Abstract
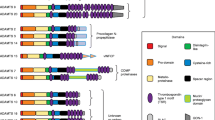
Rheumatoid arthritis (RA) and osteoarthritis (OA) are the two most common joint disorders, causing pain and disability to a significant proportion of the human population. Although the etiology of the two diseases is different, a common feature is the destruction of articular cartilage, which is largely due to the elevated proteolytic enzyme activities that degrade extracellular matrix (ECM) macromolecules in the cartilage. A number of proteinases, including serine and cysteine proteinases are produced in various tissues and cells in the articular joints, but the major proteinases that degrade cartilage matrices are the matrix metalloproteinases (MMPs) and the metallproteinases with disintegrin and thrombospondin domains (ADAMTs). In addition, the metalloproteinases with a disintegrin domain (ADAMs) that shed cell surface cytokines, growth factor and their receptors play important roles in inflammatory process. The activities of these metalloproteinases are regulated by endogenous tissue inhibitors of metalloproteinases called TIMPs. This chapter introduces the members of these metalloproteinases, and discuss their structures and how they are involved in cartilage matrix degradation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abbaszade I, Liu RQ, Yang F, Rosenfeld SA, Ross OH, Link JR, Ellis DM, Tortorella MD, Pratta MA, Hollis JM, Wynn R, Duke JL, George HJ, Hillman MC Jr, Murphy K, Wiswall BH, Copeland RA, Decicco CP, Bruckner R, Nagase H, Itoh Y, Newton RC, Magolda RL, Trzaskos JM, Burn TC (1999) Cloning and characterization of ADAMTS11, an aggrecanase from the ADAMTS family. J Biol Chem 274:23443–23450
Adrain C, Zettl M, Christova Y, Taylor N, Freeman M (2012) Tumor necrosis factor signaling requires iRhom2 to promote trafficking and activation of TACE. Science 335:225–228
Aigner T, Soder S, Gebhard PM, McAlinden A, Haag J (2007) Mechanisms of disease: role of chondrocytes in the pathogenesis of osteoarthritis–structure, chaos and senescence. Nat Clin Pract Rheumatol 3:391–399
Akiyama M, Takeda S, Kokame K, Takagi J, Miyata T (2009) Crystal structures of the noncatalytic domains of ADAMTS13 reveal multiple discontinuous exosites for von Willebrand factor. Proc Natl Acad Sci U S A 106:19274–19279
Amour A, Slocombe PM, Webster A, Butler M, Knight CG, Smith BJ, Stephens PE, Shelley C, Hutton M, Knäuper V, Docherty AJP, Murphy G (1998) TNF-Alpha converting enzyme (TACE) is inhibited by TIMP-3. FEBS Lett 435:39–44
Barrett AJ (1981) α2-Macroglobulin. Methods Enzymol 80:737–754
Barrett AJ, Starkey PM (1973) The interaction of α2-macroglobulin with proteinases. Characteristics and specificity of the reaction, and a hypothesis concerning its molecular mechanism. Biochem J 133:709–724
Basu B, Correa de Sampaio P, Mohammed H, Fogarasi M, Corrie P, Watkins NA, Smethurst PA, English WR, Ouwehand WH, Murphy G (2012) Inhibition of MT1-MMP activity using functional antibody fragments selected against its hemopexin domain. Int J Biochem Cell Biol 44:393–403
Bau B, Gebhard PM, Haag J, Knorr T, Bartnik E, Aigner T (2002) Relative messenger RNA expression profiling of collagenases and aggrecanases in human articular chondrocytes in vivo and in vitro. Arthritis Rheum 46:2648–2657
Becherer JD, Blobel CP (2003) Biochemical properties and functions of membrane-anchored metalloprotease-disintegrin proteins (ADAMs). Curr Top Dev Biol 54:101–123
Becker JW, Marcy AI, Rokosz LL, Axel MG, Burbaum JJ, Fitzgerald PM, Cameron PM, Esser CK, Hagmann WK, Hermes JD et al (1995) Stromelysin-1: three-dimensional structure of the inhibited catalytic domain and of the C-truncated proenzyme. Protein Sci 4:1966–1976
