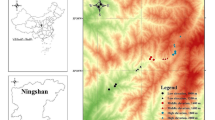
Abstract
Some parasitic fungi can increase fitness by modifying the behavior of their hosts. These behaviors are known as extended phenotypes because they favor parasitic gene propagation. Here, we studied three lineages of Ophiocordyceps, a fungus that infects ants, altering their conduct before death. According to fungal strategy, ants may die in leaf litter, with entwined legs in branches, under the moss mat, or biting plant tissue. It is critical for parasites that the corpses stay at these places because Ophiocordyceps exhibit iteroparity, possibly releasing spores in multiple life cycles. Thus, we assumed substrate cadaver permanence as a fungi reproductive proxy and corpse height as a proxy of cadaver removal. We hypothesize that biting vegetation and dying in higher places may increase the permanence of ant corpses while avoiding possible corpse predation on the forest floor. We monitored over a year more than 4000 zombie ants in approximately 15 km2 of undisturbed tropical forest in central Amazonia. Our results show a longer permanence of corpses with increasing ground height, suggesting that the parasites may have better chances of releasing spores and infecting new hosts at these places. We found that the zombie ants that last longer on the substrate die under the moss mat in tree trunks, not necessarily biting vegetation. The biting behavior appears to be the most derived and complex mechanism among Ophiocordyceps syndromes. Our results put these findings under a new perspective, proposing that seemingly less complex behavioral changes are ecologically equivalent and adaptative for other parasite lineages.





Similar content being viewed by others
References
Andersen SB, Ferrari M, Evans HC, Elliot SL, Boomsma JJ, Hughes DP (2012) Disease dynamics in a specialized parasite of ant societies. PloS one 7(5):e36352. https://doi.org/10.1371/journal.pone.0036352
Andersen SB, Gerritsma S, Yusah KM, Mayntz D, Hywel-Jones NL, Billen J, Boomsma JJ, Hughes DP (2009) The life of a dead ant: the expression of an adaptive extended phenotype. Am Nat 174:424–433
Andriolli FS, Ishikawa NK, Vargas-Isla R, Cabral TS, de Bekker C, Baccaro FB (2019) Do zombie ant fungi turn their hosts into light seekers? Behav Ecol 30(3):609–616
Araújo JPM, Evans HC, Geiser DM, Mackay WP, Hughes DP (2015) Unraveling the diversity behind the Ophiocordyceps unilateralis (Ophiocordycipitaceae) complex: three new species of zombie-ant fungi from the Brazilian Amazon. Phytotaxa 220(3):224–238
Araújo JPM, Hughes DP (2017) The fungal spore: myrmecophilous Ophiocordyceps as a case study. The fungal community: its organization and role in the ecosystem, 4th edn. CRC Press
Araújo JP, Hughes DP (2019) Zombie-ant fungi emerged from non-manipulating, beetle-infecting ancestors. Curr Biol 29(21):3735–8. https://doi.org/10.1016/j.cub.2019.09.004
Araújo JPM, Evans HC, Kepler R, Hughes DP (2018) Zombie-ant fungi across continents: 15 new species and new combinations within Ophiocordyceps I. Myrmecophilous hirsutelloid species. Stud Mycol 90:1191e60
Araújo JPM, Moriguchi MG, Uchiyama S, Kinjo N, Matsuura Y (2021) Ophiocordyceps salganeicola, a parasite of social cockroaches in Japan and insights into the evolution of other closely related Blattodea-associated lineages. IMA Fungus 12:1–17
Baccaro FB, Feitosa RM, Fernandez F, Fernandes IO, Izzo TJ, Souza JP, Solar R (2015) Guia para os gêneros de formigas do Brasil. Manaus, INPA
Barbosa BC, Halfeld VR, de Araújo JPM, Maciel TT, Prezoto F (2015) Record of Ophiocordyceps unilateralis sensu lato, the zombie-ant fungus, parasitizing Camponotus in an urban fragment of Atlantic Rainforest in southeastern Brazil. Stud Neotropical Fauna Environ 50(1):21–23
Cardoso Neto JA, Leal LC, Baccaro FB (2019) Temporal and spatial gradients of humidity shape the occurrence and the behavioral manipulation of ants infected by entomopathogenic fungi in Central Amazon. Fungal Ecol 42:100871
Carroll CR, Janzen DH (1973) Ecology of foraging by ants. Annu Rev Ecol Syst 4(1):231–257
Chung TY, Sun PF, Kuo JI, Lee YI, Lin CC, Chou JY (2017) Zombie ant heads are oriented relative to solar cues. Fungal Ecol 25:22–28
Costa FV, Costa FRC, Magnusson WE, Franklin E, Zuanon J, Cintra R, Luizão F, Camargo JLC, Andrade A, Laurance WF, Baccaro F, Pereira Souza JL, Espírito-Santo H (2015) Synthesis of the first 10 years of long-term ecological research in Amazonian forest ecosystem - implications for conservation and management. Nat Conservação 13:3–14
