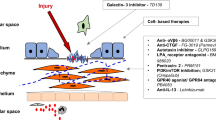
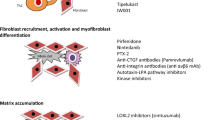
Abstract
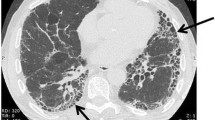
Idiopathic pulmonary fibrosis (IPF) is a persistent and an aggravating interstitial alveolar disease, marked by thickening and scarring of the lung parenchyma with unknown etiology leading to early death among the elderly population worldwide. The vast knowledge on the understanding of the pathogenesis, diagnosis, and therapeutic management has been raised significantly in recent years to delay the progression of IPF. Currently, nintedanib and pirfenidone are two medications used to treat IPF which significantly restores the alveolar epithelial functions; however, it is associated with a few demerits. Thus, new approaches are needed to overcome hurdles raised from IPF pathogenesis. Up-to-date approaches in pulmonary rehabilitation, non-pharmacological strategies, lung transplantation, and comorbidity management are mainly involved in subsiding the symptoms and thereby attempt to improve patient’s health outcomes. Hence clinical trial studies are in search of novel molecular and cellular targets to overcome IPF. This chapter highlights the understanding of up-to-date molecular and cellular targets and potential approaches which will create an avenue to design and develop novel therapeutics to defend against this complex and injurious disease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Selman M, King TE, Pardo A (2001) Idiopathic pulmonary fibrosis: prevailing and evolving hypotheses about its pathogenesis and implications for therapy. Ann Intern Med 134:136–151
Wolters PJ, Blackwell TS, Eickelberg O, Loyd JE, Kaminski N et al (2018) Time for a change: Is idiopathic pulmonary fibrosis still idiopathic and only fibrotic? Lancet Respir Med 6:154–160
Jenskin G (2020) Demystifying pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 319:L554–L559
Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J et al (2011) An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 183:788–824
Iwata T, Yoshida S, Nagato K, Nakajima T, Suzuki H et al (2015) Experience with perioperative pirfenidone for lung cancer surgery in patients with idiopathic pulmonary fibrosis. Surg Today 45:1263–1270
Iwata T, Yoshida S, Fujiwara T, Wada H, Nakajima T (2016) Effect of perioperative Pirfenidone treatment in lung cancer patients with idiopathic pulmonary fibrosis. Ann Thorac Surg 102:1905–1910
Karampitsakos T, Tzilas V, Tringidou R, Steiropoulos P, Aidinis V et al (2017) Lung cancer in patients with idiopathic pulmonary fibrosis. Pulm Pharmacol Ther 45:1–10
Goto T (2018) Measuring surgery outcomes of lung Cancer patients with concomitant pulmonary fibrosis: a review of the literature. Cancers 10:223
Daniels CE, Jett JR (2005) Does interstitial lung disease predispose to lung cancer? Curr Opin Pulm Med 11:431–437
Hendriks LE, Drent M, van Haren EH, Verschakelen JA, Verleden GM (2012) Lung cancer in idiopathic pulmonary fibrosis patients diagnosed during or after lung transplantation. Respir Med Case Rep 5:37–39
King TE Jr, Schwarz MI, Brown K, Tooze JA, Colby TV et al (2001) Idiopathic pulmonary fibrosis: relationship between histopathologic features and mortality. Am J Respir Crit Care Med 164:1025–1032
Cha SI, Chang CS, Kim EK, Lee JW, Matthay MA et al (2012) Lung mast cell density defines a subpopulation of patients with idiopathic pulmonary fibrosis. Histopathology 61:98–106