Benito MJ, Veale DJ, FitzGerald O, van den Berg WB, Bresnihan B (2005) Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis 64:1263–1267
Bertini I, Calderone V, Fragai M, Jaiswal R, Luchinat C, Melikian M, Mylonas E, Svergun DI (2008) Evidence of reciprocal reorientation of the catalytic and hemopexin-like domains of full-length MMP-12. J Am Chem Soc 130:7011–7021
Bertini I, Fragai M, Luchinat C, Melikian M, Mylonas E, Sarti N, Svergun DI (2009) Interdomain flexibility in full-length matrix metalloproteinase-1 (MMP-1). J Biol Chem 284:12821–12828
Bertini I, Fragai M, Luchinat C, Melikian M, Toccafondi M, Lauer JL, Fields GB (2012) Structural basis for matrix metalloproteinase 1-catalyzed collagenolysis. J Am Chem Soc 134:2100–2110
Blain EJ, Gilbert SJ, Wardale RJ, Capper SJ, Mason DJ, Duance VC (2001) Up-regulation of matrix metalloproteinase expression and activation following cyclical compressive loading of articular cartilage in vitro. Arch Biochem Biophys 396:49–55
Bode W, Gomis-Rüth FX, Stöcker W (1993) Astacins, serralysins, snake venom and matrix metalloproteinases exhibit identical zinc-binding environments (HEXXHXXGXXH and Met-turn) and topologies and should be grouped into a common family, the ‘metzincins’. FEBS Lett 331:134–140
Bode W (1995) A hel** hand for collagenases: the haemopexin-like domain. Structure 3:527–530
Böhm BB, Aigner T, Blobel CP, Kalden JR, Burkhardt H (2001) Highly enhanced expression of the disintegrin metalloproteinase MDC15 (metargidin) in rheumatoid synovial tissue. Arthritis Rheum 44:2046–2054
Böhm BB, Aigner T, Roy B, Brodie TA, Blobel CP, Burkhardt H (2005) Homeostatic effects of the metalloproteinase disintegrin ADAM15 in degenerative cartilage remodeling. Arthritis Rheum 52:1100–1109
Böhm B, Hess S, Krause K, Schirner A, Ewald W, Aigner T, Burkhardt H (2010) ADAM15 exerts an antiapoptotic effect on osteoarthritic chondrocytes via up-regulation of the X-linked inhibitor of apoptosis. Arthritis Rheum 62:1372–1382
Boldt HB, Kjaer-Sorensen K, Overgaard MT, Weyer K, Poulsen CB, Sottrup-Jensen L, Conover CA, Giudice LC, Oxvig C (2004) The Lin12-notch repeats of pregnancy-associated plasma protein-A bind calcium and determine its proteolytic specificity. J Biol Chem 279:38525–38531
Bolz H, Ramirez A, von Brederlow B, Kubisch C (2001) Characterization of ADAMTS14, a novel member of the ADAMTS metalloproteinase family. Biochim Biophys Acta 1522:221–225
Brew K, Nagase H (2010) The tissue inhibitors of metalloproteinases (TIMPs): an ancient family with structural and functional diversity. Biochim Biophys Acta 1803:55–71
Chung L, Dinakarpandian D, Yoshida N, Lauer-Fields JL, Fields GB, Visse R, Nagase H (2004) Collagenase unwinds triple-helical collagen prior to peptide bond hydrolysis. EMBO J 23:3020–3030
Colige A, Li SW, Sieron AL, Nusgens BV, Prockop DJ, Lapière CM (1997) cDNA cloning and expression of bovine procollagen I N-proteinase: a new member of the superfamily of zinc-metalloproteinases with binding sites for cells and other matrix components. Proc Natl Acad Sci U S A 94:2374–2379
Colige A, Vandenberghe I, Thiry M, Lambert CA, Van Beeumen J, Li SW, Prockop DJ, Lapière CM, Nusgens BV (2002) Cloning and characterization of ADAMTS-14, a novel ADAMTS displaying high homology with ADAMTS-2 and ADAMTS-3. J Biol Chem 277:5756–5766
Conway JG, Wakefield JA, Brown RH, Marron BE, Sekut L, Stimpson SA, McElroy A, Menius JA, Jeffreys JJ, Clark RL et al (1995) Inhibition of cartilage and bone destruction in adjuvant arthritis in the rat by a matrix metalloproteinase inhibitor. J Exp Med 182:449–457
