or.2013.2394
Horn HF, Vousden KH (2007) Co** with stress: multiple ways to activate p53. Oncogene 26(9):1306–1316. doi:10.1038/sj.onc.1210263
Jones W, Li X, Qu ZC, Perriott L, Whitesell RR, May JM (2002) Uptake, recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med 33(1):83–93
Kim JW, Tchernyshyov I, Semenza GL, Dang CV (2006) HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab 3(3):177–185. doi:10.1016/j.cmet.2006.02.002
Kinner A, Wu W, Staudt C, Iliakis G (2008) Gamma-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res 36(17):5678–5694. doi:10.1093/nar/gkn550
Kopetz S, Chang GJ, Overman MJ et al (2009) Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol 27(22):3677–3683. doi:10.1200/JCO.2008.20.5278
Korotchkina LG, Sidhu S, Patel MS (2004) R-lipoic acid inhibits mammalian pyruvate dehydrogenase kinase. Free Radic Res 38(10):1083–1092. doi:10.1080/10715760400004168
Kralj M, Husnjak K, Korbler T, Pavelic J (2003) Endogenous p21WAF1/CIP1 status predicts the response of human tumor cells to wild-type p53 and p21WAF1/CIP1 overexpression. Cancer Gene Ther 10(6):457–467. doi:10.1038/sj.cgt.7700588
Liu Y, Bodmer WF (2006) Analysis of P53 mutations and their expression in 56 colorectal cancer cell lines. Proc Natl Acad Sci USA 103(4):976–981. doi:10.1073/pnas.0510146103
Mangerich A, Bürkle A (2011) How to kill tumor cells with inhibitors of poly(ADP-ribosyl)ation. Int J Cancer 128(2):251–265. doi:10.1002/ijc.25683
Marques C, Oliveira CS, Alves S et al (2013) Acetate-induced apoptosis in colorectal carcinoma cells involves lysosomal membrane permeabilization and cathepsin D release. Cell Death Dis 4:e507. doi:10.1038/cddis.2013.29
Michikoshi H, Nakamura T, Sakai K et al (2013) Alpha-lipoic acid-induced inhibition of proliferation and met phosphorylation in human non-small cell lung cancer cells. Cancer Lett 335(2):472–478. doi:10.1016/j.canlet.2013.03.008
Moungjaroen J, Nimmannit U, Callery PS et al (2006) Reactive oxygen species mediate caspase activation and apoptosis induced by lipoic acid in human lung epithelial cancer cells through Bcl-2 down-regulation. J Pharmacol Exp Ther 319(3):1062–1069. doi:10.1124/jpet.106.110965
Olivier M, Hollstein M, Hainaut P (2010) TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb Perspect Biol 2(1):a001008. doi:10.1101/cshperspect.a001008
Pack RA, Hardy K, Madigan MC, Hunt NH (2002) Differential effects of the antioxidant alpha-lipoic acid on the proliferation of mitogen-stimulated peripheral blood lymphocytes and leukaemic T cells. Mol Immunol 38(10):733–745
Pettersen HS, Visnes T, Vagbo CB et al (2011) UNG-initiated base excision repair is the major repair route for 5-fluorouracil in DNA, but 5-fluorouracil cytotoxicity depends mainly on RNA incorporation. Nucleic Acids Res 39(19):8430–8444. doi:10.1093/nar/gkr563
Pop C, Salvesen GS (2009) Human caspases: activation, specificity, and regulation. J Biol Chem 284(33):21777–21781. doi:10.1074/jbc.R800084200
Prasad PD, Wang H, Kekuda R et al (1998) Cloning and functional expression of a cDNA encoding a mammalian sodium-dependent vitamin transporter mediating the uptake of pantothenate, biotin, and lipoate. J Biol Chem 273(13):7501–7506
Reljanovic M, Reichel G, Rett K et al (1999) Treatment of diabetic polyneuropathy with the antioxidant thioctic acid (alpha-lipoic acid): a two year multicenter randomized double-blind placebo-controlled trial (ALADIN II). Alpha lipoic acid in diabetic neuropathy. Free Radic Res 31(3):171–179
Rochette L, Ghibu S, Richard C, Zeller M, Cottin Y, Vergely C (2013) Direct and indirect antioxidant properties of alpha-lipoic acid and therapeutic potential. Mol Nutr Food Res 57(1):114–125. doi:10.1002/mnfr.201200608
Rodrigues NR, Rowan A, Smith ME et al (1990) p53 mutations in colorectal cancer. Proc Natl Acad Sci USA 87(19):7555–7559
Ruemmele FM, Schwartz S, Seidman EG, Dionne S, Levy E, Lentze MJ (2003) Butyrate induced Caco-2 cell apoptosis is mediated via the mitochondrial pathway. Gut 52(1):94–100
Scian MJ, Carchman EH, Mohanraj L et al (2008) Wild-type p53 and p73 negatively regulate expression of proliferation related genes. Oncogene 27(18):2583–2593. doi:10.1038/sj.onc.1210898