Noth I, Zhang Y, Ma SF, Flores C, Barber M et al (2013) Genetic variants associated with idiopathic pulmonary fibrosis susceptibility and mortality: a genome-wide association study. Lancet Resp Med 1:309–317
Prasse A, Probst C, Bargagli E, Zissel G, Toews GB et al (2009) Serum CC-chemokine ligand 18 concentration predicts outcome in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 179:717–723
Rosas IO, Richards TJ, Konishi K, Zhang Y, Gibson K et al (2008) MMP1 and MMP7 as potential peripheral blood biomarkers in idiopathic pulmonary fibrosis. PLoS Med 5:e93
Wolters PJ, Collard HR, Jones KD (2014) Pathogenesis of idiopathic pulmonary fibrosis. Annu Rev Pathol 9:157–179
Ley B, Collard HR (2013) Epidemiology of idiopathic pulmonary fibrosis. Clin Epidemiol 5:483–492
Hoffman TW, van Moorsel CHM, Borie R, Crestani B (2018) Pulmonary phenotypes associated with genetic variation in telomere-related genes. Curr Opin Pulm Med 24:269–280
Cunningham PS, Meijer P, Nazgiewicz A, Anderson SG, Borthwick LA et al (2020) The circadian clock protein REVERBα inhibits pulmonary fibrosis development. Proc Natl Acad Sci U S A 117:1139–1147
Takezaki A, Tsukumo S, Setoguchi Y, Ledford JG, Goto H et al (2019) A homozygous SFTPA1 mutation drives necroptosis of type II alveolar epithelial cells in patients with idiopathic pulmonary fibrosis. J Exp Med 216:2724–2735
Meltzer EB, Noble P (2008) Idiopathic pulmonary fibrosis. Orphanet J Rare Dis 3:3–8
Cuddapah S, Barski A, Zhao K (2010) Epigenomics of T cell activation, differentiation, and memory. Curr Opin Immunol 22:341–347
Arrowsmith CH, Bountra C, Fish PV, Lee K, Schapira M (2012) Epigenetic protein families: a new frontier for drug discovery. Nat Rev Drug Discov 11:384–400
Rivera CM, Ren B (2013) Map** human epigenomes. Cell 155:39–55
Li H, Zhao X, Shan H, Liang H (2016) MicroRNAs in idiopathic pulmonary fibrosis: involvement in pathogenesis and potential use in diagnosis and therapeutics. Acta Pharm Sin B 6:531–539
Kidd C, Hayden BY (2015) The psychology and neuroscience of curiosity Celeste. Neuron 88:449–460
Helling BA, Yang IV (2015) Epigenetics in lung fibrosis: from pathobiology to treatment perspective. Curr Opin Pulm Med 21:454–462
Keane MP, Strieter RM (2002) The importance of balanced pro-inflammatory and anti-inflammatory mechanisms in diffuse lung disease. Respir Res 3:5
Telugu AN, Chen H, Weng T, Bhaskaran M, ** N et al (2006) Expression profile of IGF system during lung injury and recovery in rats exposed to hyperoxia: a possible role of IGF-1 in alveolar epithelial cell proliferation and differentiation. Cell Biochem 97:984–998
Camelo A, Dunmore R, Sleeman MA, Clarke DL (2013) The epithelium in idiopathic pulmonary fibrosis: breaking the barrier. Front Pharmacol 4:173
Souto JT, Aliberti JC, Campanelli AP, Livonesi MC, Maffei CML et al (2003) Chemokine production and leukocyte recruitment to the lungs of Paracoccidioides brasiliensis-infected mice is modulated by interferon-γ. Am J Pathol 163:583–590
Linthout SV, Miteva K, Tschöpe C (2014) Crosstalk between fibroblasts and inflammatory cells. Cardiovasc Res 102:258–269
Kolb M, Margetts PJ, Anthony DC, Pitossi F, Gauldie J (2001) Transient expression of IL-1beta induces acute lung injury and chronic repair leading to pulmonary fibrosis. J Clin Invest 107:1529–1536
Genersch E, Hayess K, Neuenfeld Y, Haller H (2000) Sustained ERK phosphorylation is necessary but not sufficient for MMP-9 regulation in endothelial cells: involvement of Ras-dependent and -independent pathways. J Cell Sci 113:4319–4330