Cox JH, Starr AE, Kappelhoff R, Yan R, Roberts CR, Overall CM (2010) Matrix metalloproteinase 8 deficiency in mice exacerbates inflammatory arthritis through delayed neutrophil apoptosis and reduced caspase 11 expression. Arthritis Rheum 62:3645–3655
Dean DD, Martel-Pelletier J, Pelletier JP, Howell DS, Woessner JF Jr (1989) Evidence for metalloproteinase and metalloproteinase inhibitor imbalance in human osteoarthritic cartilage. J Clin Invest 84:678–685
Dodge GR, Poole AR (1989) Immunohistochemical detection and immunochemical analysis of type II collagen degradation in human normal, rheumatoid, and osteoarthritic articular cartilages and in explants of bovine articular cartilage cultured with interleukin 1. J Clin Invest 83:647–661
Durigova M, Nagase H, Mort JS, Roughley PJ (2011) MMPs are less efficient than ADAMTS5 in cleaving aggrecan core protein. Matrix Biol 30:145–153
Edwards DR, Handsley MM, Pennington CJ (2008) The ADAM metalloproteinases. Mol Aspects Med 29:258–289
Elkins PA, Ho YS, Smith WW, Janson CA, D’Alessio KJ, McQueney MS, Cummings MD, Romanic AM (2002) Structure of the C-terminally truncated human ProMMP9, a gelatin-binding matrix metalloproteinase. Acta Crystallogr D Biol Crystallogr 58:1182–1192
Engel CK, Pirard B, Schimanski S, Kirsch R, Habermann J, Klingler O, Schlotte V, Weithmann KU, Wendt KU (2005) Structural basis for the highly selective inhibition of MMP-13. Chem Biol 12:181–189
Fernandes RJ, Hirohata S, Engle JM, Colige A, Cohn DH, Eyre DR, Apte SS (2001) Procollagen II amino propeptide processing by ADAMTS-3. Insights on dermatosparaxis. J Biol Chem 276:31502–31509
Flannery CR, Little CB, Hughes CE, Caterson B (1999) Expression of ADAMTS homologues in articular cartilage. Biochem Biophys Res Commun 260:318–322
Flory ED, Clarris BJ, Muirden KD (1982) Deposits of α2M in the rheumatoid synovial membrane. Ann Rheum Dis 41:520–526
Fosang AJ, Rogerson FM, East CJ, Stanton H (2008) ADAMTS-5: the story so far. Eur Cell Mater 15:11–26
Fried D, Bohm BB, Krause K, Burkhardt H (2012) ADAM15 protein amplifies focal adhesion kinase phosphorylation under genotoxic stress conditions. J Biol Chem 287:21214–21223
Fu X, Kao JL, Bergt C, Kassim SY, Huq NP, d’Avignon A, Parks WC, Mecham RP, Heinecke JW (2004) Oxidative cross-linking of tryptophan to glycine restrains matrix metalloproteinase activity: specific structural motifs control protein oxidation. J Biol Chem 279:6209–6212
Fujii Y, Okuda D, Fujimoto Z, Horii K, Morita T, Mizuno H (2003) Crystal structure of trimestatin, a disintegrin containing a cell adhesion recognition motif RGD. J Mol Biol 332:1115–1122
Fukata Y, Adesnik H, Iwanaga T, Bredt DS, Nicoll RA, Fukata M (2006) Epilepsy-related ligand/receptor complex LGI1 and ADAM22 regulate synaptic transmission. Science 313:1792–1795
Fulcher YG, Van Doren SR (2011) Remote exosites of the catalytic domain of matrix metalloproteinase-12 enhance elastin degradation. Biochemistry 50:9488–9499
Fushimi K, Troeberg L, Nakamura H, Lim NH, Nagase H (2008) Functional differences of the catalytic and non-catalytic domains in human ADAMTS-4 and ADAMTS-5 in aggrecanolytic activity. J Biol Chem 283:6706–6716
Gao G, Plaas A, Thompson VP, ** S, Zuo F, Sandy JD (2004) ADAMTS4 (aggrecanase-1) activation on the cell surface involves C-terminal cleavage by glycosylphosphatidyl inositol-anchored membrane type 4-matrix metalloproteinase and binding of the activated proteinase to chondroitin sulfate and heparan sulfate on syndecan-1. J Biol Chem 279:10042–10051
Garnero P, Borel O, Byrjalsen I, Ferreras M, Drake FH, McQueney MS, Foged NT, Delmas PD, Delaisse JM (1998) The collagenolytic activity of cathepsin K is unique among mammalian proteinases. J Biol Chem 273:32347–32352
