Abstract
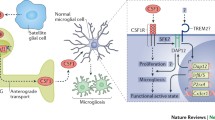
Injury to the nervous system often causes a debilitating chronic pain syndrome, termed neuropathic pain. Neuropathic pain is refractory to currently available analgesics. Accumulating evidence indicates that spinal microglia react and undergo a series of changes that influence directly establishing the pain. P2X4 receptor (P2X4R), a subtype of ionotropic ATP receptors, is upregulated in spinal microglia after nerve injury by several factors including the CC chemokine CCL21 derived from damaged neurons, CC chemokine receptor CCR2, the extracellular matrix protein fibronectin in the spinal cord, and the transcription factor interferon regulatory factor 8 and 5 expressed in microglia. The inhibition of the function of P2X4R and P2X4R-regulating molecules suppresses the excitability of dorsal horn neurons and neuropathic pain. These findings indicate that microglia overexpressing P2X4R are a central player in neuropathic pain.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Beggs S, Liu XJ, Kwan C et al (2010) Peripheral nerve injury and TRPV1-expressing primary afferent C-fibers cause opening of the blood-brain barrier. Mol Pain 6:74
Biber K, Tsuda M, Tozaki-Saitoh H et al (2011) Neuronal CCL21 up-regulates microglia P2X4 expression and initiates neuropathic pain development. EMBO J 30:1864–1873
Cavaliere F, Florenzano F, Amadio S et al (2003) Up-regulation of P2X2, P2X4 receptor and ischemic cell death: prevention by P2 antagonists. Neuroscience 120:85–98
Costigan M, Scholz J, Woolf CJ (2009) Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci 32:1–32
Coull JA, Beggs S, Boudreau D et al (2005) BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 438:1017–1021
Davalos D, Grutzendler J, Yang G et al (2005) ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci 8:752–758
Echeverry S, Shi XQ, Rivest S et al (2011) Peripheral nerve injury alters blood-spinal cord barrier functional and molecular integrity through a selective inflammatory pathway. J Neurosci 31:10819–10828
Ferrini F, Trang T, Mattioli TA et al (2013) Morphine hyperalgesia gated through microglia-mediated disruption of neuronal Cl(−) homeostasis. Nat Neurosci 16:183–192
Ginhoux F, Greter M, Leboeuf M et al (2010) Fate map** analysis reveals that adult microglia derive from primitive macrophages. Science 330:841–845
Guo LH, Schluesener HJ (2005) Lesional accumulation of P2X(4) receptor(+) macrophages in rat CNS during experimental autoimmune encephalomyelitis. Neuroscience 134:199–205
Guo LH, Trautmann K, Schluesener HJ (2004) Expression of P2X4 receptor in rat C6 glioma by tumor-associated macrophages and activated microglia. J Neuroimmunol 152:67–72
Horiuchi M, Wakayama K, Itoh A et al (2012) Interferon regulatory factor 8/interferon consensus sequence binding protein is a critical transcription factor for the physiological phenotype of microglia. J Neuroinflammation 9:227
Inoue K (2006) The function of microglia through purinergic receptors: neuropathic pain and cytokine release. Pharmacol Ther 109:210–226
Katsura H, Obata K, Mizushima T et al (2006) Activation of Src-family kinases in spinal microglia contributes to mechanical hypersensitivity after nerve injury. J Neurosci 26:8680–8690
Kierdorf K, Erny D, Goldmann T, Sander V et al (2013) Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat Neurosci 16:273–280
Masuda T, Tsuda M, Yoshinaga R et al (2012) IRF8 is a critical transcription factor for transforming microglia into a reactive phenotype. Cell Rep 1:334–340
McMahon SB, Malcangio M (2009) Current challenges in glia-pain biology. Neuron 64:46–54
Minten C, Terry R, Deffrasnes C et al (2012) IFN regulatory factor 8 is a key constitutive determinant of the morphological and molecular properties of microglia in the CNS. PLoS One 7, e49851
Nasu-Tada K, Koizumi S, Tsuda M et al (2006) Possible involvement of increase in spinal fibronectin following peripheral nerve injury in upregulation of microglial P2X(4), a key molecule for mechanical allodynia. Glia 53:769–775