Sime PJ, Marr RA, Gauldie D, **ng Z, Hewlett BR (1998) Transfer of tumor necrosis factor-alpha to rat lung induces severe pulmonary inflammation and patchy interstitial fibrogenesis with induction of transforming growth factorbeta1 and myofibroblasts. Am J Pathol 153:825–832
Hyldgaard C, Hilberg O, Bendstrup E (2014) How does comorbidity influence survival in idiopathic pulmonary fibrosis? Respir Med 108:647–653
Kreuter M, Ehlers-Tenenbaum S, Palmowski K, Bruhwyler J, Oltmanns U et al (2016) Impact of comorbidities on mortality in patients with idiopathic pulmonary fibrosis. PLoS One 11:151425
Tsakiri KD, Cronkhite JT, Kuan PJ, **ng C, Raghu G et al (2007) Adult-onset pulmonary fibrosis caused by mutations in telomerase. Proc Natl Acad Sci U S A 104:7552–7557
Vancheri C, Failla M, Crimi N, Raghu G (2010) Idiopathic pulmonary fibrosis: a disease with similarities and links to cancer biology. Eur Respir J 35:496–504
Raghu G (2003) The role of gastroesophageal reflux in idiopathic pulmonary fibrosis. Am J Med 115:60–64
Antonio GE, Wong KT, Hui DS, Wu A, Lee N et al (2003) Thin-section CT in patients with severe acute respiratory syndrome following hospital discharge: preliminary experience. Radiology 228:810–815
Hui DS, Joynt GM, Wong KT, Gomersall CD, Li TS et al (2005) Impact of severe acute respiratory syndrome (SARS) on pulmonary function, functional capacity and quality of life in a cohort of survivors. Thorax 60:401–409
Barkauskas CE, Noble PW (2014) Cellular mechanisms of tissue fibrosis. New insights into the cellular mechanisms of pulmonary fibrosis. Am J Physiol Cell Physiol 306:C987–C996
Wynn TA (2011) Integrating mechanisms of pulmonary fibrosis. J Exp Med 208:1339–1350
Kropski JA, Blackwell TS (2019) Progress in understanding and treating idiopathic pulmonary fibrosis. Annu Rev Med 70:211–224
Yanagihara T, Sato S, Upagupta C, Kolb M (2019) What have we learned from basic science studies on idiopathic pulmonary fibrosis? Eur Respir Rev 28:190029
Selman M, Pardo A (2014) Revealing the pathogenic and aging-related mechanisms of the enigmatic idiopathic pulmonary fibrosis. An integral model. Am J Respir Crit Care Med 189:1161–1172
Lehmann M, Korfei M, Mutze K, Klee S, Skronska-Wasek W et al (2017) Senolytic drugs target alveolar epithelial cell function and attenuate experimental lung fibrosis ex vivo. Eur Respir J 50:1602367
Schafer MJ, White TA, Iijima K, Haak AJ, Ligresti G et al (2017) Cellular senescence mediates fibrotic pulmonary disease. Nat Commun 8:14532
Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H et al (2015) The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell 14:644–658
Rangarajan S, Bone NB, Zmijewska AA, Jiang S, Won-park D et al (2018) Metformin reverses established lung fibrosis in a bleomycin model. Nat Med 24:1121–1127
Sato N, Takasaka N, Yoshida M, Tsubouchi K, Minagawa S et al (2016) Metformin attenuates lung fibrosis development via NOX4 suppression. Respir Res 17:107
Xu Y, Mizuno T, Sridharan A, Du Y, Guo M et al (2016) Single-cell RNA sequencing identifies diverse roles of epithelial cells in idiopathic pulmonary fibrosis. JCI Insight 1:e90558
Horan GS, Wood S, Ona V, Li DJ, Lukashev ME et al (2008) Partial inhibition of integrin alpha (v) beta6 prevents pulmonary fibrosis without exacerbating inflammation. Am J Respir Crit Care Med 177:56–65
Munger JS, Huang X, Kawakatsu H, Griffiths MJ, Dalton SL et al (1999) The integrin alpha v beta 6 binds and activates latent TGF beta 1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96:319–328