Gendron C, Kashiwagi M, Lim NH, Enghild JJ, Thogersen IB, Hughes C, Caterson B, Nagase H (2007) Proteolytic activities of human ADAMTS-5: comparative studies with ADAMTS-4. J Biol Chem 282:18294–18306
Geng Y, McQuillan D, Roughley PJ (2006) SLRP interaction can protect collagen fibrils from cleavage by collagenases. Matrix Biol 25:484–491
Gerhardt S, Hassall G, Hawtin P, McCall E, Flavell L, Minshull C, Hargreaves D, Ting A, Pauptit RA, Parker AE, Abbott WM (2007) Crystal structures of human ADAMTS-1 reveal a conserved catalytic domain and a disintegrin-like domain with a fold homologous to cysteine-rich domains. J Mol Biol 373:891–902
Ghosh P (1999) The pathobiology of osteoarthritis and the rationale for the use of pentosan polysulfate for its treatment. Semin Arthritis Rheum 28:211–267
Glasson SS, Askew R, Sheppard B, Carito BA, Blanchet T, Ma HL, Flannery CR, Kanki K, Wang E, Peluso D, Yang Z, Majumdar MK, Morris EA (2004) Characterization of and osteoarthritis susceptibility in ADAMTS-4-knockout mice. Arthritis Rheum 50:2547–2558
Glasson SS, Askew R, Sheppard B, Carito B, Blanchet T, Ma HL, Flannery CR, Peluso D, Kanki K, Yang Z, Majumdar MK, Morris EA (2005) Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434:644–648
Gomis-Rüth FX (2003) Structural aspects of the metzincin clan of metalloendopeptidases. Mol Biotechnol 24:157–202
Gomis-Rüth FX (2009) Catalytic domain architecture of metzincin metalloproteases. J Biol Chem 284:15353–15357
Gomis-Rüth FX, Kress LF, Bode W (1993) First structure of a snake venom metalloproteinase: a prototype for matrix metalloproteinases/collagenases. EMBO J 12:4151–4157
Gomis-Rüth FX, Gohlke U, Betz M, Knauper V, Murphy G, López-Otìn C, Bode W (1996) The hel** hand of collagenase-3 (MMP-13): 2.7 Å crystal structure of its C-terminal haemopexin-like domain. J Mol Biol 264:556–566
Gomis-Rüth FX, Maskos K, Betz M, Bergner A, Huber R, Suzuki K, Yoshida N, Nagase H, Brew K, Bourenkov GP, Bartunik H, Bode W (1997) Mechanism of inhibition of the human matrix metalloproteinase stromelysin-1 by TIMP-1. Nature 389:77–81
Gonzales PE, Solomon A, Miller AB, Leesnitzer MA, Sagi I, Milla ME (2004) Inhibition of the tumor necrosis factor-alpha-converting enzyme by its pro domain. J Biol Chem 279:31638–31645
Gorovoy M, Gaultier A, Campana WM, Firestein GS, Gonias SL (2010) Inflammatory mediators promote production of shed LRP1/CD91, which regulates cell signaling and cytokine expression by macrophages. J Leukoc Biol 88:769–778
Goulas T, Arolas JL, Gomis-Rüth FX (2011) Structure, function and latency regulation of a bacterial enterotoxin potentially derived from a mammalian adamalysin/ADAM xenolog. Proc Natl Acad Sci U S A 108:1856–1861
Gould RJ, Polokoff MA, Friedman PA, Huang TF, Holt JC, Cook JJ, Niewiarowski S (1990) Disintegrins: a family of integrin inhibitory proteins from viper venoms. Proc Soc Exp Biol Med 195:168–171
Grams F, Reinemer P, Powers JC, Kleine T, Pieper M, Tschesche H, Huber R, Bode W (1995) X-ray structures of human neutrophil collagenase complexed with peptide hydroxamate and peptide thiol inhibitors. Implications for substrate binding and rational drug design. Eur J Biochem 228:830–841
Grimaud E, Heymann D, Redini F (2002) Recent advances in TGF-β effects on chondrocyte metabolism. Potential therapeutic roles of TGF-beta in cartilage disorders. Cytokine Growth Factor Rev 13:241–257
Gu Z, Kaul M, Yan B, Kridel SJ, Cui J, Strongin A, Smith JW, Liddington RC, Lipton SA (2002) S-nitrosylation of matrix metalloproteinases: signaling pathway to neuronal cell death. Science 297:1186–1190