Costa FRC, Zuanon JAS, Baccaro FB, Schietti J, Menger JS, Souza JLP, Borba GC, Esteban EJL, Bertin VM, Gerolamo CS et al (2020) Effects of climate change on central Amazonian forests: a two decades synthesis of monitoring tropical biodiversity. Oecol Australis 24:317–335
Cox D (1972) Regression models and life-tables. J Roy Stat Soc: Ser B (Methodol) 34(2):187–220
Dawkins R & Krebs JR (1979) Arms races between and within species. Proc R Soc Lond B 205(1161):489-511
Dawkins R (1982) The extended phenotype: the long reach of the gene. Oxford University Press, Oxford (UK)
de Bekker C, Ohm RA, Loreto RG, Sebastian A, Albert I, Merrow M, Brachmann A, Hughes DP (2015) Gene expression during zombie ant biting behavior reflects the complexity underlying fungal parasitic behavioral manipulation. BMC Genomics 16:620
de Bekker C, Will I, Hughes DP, Brachmann A, Merrow M (2017) Daily rhythms and enrichment patterns in the transcriptome of the behavior-manipulating parasite Ophiocordyceps kimflemingiae. PLoS ONE 12:e0187170
de Bekker C, Das B (2022) Hijacking time: how Ophiocordyceps fungi could be using ant host clocks to manipulate behavior. Parasite Immunol 44:e12909
de Meeûs T, Michalakis Y, Renaud F (1998) Santa Rosalia revisited: or why are there so many kinds of parasites in ‘The Garden of Earthly Delights’? Parasitol Today 14:10–13
Eldridge DJ, Guirado E, Reich PB et al (2023) The global contribution of soil mosses to ecosystem services. Nat Geosci 16:430–438
Evans HC (1982) Entomogenous fungi in the tropical forest ecosystems: an appraisal. Ecol Entomol 7:47–60
Evans HC, Elliot SL, Hughes DP (2011) Hidden diversity behind the zombie-ant fungus Ophiocordyceps unilateralis: four new species described from carpenter ants in Minas Gerais, Brazil. Plos One 6:e17024
Evans HC, Samson RA (1982) Cordyceps species and their anamorphs pathogenic on ants (Formicidae) in tropical forest ecosystems I. The Cephalotes (Formicinae) complex. Trans Br Mycol Soc 79:431e453
Evans HC, Samson RA (1984) Cordyceps species and their anamorphs pathogenic on ants (Formicidae) in tropical forest ecosystems. II. The Camponotus (Formicinae) complex. Trans Br Mycol Soc 79:127–150
Falesi IC, Cruz ES, Pereira FB, Lopes EC (1969) Os solos da área da Manaus-ltacoatiara. Instituto de Pesquisa e Experimentação Agropecuária do Norte (IPEAN). Sér Estudos Ensaios 1:116
Gracia ES, de Bekker C, Hanks EM, Hughes DP (2018) Within the fortress: a specialized parasite is not discriminated against in a social insect society. PLoS ONE 13:e0193536
Griffiths HM, Ashton LA, Walker AE, Hasan F, Evans TA, Eggleton P, Parr CL (2018) Ants are the major agents of resource removal from tropical rainforests. J Anim Ecol 87(1):293–300
Guillaumet JL (1987) Some structural and floristic aspects of the forest. pdf Experientia 43:241–251
Hirshfield MF, and Tinkle DW (1975) Natural selection and the evolution of reproductive effort. Proc Natl Acad Sci USA 72(6):2227–2231
Holldober B, Wilson EO (1990) Host tree selection by the neotropical ant Paraponera clavata (Hymenoptera Formicidae). Biotropica 22(2):213–214
Hughes DP, Andersen SB, Hywel-Jones NL, Himaman W, Billen J, Boomsma JJ (2011) Behavioral mechanisms and morphological symptoms of zombie ants dying from fungal infection. BMC Ecol 11:1–10
Hughes DP, Araújo JP, Loreto R, Quevillon L, de Bekker C, Evans HC (2016) From so simple a beginning: the evolution of behavioral manipulation by fungi. Adv Genet 94:1–33
Imirzian N, Araújo JPM, Hughes DP (2020) A new zombie ant behavior unraveled: aggregating on tree trunks. J Invertebr Pathol 177:107499
Kassambara A, Kosinski M and Biecek P (2021) Survminer: drawing survival curves using 'ggplot2'. R package version 0.4.9. https://CRAN.R-project.org/package=survminer. Accessed 13 June 2024
Kobmoo N, Mongkolsamrit S, Tasanathai K, Thanakitpipattana D, Luangsa-ard JJ (2012) Molecular phylogenies reveal host-specific divergence of Ophiocordyceps unilateralis sensu lato following its host ants. Mol Ecol 21:3022e3031
Kobmoo N, Mongkolsamrit S, Wutikhun T, Tasanathai K, Khonsanit A, Thanakitpipattana D, Luangsa-Ard JJ (2015) New species of Ophiocordyceps unilateralis, a ubiquitous pathogen of ants from Thailand. Fungal Biol 119(1):44–52