Cosgrove GP, Brown KK, Schiemann WP, Serls AE, Parr JE et al (2004) Pigment epithelium-derived factor in idiopathic pulmonary fibrosis: a role in aberrant angiogenesis. Am J Respir Crit Care Med 170:242–251
Kotani I, Sato A, Hayakawa H, Urano T, Takado Y et al (1995) Increased procoagulant and antifibrinolytic activities in the lungs with idiopathic pulmonary fibrosis. Thromb Res 77:493–504
Kim KK, Kugler MC, Wolters PJ, Robillard L, Galvez MG et al (2006) Alveolar epithelial cell mesenchymal transition develops in vivo during pulmonary fibrosis and is regulated by the extracellular matrix. Proc Natl Acad Sci U S A 103:13180–13185
Willis BC, Liebler JM, Luby-Phelps K, Nicholson AG, Crandall ED et al (2005) Induction of epithelial-mesenchymal transition in alveolar epithelial cells by transforming growth factor beta1: potential role in idiopathic pulmonary fibrosis. Am J Pathol 166:1321–1332
Nieto MA (2013) Epithelial plasticity: a common theme in embryonic and cancer cells. Science 342:1234850
Nabhan AN, Brownfield DG, Harbury PB, Krasnow MA, Desai TJ (2018) Single-cell Wnt signaling niches maintain stemness of alveolar type 2 cells. Science 359:1118–1123
Peng T, Frank DB, Kadzik RS, Morley MP, Rathi KS et al (2015) Hedgehog actively maintains adult lung quiescence and regulates repair and regeneration. Nature 526:578–582
Tsao PN, Matsuoka C, Wei SC, Sato A, Sato S, Hasegawa K et al (2016) Epithelial notch signaling regulates lung alveolar morphogenesis and airway epithelial integrity. Proc Natl Acad Sci U S A 113:8242–8247
Xu K, Nieuwenhuis E, Cohen BL, Wang W, Canty AG et al (2010) Lunatic fringe-mediated notch signaling is required for lung alveogenesis. Am J Physiol Lung Cell Mol Physiol 298:L45–L56
Selman M, Pardo A (2019) The leading role of epithelial cells in the pathogenesis of idiopathic pulmonary fibrosis. Cell Signal 66:109482
Bueno M, Zank D, Buendia-Roldan I, Fiedler K, Mays BG et al (2019) PINK1 attenuates mtDNA release in alveolar epithelial cells and TLR9 mediated profibrotic responses. PLoS One 14:e0218003
Raredon MSB, Adams TS, Suhail Y, Schupp JC, Poli S et al (2019) Single-cell connectomic analysis of adult mammalian lungs. Sci Adv 5:3851
Wang C, Cassandras M, Peng T (2019) The role of hedgehog signaling in adult lung regeneration and maintenance. J Dev Biol 7:14
Hu B, Liu J, Wu Z, Liu T, Ullenbruch MR et al (2015) Reemergence of hedgehog mediates epithelial-mesenchymal crosstalk in pulmonary fibrosis. Am J Respir Cell Mol Biol 52:418–428
Moore BB, Peters-Golden M, Christensen PJ, Lama V, Kuziel WA et al (2003) Alveolar epithelial cell inhibition of fibroblast proliferation is regulated by MCP-1/CCR2 and mediated by PGE2. Am J Physiol Lung Cell Mol Physiol 284:L342–L349
Tan Q, Ma XY, Liu W, tal MJA e (2019) Nascent lung organoids reveal epithelium- and Bone morphogenetic protein-mediated suppression of fibroblast activation. Am J Respir Cell Mol Biol 61:607–619
Jiang C, Huang H, Liu J, Wang Y, Lu Z et al (2012) Fasudil, a rho-kinase inhibitor, attenuates bleomycin-induced pulmonary fibrosis in mice. Int J Mol Sci 13:8293–8307
Bei Y, Hua-Huy T, Duong-Quy S, Nguyen V, Chen W et al (2013) Long-term treatment with fasudil improves bleomycin-induced pulmonary fibrosis and pulmonary hypertension via inhibition of Smad2/3 phosphorylation. Pulm Pharmacol Ther 26:635–643
Knipe RS, Probst CK, Lagares D, Franklin A, Spinney J et al (2018) The rho kinase isoforms ROCK1 and ROCK2 each contribute to the development of experimental pulmonary fibrosis. Am J Respir Cell Mol Biol 58:471–481