Hadler-Olsen E, Fadnes B, Sylte I, Uhlin-Hansen L, Winberg JO (2011) Regulation of matrix metalloproteinase activity in health and disease. FEBS J 278:28–45
Hamze AB, Wei S, Bahudhanapati H, Kota S, Acharya KR, Brew K (2007) Constraining specificity in the N-domain of tissue inhibitor of metalloproteinases-1; gelatinase-selective inhibitors. Protein Sci 16:1905–1913
Heinegård D, Saxne T (2011) The role of the cartilage matrix in osteoarthritis. Nat Rev Rheumatol 7:50–56
Higashi S, Miyazaki K (1999) Reactive site-modified tissue inhibitor of metalloproteinases-2 inhibits the cell-mediated activation of progelatinase A. J Biol Chem 274:10497–10504
Homandberg GA (1999) Potential regulation of cartilage metabolism in osteoarthritis by fibronectin fragments. Front Biosci 4:D713–D730 [Review] [151 refs]
Hurskainen TL, Hirohata S, Seldin MF, Apte SS (1999) ADAM-TS5, ADAM-TS6, and ADAM-TS7, novel members of a new family of zinc metalloproteases. General features and genomic distribution of the ADAM-TS family. J Biol Chem 274:25555–25563
Igarashi T, Araki S, Mori H, Takeda S (2007) Crystal structures of catrocollastatin/VAP2B reveal a dynamic, modular architecture of ADAM/adamalysin/reprolysin family proteins. FEBS Lett 581:2416–2422
Ilic MZ, Handley CJ, Robinson HC, Mok MT (1992) Mechanism of catabolism of aggrecan by articular cartilage. Arch Biochem Biophys 294:115–122
Ishikawa T, Nishigaki F, Miyata S, Hirayama Y, Minoura K, Imanishi J, Neya M, Mizutani T, Imamura Y, Naritomi Y, Murai H, Ohkubo Y, Kagayama A, Mutoh S (2005) Prevention of progressive joint destruction in collagen-induced arthritis in rats by a novel matrix metalloproteinase inhibitor, FR255031. Br J Pharmacol 144:133–143
Issuree PD, Maretzky T, McIlwain DR, Monette S, Qing X, Lang PA, Swendeman SL, Park-Min KH, Binder N, Kalliolias GD, Yarilina A, Horiuchi K, Ivashkiv LB, Mak TW, Salmon JE, Blobel CP (2013) iRHOM2 is a critical pathogenic mediator of inflammatory arthritis. J Clin Invest 123:928–932
Iyer S, Visse R, Nagase H, Acharya KR (2006) Crystal structure of an active form of human MMP-1. J Mol Biol 362(1):78–88
Janes PW, Saha N, Barton WA, Kolev MV, Wimmer-Kleikamp SH, Nievergall E, Blobel CP, Himanen JP, Lackmann M, Nikolov DB (2005) Adam meets Eph: an ADAM substrate recognition module acts as a molecular switch for ephrin cleavage in trans. Cell 123:291–304
Johnson AR, Pavlovsky AG, Ortwine DF, Prior F, Man CF, Bornemeier DA, Banotai CA, Mueller WT, McConnell P, Yan C, Baragi V, Lesch C, Roark WH, Wilson M, Datta K, Guzman R, Han HK, Dyer RD (2007) Discovery and characterization of a novel inhibitor of matrix metalloprotease-13 (MMP13) that reduces cartilage damage in vivo without joint fibroplasia side effects. J Biol Chem 282(38):27781–27791
Jozic D, Bourenkov G, Lim NH, Visse R, Nagase H, Bode W, Maskos K (2005) X-ray structure of human proMMP-1: new insights into procollagenase activation and collagen binding. J Biol Chem 280:9578–9585
Kashiwagi M, Enghild JJ, Gendron C, Hughes C, Caterson B, Itoh Y, Nagase H (2004) Altered proteolytic activities of ADAMTS-4 expressed by C-terminal processing. J Biol Chem 279:10109–10119
Kevorkian L, Young DA, Darrah C, Donell ST, Shepstone L, Porter S, Brockbank SM, Edwards DR, Parker AE, Clark IM (2004) Expression profiling of metalloproteinases and their inhibitors in cartilage. Arthritis Rheum 50:131–141
Knäuper V, Docherty AJP, Smith B, Tschesche H, Murphy G (1997) Analysis of the contribution of the hinge region of human neutrophil collagenase (HNC, MMP-8) to stability and collagenolytic activity by alanine scanning mutagenesis. FEBS Lett 405:60–64
