Abstract
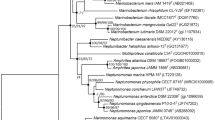
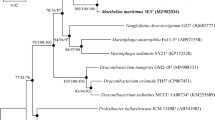
A Gram-negative, rod-shaped, motile bacterium, designated strain CAU 1620T, was isolated from a tidal flat sediment in Incheon, Republic of Korea. Strain CAU 1620T grew optimally at 30 °C and pH 8.0 in the presence of 6.0% (w/v) NaCl. The results of 16S rRNA gene sequence analysis revealed that strain CAU 1620T showed the highest similarity to Marinobacter adhaerens DSM 23420T (98.5%), followed by Marinobacter algicola DSM 16394T (98.3%) and Marinobacter maroccanus LMG 30465T (98.2%). The average nucleotide identity and digital DNA–DNA hybridisation values between strain CAU 1620T and related strains were estimated as 75.6–78.1% and 19.5–20.9%, respectively. The DNA G + C content based on the draft genome sequence was 59.2%, and the major respiratory quinone was ubiquinone-9. The predominant cellular fatty acids were C12:0, C16:0, C18:1 ω9c, and C12:0 3OH. The predominant polar lipids were diphosphatidylglycerol, phosphatidylethanolamine, and phosphatidylglycerol. According to phenotypic, genotypic, and chemotaxonomic analyses, strain CAU 1620T represents a novel species of the genus Marinobacter, for which the name Marinobacter arenosus sp. nov. is proposed. The type strain is CAU 1620T (= KCTC 82431T = MCCC 1K06079T).


Similar content being viewed by others
References
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagniko O (2008) The RAST server: rapid annotations using subsystems technology. BMC Genom 9:75. https://doi.org/10.1186/1471-2164-9-75
Blin K, Shaw S, Villebro SK, Zeimert NR, Lee SY, Medema M, Weber T (2019) Antismash 5.0: updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res 47:W81–W87. https://doi.org/10.1093/nar/gkz310
Boujida N, Palau M, Charfi S, Manresa A, Senhaji NS, Abrini J, Minana-Galbis D (2019) Marinobacter maroccanus sp. nov., a moderately halophilic bacterium isolated from a saline soil. Int J Syst Evol Microbiol 69:227–234. https://doi.org/10.1099/ijsem.0.003134
Bowman JP (2000) Description of Cellulophaga algicola sp. nov., isolated from the surfaces of Antarctic algae, and reclassification of Cytophaga uliginosa (ZoBell and Upham 1944) Reichenbach 1989 as Cellulophaga uliginosa comb. nov. Int J Syst Evol Microbiol 50:1861–1868. https://doi.org/10.1099/00207713-50-5-1861
Fang C, Wu YH, Xamxidin M, Wang CS, Xu XW (2017) Maribacter cobaltidurans sp. nov., a heavy-metal-tolerant bacterium isolated from deep-sea sediment. Int J Syst Evol Microbiol 67:5261–5267. https://doi.org/10.1099/ijsem.0.002458
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Felsenstein J (1989) PHYLIP—phylogeny inference package (version 3.2). Cladistics, pp 164–166
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Zool 20:406–416. https://doi.org/10.1093/sysbio/20.4.406
Gauthier MJ, Lafay B, Christen R, Fernandez L, Acquaviva M, Bonin P, Bertrand JC (1992) Marinobacter hydrocarbonoclasticus gen. nov., sp. nov., a new, extremely halotolerant, hydrocarbon-degrading marine bacterium. Int J Syst Evol Microbiol 42:568–576. https://doi.org/10.1099/00207713-42-4-568
Green DH, Bowman JP, Smith EA, Gutierrez T, Bolch CJ (2006) Marinobacter algicola sp. nov., isolated from laboratory cultures of paralytic shellfish toxin-producing dinoflagellates. Int J Syst Evol Microbiol 56:523–527. https://doi.org/10.1099/ijs.0.63447-0
Henz SR, Huson DH, Auch AF, Nieselt-Struwe K, Schuster SC (2005) Whole genome prokaryotic phylogeny. Bioinformatics 21:2329–2335. https://doi.org/10.1093/bioinformatics/bth324
Kaeppel EC, Gardes A, Seebah S, Grossart HP, Ullrich MS (2012) Marinobacter adhaerens sp. nov., isolated from marine aggregates formed with the diatom Thalassiosira weissflogii. Int J Syst Evol Microbiol 62:124–128. https://doi.org/10.1099/ijs.0.030189-0
Kang HS, Yang HL, Lee SD (2009) Nitratireductor kimnyeongensis sp. nov., isolated from seaweed. Int J Syst Evol Microbiol 59:1036–1039. https://doi.org/10.1099/ijs.0.65734-0
Kim JH, Ward AC, Kim W (2015) Kangiella chungangensis sp. nov. Isolated from a marine sand. Antonie Van Leeuwenhoek 107:1291–1298. https://doi.org/10.1007/s10482-015-0423-5
Komagata K, Suzuki K (1987) 4 lipid and cell-wall analysis in bacterial systematics. Methods Microbiol 19:161–208. https://doi.org/10.1016/S0580-9517(08)70410-0
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lane D (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, pp 115–175
Lee I, Kim YO, Park SC, Chun J (2016) OrthoANI: an improved algorithm and software for calculating average nucleotide identity. Int J Syst Evol Microbiol 66:1100–1103. https://doi.org/10.1099/ijsem.0.000760
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 21:14–60. https://doi.org/10.1186/1471-2105-14-60
Rodriguez-Valera F, Ruiz-Berraquero F, Ramos-Cormenzana A (1981) Characteristics of the heterotrophic bacterial populations in hypersaline environments of different salt concentrations. Microb Ecol 7:235–243. https://doi.org/10.1007/BF02010306
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Smibert RM, Krieg NR (1994) Phenotypic characterization. In: Gerhardt P, Murray RG, Wood WA, Krieg NR (eds) Methods for general and molecular bacteriology. American Society for Microbiology, Washington, pp 607–654
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Wayne LG, Brenner DJ, Colwell RR, Grimont PAD, Kandler O, Krichevsky MI, Moore LH, Moore WEC, Murray RGE, Stackebrandt E, Starr MP, Truper HG (1987) Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 37:463–464. https://doi.org/10.1099/00207713-37-4-463
Acknowledgements
This work was supported by a grant from the National Institute of Biological Resources (NIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR201902203), and the Chung-Ang University Research Grants in 2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The 16S rRNA gene sequence and the whole-genome sequence of strain CAU 1620T have been deposited in GenBank/EMBL/DDBJ as the accession numbers MW019609 and JAHVAO000000000, respectively.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, Y., Weerawongwiwat, V., Kim, JH. et al. Marinobacter arenosus sp. nov., a halotolerant bacterium isolated from a tidal flat. Arch Microbiol 204, 155 (2022). https://doi.org/10.1007/s00203-021-02696-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02696-7