Barry-Hamilton V, Spangler R, Marshall D, McCauley S, Rodiguez HM et al (2010) Allosteric inhibition of lysyl oxidase-like-2 impedes the development of a pathologic microenvironment. Nat Med 16:1009–1017
Gokey JJ, Sridharan A, Xu Y, Green J, Carraro G et al (2018) Active epithelial Hippo signaling in idiopathic pulmonary fibrosis. JCI Insight 3:e98738
Axell-House VYDB, Zhang Z, Burdick MD, Strieter RM, Mehrad B (2018) Fibrocytes in pulmonary fibrosis: double-blind placebo-controlled crossover pilot study of Sirolimus in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 197:A4353
Peter E, Huber SB, Peter P, Alexandra T, Ute W et al (2010) Reversal of established fibrosis by treatment with the anti-CTGF monoclonal antibody FG-3019 in a murine model of radiation-induced pulmonary fibrosis. Am J Respir Crit Care Med 181:A1054
Raghu G, Scholand MB, de Andrade J, Lancaster L, Mageto Y et al (2016) FG-3019 anti-connective tissue growth factor monoclonal antibody: results of an open-label clinical trial in idiopathic pulmonary fibrosis. Eur Respir J 47:1481–1491
Melboucy-Belkhir S, Pradere P, Tadbiri S, Habib S, Baccrat A et al (2014) Forkhead box F1 represses cell growth and inhibits COL1 and ARPC2 expression in lung fibroblasts in vitro. Am J Physiol Lung Cell Mol Physiol 307:L838–L847
Shi M, Shu J, Wang R, Chen X, Mi L et al (2011) Latent TGF-beta structure and activation. Nature 474:343–349
Henderson NC, Arnold TD, Katamura Y, Giacomini MM, Rodriguez JD et al (2013) Targeting of alpha integrin identifies a core molecular pathway that regulates fibrosis in several organs. Nat Med 19:1617–1624
Tager AM, LaCamera P, Shea BS, Campanella GS, Selman M et al (2008) The lysophosphatidic acid receptor LPA1 links pulmonary fibrosis to lung injury by mediating fibroblast recruitment and vascular leak. Nat Med 14:45–54
Zhu Z, Homer RJ, Wang Z, Chen Q, Geba GP (1999) Pulmonary expression of interleukin-13 causes inflammation, mucus hypersecretion, subepithelial fibrosis, physiologic abnormalities, and eotaxin production. J Clin Invest 103:779–788
Belperio JA, Dy M, Burdick MD, Xue YY, Li K et al (2002) Interaction of IL-13 and C10 in the pathogenesis of bleomycin-induced pulmonary fibrosis. Am J Respir Cell Mol Biol 27:419–427
Murray LA, Zhang H, Oak SR, Coelho AL, Herath A et al (2014) Targeting interleukin-13 with tralokinumab attenuates lung fibrosis and epithelial damage in a humanized SCID idiopathic pulmonary fibrosis model. Am J Respir Cell Mol Biol 50:985–994
Saito S, Zhuang Y, Shan B, Danchuk S et al (2017) Tubastatin ameliorates pulmonary fibrosis by targeting the TGFβ-PI3K-Akt pathway. PLoS One 12:e0186615
Sellares J, Veraldi KL, Thiel KJ et al (2019) Intracellular heat shock protein 70 deficiency in pulmonary fibrosis. Am J Respir Cell Mol Biol 60:629–636
Agarwal SK (2014) Integrins and cadherins as therapeutic targets in fibrosis. Front Pharmacol 5:1–7
Annes JP, Chen Y, Munger JS, Rifkin DB (2004) Integrin alphaVbeta6-mediated activation of latent TGF-beta requires the latent TGF-beta binding protein-1. J Cell Biol 165:723–734
Morris DG, Huang X, Kaminski N, Wang Y, Shapiro SD et al (2003) Loss of integrin alpha (v) beta6-mediated TGF-beta activation causes Mmp12-dependent emphysema. Nature 422:169–173
Craig VJ, Zhang L, Hagood JS, Owen CA (2015) Matrix metalloproteinases as therapeutic targets for idiopathic pulmonary fibrosis. Am J Respir Cell Mol Biol 53:585–600