Komiya K, Enomoto H, Inoki I, Okazaki S, Fujita Y, Ikeda E, Ohuchi E, Matsumoto H, Toyama Y, Okada Y (2005) Expression of ADAM15 in rheumatoid synovium: up-regulation by vascular endothelial growth factor and possible implications for angiogenesis. Arthritis Res Ther 7:R1158–R1173
Koo BH, Longpre JM, Somerville RP, Alexander JP, Leduc R, Apte SS (2006) Cell-surface processing of pro-adams9 by furin. J Biol Chem 281(18):12485–12494
Koo BH, Longpre JM, Somerville RP, Alexander JP, Leduc R, Apte SS (2007) Regulation of ADAMTS9 secretion and enzymatic activity by its propeptide. J Biol Chem 282:16146–16154
Kramer RZ, Bella J, Brodsky B, Berman HM (2001) The crystal and molecular structure of a collagen-like peptide with a biologically relevant sequence. J Mol Biol 311:131–147
Kuno K, Kanada N, Nakashima E, Fujiki F, Ichimura F, Matsushima K (1997) Molecular cloning of a gene encoding a new type of metalloproteinase-disintegrin family protein with thrombospondin motifs as an inflammation associated gene. J Biol Chem 272:556–562
Kuno K, Terashima Y, Matsushima K (1999) ADAMTS-1 is an active metalloproteinase associated with the extracellular matrix. J Biol Chem 274:18821–18826
Lark MW, Bayne EK, Flanagan J, Harper CF, Hoerrner LA, Hutchinson NI, Singer II, Donatelli SA, Weidner JR, Williams HR, Mumford RA, Lohmander LS (1997) Aggrecan degradation in human cartilage – evidence for both matrix metalloproteinase and aggrecanase activity in normal, osteoarthritic, and rheumatoid joints. J Clin Invest 100:93–106
Le Gall SM, Maretzky T, Issuree PD, Niu XD, Reiss K, Saftig P, Khokha R, Lundell D, Blobel CP (2010) ADAM17 is regulated by a rapid and reversible mechanism that controls access to its catalytic site. J Cell Sci 123:3913–3922
Lee MH, Rapti M, Knäuper V, Murphy G (2004a) Threonine 98, the pivotal residue of tissue inhibitor of metalloproteinases (TIMP)-1 in metalloproteinase recognition. J Biol Chem 279:17562–17569
Lee MH, Rapti M, Murphy G (2004b) Delineating the molecular basis of the inactivity of tissue inhibitor of metalloproteinase-2 against tumor necrosis factor-alpha-converting enzyme. J Biol Chem 279:45121–45129
Lee JH, Fitzgerald JB, DiMicco MA, Grodzinsky AJ (2005a) Mechanical injury of cartilage explants causes specific time-dependent changes in chondrocyte gene expression. Arthritis Rheum 52:2386–2395
Lee MH, Rapti M, Murphy G (2005b) Total conversion of tissue inhibitor of metalloproteinase (TIMP) for specific metalloproteinase targeting: fine-tuning TIMP-4 for optimal inhibition of tumor necrosis factor-{alpha}-converting enzyme. J Biol Chem 280:15967–15975
Leonard JD, Lin F, Milla ME (2005) Chaperone-like properties of the prodomain of TNFalpha-converting enzyme (TACE) and the functional role of its cysteine switch. Biochem J 387:797–805
Levy GG, Nichols WC, Lian EC, Foroud T, McClintick JN, McGee BM, Yang AY, Siemieniak DR, Stark KR, Gruppo R, Sarode R, Shurin SB, Chandrasekaran V, Stabler SP, Sabio H, Bouhassira EE, Upshaw JD Jr, Ginsburg D, Tsai HM (2001) Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature 413:488–494
Li J, Brick P, O’Hare MC, Skarzynski T, Lloyd LF, Curry VA, Clark IM, Bigg HF, Hazleman BL, Cawston TE, Blow DM (1995) Structure of full-length porcine synovial collagenase reveals a C-terminal domain containing a calcium-linked, four-bladed beta-propeller. Structure 3:541–549
Libson AM, Gittis AG, Collier IE, Marmer BL, Goldberg GI, Lattman EE (1995) Crystal structure of the haemopexin-like C-terminal domain of gelatinase A [letter]. Nat Struct Biol 2:938–942
Lillis AP, Van Duyn LB, Murphy-Ullrich JE, Strickland DK (2008) LDL receptor-related protein 1: unique tissue-specific functions revealed by selective gene knockout studies. Physiol Rev 88:887–918