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Pocock JM, Kettenmann H (2007) Neurotransmitter receptors on microglia. Trends Neurosci 30:527–535
Ren K, Dubner R (2010) Interactions between the immune and nervous systems in pain. Nat Med 16:1267–1276
Salter MW, Kalia LV (2004) Src kinases: a hub for NMDA receptor regulation. Nat Rev Neurosci 5:317–328
Schwab JM, Guo L, Schluesener HJ (2005) Spinal cord injury induces early and persistent lesional P2X4 receptor expression. J Neuroimmunol 163:185–189
Suter MR, Wen YR, Decosterd I et al (2007) Do glial cells control pain? Neuron Glia Biol 3:255–268
Tamura T, Yanai H, Savitsky D et al (2008) The IRF family transcription factors in immunity and oncogenesis. Annu Rev Immunol 26:535–584
Trang T, Beggs S, Wan X et al (2009) P2X4-receptor-mediated synthesis and release of brain-derived neurotrophic factor in microglia is dependent on calcium and p38-mitogen-activated protein kinase activation. J Neurosci 29:3518–3528
Tsuda M, Shigemoto-Mogami Y, Koizumi S et al (2003) P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424:778–783
Tsuda M, Inoue K, Salter MW (2005) Neuropathic pain and spinal microglia: a big problem from molecules in “small” glia. Trends Neurosci 28:101–107
Tsuda M, Toyomitsu E, Komatsu T et al (2008a) Fibronectin/integrin system is involved in P2X(4) receptor upregulation in the spinal cord and neuropathic pain after nerve injury. Glia 56:579–585
Tsuda M, Tozaki-Saitoh H, Masuda T et al (2008b) Lyn tyrosine kinase is required for P2X(4) receptor upregulation and neuropathic pain after peripheral nerve injury. Glia 56:50–58
Tsuda M, Kuboyama K, Inoue T et al (2009a) Behavioral phenotypes of mice lacking purinergic P2X4 receptors in acute and chronic pain assays. Mol Pain 5:28
Tsuda M, Masuda T, Kitano J et al (2009b) IFN-gamma receptor signaling mediates spinal microglia activation driving neuropathic pain. Proc Natl Acad Sci U S A 106:8032–8037
Tsuda M, Toyomitsu E, Kometani M et al (2009c) Mechanisms underlying fibronectin-induced upregulation of P2XR expression in microglia: distinct roles of PI3K-Akt and MEK-ERK signaling pathways. J Cell Mol Med 13:3251–3259
Tsuda M, Tozaki-Saitoh H, Inoue K (2012) Purinergic system, microglia and neuropathic pain. Curr Opin Pharmacol 12:74–79
Tsuda M, Masuda T, Tozaki-Saitoh H et al (2013) Microglial regulation of neuropathic pain. J Pharmacol Sci 121:89–94
Ulmann L, Hatcher JP, Hughes JP et al (2008) Up-regulation of P2X4 receptors in spinal microglia after peripheral nerve injury mediates BDNF release and neuropathic pain. J Neurosci 28:11263–11268
Ulmann L, Levavasseur F, Avignone E et al (2013) Involvement of P2X4 receptors in hippocampal microglial activation after status epilepticus. Glia 61:1306–1319
Watkins LR, Milligan ED, Maier SF (2001) Glial activation: a driving force for pathological pain. Trends Neurosci 24:450–455
Woolf CJ, Salter MW (2000) Neuronal plasticity: increasing the gain in pain. Science 288:1765–1769
Zhang Z, Zhang Z, Artelt M et al (2007) Dexamethasone attenuates early expression of three molecules associated with microglia/macrophages activation following rat traumatic brain injury. Acta Neuropathol (Berl) 113(6):675–682
Zhang Z, Zhang Z, Fauser U et al (2008) Mechanical allodynia and spinal up-regulation of P2X4 receptor in experimental autoimmune neuritis rats. Neuroscience 152:495–501
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Inoue, K., Tsuda, M. (2016). The Research for the Mechanism of Chronically Intractable Pain Based on the Functions of Microglia as Brain Immunocompetent Cell. In: Miyasaka, M., Takatsu, K. (eds) Chronic Inflammation. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56068-5_48
Download citation
DOI: https://doi.org/10.1007/978-4-431-56068-5_48
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56066-1
Online ISBN: 978-4-431-56068-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)