Kolb C, Mauch S, Peter HH, Krawinkel U, Sedlacek R (1997) The matrix metalloproteinase RASI-1 is expressed in synovial blood vessels of a rheumatoid arthritis patient. Immunol Lett 57:83–88
Sadowski T, Dietrich S, Koschinsky F, Ludwig A, Proksch E et al (2005) Matrix metalloproteinase 19 processes the laminin 5 gamma 2 chain and induces epithelial cell migration. Cell Mol Life Sci 62:870–880
Carey WA, Taylor GD, Dean WB, Bristow JD (2010) Tenascin-C deficiency attenuates TGF-s mediated fibrosis following murine lung injury. Am J Physiol Lung Cell Mol Physiol 299:L785–L793
Jara P, Calyeca J, Romero Y, Placido L, Yu G et al (2015) Matrix metalloproteinase (MMP)-19-deficient fibroblasts display a profibrotic phenotype. Am J Physiol Lung Cell Mol Physiol 308:L511–L522
Yu G, Kovkarova-Naumovski E, Jara P, Parwani A, Kass D et al (2012) Matrix metalloproteinase-19 is a key regulator of lung fibrosis in mice and humans. Am J Respir Crit Care Med 186:752–762
Smith WL, Langenbach R (2001) Why there are two cyclooxygenase isozymes. J Clin Invest 107:1491–1495
White ES, Atrasz RG, Dickie EG, Aronoff DM, Stambolic V, Mak TW (2005) Prostaglandin E(2) inhibits fibroblast migration by E-prostanoid 2 receptor-mediated increase in PTEN activity. Am J Respir Cell Mol Biol 32:135–141
Anderson RG (1998) The caveolae membrane system. Annu Rev Biochem 67:199–225
Okamoto T, Schlegel A, Scherer PE, Lisanti MP (1998) Caveolins, a family of scaffolding proteins for organizing "preassembled signaling complexes" at the plasma membrane. J Biol Chem 273:5419–5422
Labrecque L, Royal I, Surprenant DS, Patterson C, Gingras D, Beliveau R (2003) Regulation of vascular endothelial growth factor receptor-2 activity by caveolin-1 and plasma membrane cholesterol. Mol Biol Cell 14:334–347
Wang XM, Zhang Y, Kim HP, Zhou Z, Feghali-Bostwick CA et al (2006) Caveolin-1: a critical regulator of lung fibrosis in idiopathic pulmonary fibrosis. J Exp Med 203:2895–2906
Shivshankar P, Brampton C, Miyasato S, Kasper M, Thannickal VJ et al (2012) Caveolin- 1 deficiency protects from pulmonary fibrosis by modulating epithelial cell senescence in mice. Am J Respir Cell Mol Biol 47:28–36
Marudamuthu AS, Bhandary YP, Fan L, Radhakrishnan V, MacKenzie B et al (2019) Caveolin-1–derived peptide limits development of pulmonary fibrosis. Caveolin-1–derived peptide limits development of pulmonary fibrosis. Sci Transl Med 11:2848
Pasterkamp RJ, Peschon JJ, Spriggs MK, Kolodkin AL (2003) Semaphorin 7A promotes axonoutgrowth through integrins and MAPKs. Nature 424:398–405
Kang HR, Lee CG, Homer RJ, Elias JA (2007) Semaphorin 7A plays a critical role in TGF-beta1-induced pulmonary fibrosis. J Exp Med 204:1083–1093
Reilkoff RA, Peng H, Murray LA, Peng X, Russell T et al (2013) Semaphorin 7a+ regulatory T cells are associated with progressive idiopathic pulmonary fibrosis and are implicated in transforming growth factor-beta1-induced pulmonary fibrosis. Am J Respir Crit Care Med 187:180–188
Roach KM, Duffy SM, Coward W, Feghali-Bostwick C, Wulff H (2013) The K+channel KCa3.1 as a novel target for idiopathic pulmonary fibrosis. PLoS One 8:e85244
Roach KM, Wulff H, Feghali-Bostwick C, Amrani Y, Bradding P (2014) Increased constitutive alphaSMA and Smad2/3 expression in idiopathic pulmonary fibrosis myofibroblasts is KCa3.1- dependent. Respir Res 15:155
Roach KM, Feghali-Bostwick C, Wulff H, Amrani Y, Bradding P (2015) Human lung myofibroblast TGFbeta1-dependent Smad2/3 signalling is ca(2+)-dependent and regulated by KCa3.1 K(+) channels. Fibrogenesis Tissue Repair 8:5