Lim NH, Kashiwagi M, Visse R, Jones J, Enghild JJ, Brew K, Nagase H (2010) Reactive-site mutants of N-TIMP-3 that selectively inhibit ADAMTS-4 and ADAMTS-5: biological and structural implications. Biochem J 431:113–122
Little CB, Barai A, Burkhardt D, Smith SM, Fosang AJ, Werb Z, Shah M, Thompson EW (2009) Matrix metalloproteinase 13-deficient mice are resistant to osteoarthritic cartilage erosion but not chondrocyte hypertrophy or osteophyte development. Arthritis Rheum 60:3723–3733
Liu H, Shim AH, He X (2009) Structural characterization of the ectodomain of a disintegrin and metalloproteinase-22 (ADAM22), a neural adhesion receptor instead of metalloproteinase: insights on ADAM function. J Biol Chem 284:29077–29086
Loechel F, Gilpin BJ, Engvall E, Albrechtsen R, Wewer UM (1998) Human ADAM 12 (meltrin alpha) is an active metalloprotease. J Biol Chem 273:16993–16997
Lohmander LS, Neame PJ, Sandy JD (1993) The structure of aggrecan fragments in human synovial fluid. Evidence that aggrecanase mediates cartilage degradation in inflammatory joint disease, joint injury, and osteoarthritis. Arthritis Rheum 36:1214–1222
Longpre JM, Leduc R (2004) Identification of prodomain determinants involved in ADAMTS-1 biosynthesis. J Biol Chem 279:33237–33245
Longpre JM, McCulloch DR, Koo BH, Alexander JP, Apte SS, Leduc R (2009) Characterization of proADAMTS5 processing by proprotein convertases. Int J Biochem Cell Biol 41:1116–1126
Lotta LA, Garagiola I, Palla R, Cairo A, Peyvandi F (2010) ADAMTS13 mutations and polymorphisms in congenital thrombotic thrombocytopenic purpura. Hum Mutat 31:11–19
Loulakis P, Shrikhande A, Davis G, Maniglia CA (1992) N-terminal sequence of proteoglycan fragments isolated from medium of interleukin-1-treated articular-cartilage cultures. Putative site(s) of enzymic cleavage. Biochem J 284:589–593
Luken BM, Turenhout EA, Kaijen PH, Greuter MJ, Pos W, van Mourik JA, Fijnheer R, Voorberg J (2006) Amino acid regions 572-579 and 657-666 of the spacer domain of ADAMTS13 provide a common antigenic core required for binding of antibodies in patients with acquired TTP. Thromb Haemost 96:295–301
Mahmoodi M, Sahebjam S, Smookler D, Khokha R, Mort JS (2005) Lack of tissue inhibitor of metalloproteinases-3 results in an enhanced inflammatory response in antigen-induced arthritis. Am J Pathol 166:1733–1740
Manka SW, Carafoli F, Visse R, Bihan D, Raynal N, Farndale RW, Murphy G, Enghild JJ, Hohenester E, Nagase H (2012) Structural insights into triple-helical collagen cleavage by matrix metalloproteinase 1. Proc Natl Acad Sci U S A 109:12461–12466
Maretzky T, Evers A, Zhou W, Swendeman SL, Wong PM, Rafii S, Reiss K, Blobel CP (2011) Migration of growth factor-stimulated epithelial and endothelial cells depends on EGFR transactivation by ADAM17. Nat Commun 2:229
Maskos K (2005) Crystal structures of MMPs in complex with physiological and pharmacological inhibitors. Biochimie 87:249–263
Maskos K, Fernandez-Catalan C, Huber R, Bourenkov GP, Bartunik H, Ellestad GA, Reddy P, Wolfson MF, Rauch CT, Castner BJ, Davis R, Clarke HR, Petersen M, Fitzner JN, Cerretti DP, March CJ, Paxton RJ, Black RA, Bode W (1998) Crystal structure of the catalytic domain of human tumor necrosis factor-alpha-converting enzyme. Proc Natl Acad Sci U S A 95:3408–3412
McIlwain DR, Lang PA, Maretzky T, Hamada K, Ohishi K, Maney SK, Berger T, Murthy A, Duncan G, Xu HC, Lang KS, Haussinger D, Wakeham A, Itie-Youten A, Khokha R, Ohashi PS, Blobel CP, Mak TW (2012) iRhom2 regulation of TACE controls TNF-mediated protection against Listeria and responses to LPS. Science 335:229–232