Loreto RG, Araújo JPM, Kepler RM, Fleming KR, Moreau CS, Hughes DP (2018) Evidence for convergent evolution of host parasitic manipulation in response to environmental conditions. Evolution 72(10):2144–2155
Loreto RG, Elliot SL, Freitas MLR, Pereira TM, Hughes DP (2014) Long-term disease dynamics for a specialized parasite of ant societies: a field study. PLoS ONE 9(8):e103516
MacKay WP, MacKay E (2010) The systematics and biology of the New World ants of the genus Pachycondyla (Hymenoptera: Formicidae). Edwin Mellen Press, Lewiston, New York
Mongkolsamrit S, Kobmoo N, Tasanathai K, Khonsanit A, Noisripoom, et al (2012) Life cycle, host range and temporal variation of Ophiocordyceps unilateralis/Hirsutella formicarum on Formicine ants. J Invertebr Pathol 111(3):217–224
Moore J (2002) Parasites and the behavior of animals. Oxford University Press, New York
Muenchow G (1986) Ecological use of failure time analysis. Ecology 67:246–250
Okamura B, Hartigan A, Naldoni J (2018) Extensive uncharted biodiversity: the parasite dimension. Integr Comp Biol 58:1132–1145
Ortiz CM, Fernández F (2011) Hormigas del género Dolichoderus Lund (Formicidae: Dolichoderinae). Universidad Nacional de Colombia, Bogotá
Poulin R, Morand S (2000) The diversity of parasites. Q Rev Biol 75(3):277–293
Price PW (1980) Evolutionary biology of parasites. Princeton University Press, Princeton (NJ)
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. Accessed 13 June 2024
Ribeiro MNG (1976) Aspectos climatológicos de Manaus. Acta Amazon 6(2):229–233
Sakolrak B, Blatrix R, Sangwanit U, Kobmoo N (2018) Experimental infection of the ant polyrhachis furcata with ophiocordyceps reveals specificity of behavioral manipulation. Fungal Ecol 33:122–124
Schmid-Hempel P (2011) Evolutionary parasitology. The integrated study of infections, immunology, ecology, and genetics. Oxford University Press, Oxford, UK
Somavilla A, Bartholomay P, Soares M (2019) Behavior manipulation of Crabronidae and Pompilidae (Hymenoptera) by the entomopathogenic fungus Ophiocordyceps humbertii (Ascomycota: Hypocreales) in an Amazonian Rainforest, Brazil. Rev Bras Zoociências 20(2):1–7
Therneau T (2021) A package for survival analysis in R. R package version 3.2–11. https://CRAN.R-project.org/package=survival. Accessed 13 June 2024
Wickham et al (2019) Welcome to the tidyverse. J Open-Source Softw 4(43):1686. https://doi.org/10.21105/joss.01686
Wickham H (2016) Ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Will I, Linehan S, Jenkins DG, de Bekker C (2023) Natural history and ecological effects on the establishment and fate of Florida carpenter ant cadavers infected by the parasitic manipulator Ophiocordyceps camponoti-floridani. Funct Ecol 37(4):886–99. https://doi.org/10.1111/1365-2435.14224
Windsor DA (1998) Most of species on earth are parasites. Int J Parasitol 28:1939–1941
Acknowledgements
We are thankful to J. da Silva Lopes for fieldwork assistance, to L. O. Demarchi and S. P. de Lima for the helpful comments on the manuscript, and to N.M. Kinap for the study area figure. Also, we acknowledge the Brazilian Biodiversity Research Program (PPBio) and the National Institute for Amazonian Biodiversity Research (CENBAM). This work was only possible due to 20 years of research under the Brazilian LTER – CHAMADA PÚBLICA Nº 021/2020 – PELD/CNPq/FAPEAM.
Funding
This study was financed in part by the Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM)—POSGRAD/scholarship/ financial support and by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. The Fieldwork was financed by CHAMADA PÚBLICA Nº 021/2020—PELD/CNPq/FAPEAM. FBB receives continuous support by CNPq grant (#313986/2020–7).
Author information
Authors and Affiliations
Contributions
FSA and FBB contributed to the study’s conception and design. JACN conducted the data collection. FSA and FBB performed the analysis. JWM and FBB supervised the project and reviewed versions of the manuscript. All authors contributed, commented, and approved the submitted manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by: Sean O'Donnell
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andriolli, F.S., Cardoso Neto, J.A., de Morais, J.W. et al. With the dead under the mat: the zombie ant extended phenotype under a new perspective. Sci Nat 111, 33 (2024). https://doi.org/10.1007/s00114-024-01920-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-024-01920-w