Liang H, Xu C, Pan Z, Zhang Y, Xu Z et al (2014) The antifibrotic effects and mechanisms of microRNA-26a action in idiopathic pulmonary fibrosis. Mol Ther J Am Soc Gene Ther 22:1122–1133
Pandit KV, Milosevic J (2015) MicroRNA regulatory networks in idiopathic pulmonary fibrosis. Biochem Cell Biol 93:129–137
Pandit KV, Milosevic J, Kaminski N (2011) MicroRNAs in idiopathic pulmonary fibrosis. Transl Res J Clin Med 157:191–199
Pandit KV, Corcoran D, Yousef H, Yarlagadda M, Tzouvelekis A et al (2010) Inhibition and role of let-7d in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 182:220–229
Lorand L, Graham RM (2003) Transglutaminases: crosslinking enzymes with pleiotropic functions. Nat Rev Mol Cell Biol 4:140–156
Olsen KC, Sapinoro RE, Kottmann RM, Kulkarni AA, Iismaa SE et al (2011) Transglutaminase 2 and its role in pulmonary fibrosis. Am J Respir Crit Care Med 184:699–707
Akimov SS, Belkin AM (2001) Cell-surface transglutaminase promotes fibronectin assembly via interaction with the gelatin-binding domain of fibronectin: a role in TGF beta-dependent matrix deposition. J Cell Sci 114:2989–3000
Spagnolo P, Wells AU, Collard HR (2015) Pharmacological treatment of idiopathic pulmonary fibrosis: an update. Drug Discov Today 20:514–524
du Bois RM (2010) Strategies for treating idiopathic pulmonary fibrosis. Nat Rev Drug Discov 9:129–140
Taniguchi H, Ebina M, Kondoh Y, Ogura T, Azuma A et al (2010) Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J 35:821–829
King TE Jr, Bradford WZ, Castro-Bernardini S, Fagan EA, Glaspole I et al (2014) A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med 370:2083–2092
Hilberg F, Roth GJ, Krssak M, Kautschitsch S, Sommergruber W et al (2008) BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res 68:4774–4782
Richeldi L, du Bois RM, Raghu G, Azuma A, Brown KK et al (2014) Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med 370:2071–2082
Spagnolo P, Rossi G, Cavazza A (2014) Pathogenesis of idiopathic pulmonary fibrosis and its clinical implications. Expert Rev Clin Immunol 10:1005–1017
Caminati A, Lonati C, Cassandro R, Elia D, Pelosi G, Torre O, Zompatori M, Uslenghi E, Harari S (2019) Comorbidities in idiopathic pulmonary fibrosis: an underestimated issue. Eur Respir Rev 28:190044
Alsafadi HN, Staab-Weijnitz CA, Lehmann M, Lindner M, Peschel B et al (2017) An ex vivo model to induce early fibrosis-like changes in human precision-cut lung slices. Am J Physiol Lung Cell Mol Physiol 312:L896–L902
Konar D, Devarasetty M, Yildiz DV, Atala A, Murphy SV (2016) Lung-on-a-chip technologies for disease modeling and drug development. Biomed Eng Comput Biol 7:17–27
Tamò L, Hibaoui Y, Kallol S, Alves MP, Albrecht C et al (2018) Generation of an alveolar epithelial type II cell line from induced pluripotent stem cells. Am J Physiol Lung Cell Mol Physiol 315:L921–L932
Acknowledgments
The work was supported in part by TRR fund, SASTRA Deemed-to-be University, Thanjavur, Tamilnadu, India.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Sivanantham, B., Bose, V. (2021). Targeting Molecular and Cellular Mechanisms in Idiopathic Pulmonary Fibrosis. In: Dua, K., Löbenberg, R., Malheiros Luzo, Â.C., Shukla, S., Satija, S. (eds) Targeting Cellular Signalling Pathways in Lung Diseases. Springer, Singapore. https://doi.org/10.1007/978-981-33-6827-9_12
Download citation
DOI: https://doi.org/10.1007/978-981-33-6827-9_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-33-6826-2
Online ISBN: 978-981-33-6827-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)