Shay KP, Hagen TM (2009) Age-associated impairment of Akt phosphorylation in primary rat hepatocytes is remediated by alpha-lipoic acid through PI3 kinase, PTEN, and PP2A. Biogerontology 10(4):443–456. doi:10.1007/s10522-008-9187-x
Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM (2009) Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta 1790(10):1149–1160. doi:10.1016/j.bbagen.2009.07.026
Shi DY, Liu HL, Stern JS, Yu PZ, Liu SL (2008) Alpha-lipoic acid induces apoptosis in hepatoma cells via the PTEN/Akt pathway. FEBS Lett 582(12):1667–1671. doi:10.1016/j.febslet.2008.04.021
Simbula G, Columbano A, Ledda-Columbano GM et al (2007) Increased ROS generation and p53 activation in alpha-lipoic acid-induced apoptosis of hepatoma cells. Apoptosis 12(1):113–123. doi:10.1007/s10495-006-0487-9
Smith AR, Shenvi SV, Widlansky M, Suh JH, Hagen TM (2004) Lipoic acid as a potential therapy for chronic diseases associated with oxidative stress. Curr Med Chem 11(9):1135–1146
Soldani C, Scovassi AI (2002) Poly(ADP-ribose) polymerase-1 cleavage during apoptosis: an update. Apoptosis Int J Progr Cell Death 7(4):321–328
Suh JH, Shenvi SV, Dixon BM et al (2004) Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc Natl Acad Sci USA 101(10):3381–3386. doi:10.1073/pnas.0400282101
Takaishi N, Yoshida K, Satsu H, Shimizu M (2007) Transepithelial transport of alpha-lipoic acid across human intestinal Caco-2 cell monolayers. J Agric Food Chem 55(13):5253–5259. doi:10.1021/jf063624i
Teichert J, Kern J, Tritschler HJ, Ulrich H, Preiss R (1998) Investigations on the pharmacokinetics of alpha-lipoic acid in healthy volunteers. Int J Clin Pharmacol Ther 36(12):625–628
Trivedi PP, Jena GB (2013) Role of alpha-lipoic acid in dextran sulfate sodium-induced ulcerative colitis in mice: studies on inflammation, oxidative stress, DNA damage and fibrosis. Food Chem Toxicol 59:339–355. doi:10.1016/j.fct.2013.06.019
Vakifahmetoglu-Norberg H, Zhivotovsky B (2010) The unpredictable caspase-2: what can it do? Trends Cell Biol 20(3):150–159. doi:10.1016/j.tcb.2009.12.006
van de Mark K, Chen JS, Steliou K, Perrine SP, Faller DV (2003) Alpha-lipoic acid induces p27Kip-dependent cell cycle arrest in non-transformed cell lines and apoptosis in tumor cell lines. J Cell Physiol 194(3):325–340. doi:10.1002/jcp.10205
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324(5930):1029–1033. doi:10.1126/science.1160809
Wenzel U, Nickel A, Daniel H (2005) Alpha-lipoic acid induces apoptosis in human colon cancer cells by increasing mitochondrial respiration with a concomitant O2-*-generation. Apoptosis 10(2):359–368. doi:10.1007/s10495-005-0810-x
White E (2012) Deconvoluting the context-dependent role for autophagy in cancer. Nat Rev Cancer 12(6):401–410. doi:10.1038/nrc3262
Yadav V, Marracci G, Lovera J et al (2005) Lipoic acid in multiple sclerosis: a pilot study. Multiple sclerosis 11(2):159–165
Yoo TH, Lee JH, Chun HS, Chi SG (2013) alpha-Lipoic acid prevents p53 degradation in colon cancer cells by blocking NF-kappaB induction of RPS6KA4. Anticancer Drugs 24(6):555–565. doi:10.1097/CAD.0b013e32836181eb
Zachar Z, Marecek J, Maturo C et al (2011) Non-redox-active lipoate derivates disrupt cancer cell mitochondrial metabolism and are potent anticancer agents in vivo. J Mol Med (Berl) 89(11):1137–1148. doi:10.1007/s00109-011-0785-8
Ziegler D, Hanefeld M, Ruhnau KJ et al (1995) Treatment of symptomatic diabetic peripheral neuropathy with the anti-oxidant alpha-lipoic acid. A 3-week multicentre randomized controlled trial (ALADIN Study). Diabetologia 38(12):1425–1433
Acknowledgments
The author would like to thank Dr. Bert Vogelstein (Baltimore, USA) for providing HCT116 cells. We are grateful to Anna Frumkina (Mainz, Germany) for performing the alkaline comet assay. This work was supported by the University of Mainz (Stufe I, grant to J.F.) and University Medical Center Mainz (MAIFOR).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dörsam, B., Göder, A., Seiwert, N. et al. Lipoic acid induces p53-independent cell death in colorectal cancer cells and potentiates the cytotoxicity of 5-fluorouracil. Arch Toxicol 89, 1829–1846 (2015). https://doi.org/10.1007/s00204-014-1434-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1434-0