Meng Q, Malinovskii V, Huang W, Hu YJ, Chung L, Nagase H, Bode W, Maskos K, Brew K (1999) Residue 2 of TIMP-1 is a major determinant of affinity and specificity for matrix metalloproteinases but effects of substitutions do not correlate with those of the corresponding P1’ residue of substrate. J Biol Chem 274:10184–10189
Miller M-C, Manning HB, Jain A, Troeberg L, Dudhia J, Essex D, Sandison A, Seiki M, Nanchahal J, Nagase H, Itoh Y (2009) Membrane type 1 matrix metalloproteinase is a crucial promoter of synovial invasion in human rheumatoid arthritis. Arthritis Rheum 60:686–697
Milner JM, Cawston TE (2005) Matrix metalloproteinase knockout studies and the potential use of matrix metalloproteinase inhibitors in the rheumatic diseases. Curr Drug Targets Inflamm Allergy 4:363–375
Morgunova E, Tuuttila A, Bergmann U, Isupov M, Lindqvist Y, Schneider G, Tryggvason K (1999) Structure of human pro-matrix metalloproteinase-2: activation mechanism revealed. Science 284:1667–1670
Morgunova E, Tuuttila A, Bergmann U, Tryggvason K (2002) Structural insight into the complex formation of latent matrix metalloproteinase 2 with tissue inhibitor of metalloproteinase 2. Proc Natl Acad Sci U S A 99:7414–7419
Mosyak L, Georgiadis K, Shane T, Svenson K, Hebert T, McDonagh T, Mackie S, Olland S, Lin L, Zhong X, Kriz R, Reifenberg EL, Collins-Racie LA, Corcoran C, Freeman B, Zollner R, Marvell T, Vera M, Sum PE, LaVallie ER, Stahl M, Somers W (2008) Crystal structures of the two major aggrecan degrading enzymes, ADAMTS4 and ADAMTS5. Protein Sci 17:16–21
Nagase H (1997) Activation mechanisms of matrix metalloproteinases. Biol Chem 378:151–160
Nagase H, Itoh Y, Binner S (1994) Interaction of α 2-macroglobulin with matrix metalloproteinases and its use for identification of their active forms. Ann NY Acad Sci 732:294–302
Nagase H, Visse R, Murphy G (2006) Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res 69:562–573
Naito S, Shiomi T, Okada A, Kimura T, Chijiiwa M, Fujita Y, Yatabe T, Komiya K, Enomoto H, Fujikawa K, Okada Y (2007) Expression of ADAMTS4 (aggrecanase-1) in human osteoarthritic cartilage. Pathol Int 57:703–711
Nerenberg PS, Salsas-Escat R, Stultz CM (2008) Do collagenases unwind triple-helical collagen before peptide bond hydrolysis? Reinterpreting experimental observations with mathematical models. Proteins 70:1154–1161
Okada A, Mochizuki S, Yatabe T, Kimura T, Shiomi T, Fujita Y, Matsumoto H, Sehara-Fujisawa A, Iwamoto Y, Okada Y (2008) ADAM-12 (meltrin alpha) is involved in chondrocyte proliferation via cleavage of insulin-like growth factor binding protein 5 in osteoarthritic cartilage. Arthritis Rheum 58:778–789
Orth P, Reichert P, Wang W, Prosise WW, Yarosh-Tomaine T, Hammond G, Ingram RN, **ao L, Mirza UA, Zou J, Strickland C, Taremi SS, Le HV, Madison V (2004) Crystal structure of the catalytic domain of human ADAM33. J Mol Biol 335:129–137
Overall CM (2002) Molecular determinants of metalloproteinase substrate specificity: matrix metalloproteinase substrate binding domains, modules, and exosites. MolBiotechnol 22:51–86
Palmier MO, Fulcher YG, Bhaskaran R, Duong VQ, Fields GB, Van Doren SR (2010) NMR and bioinformatics discovery of exosites that tune metalloelastase specificity for solubilized elastin and collagen triple helices. J Biol Chem 285:30918–30930
Peppin GJ, Weiss SJ (1986) Activation of the endogenous metalloproteinase, gelatinase, by triggered human neutrophils. Proc Natl Acad Sci U S A 83:4322–4326
Perumal S, Antipova O, Orgel JP (2008) Collagen fibril architecture, domain organization, and triple-helical conformation govern its proteolysis. Proc Natl Acad Sci U S A 105:2824–2829
Quinn KA, Grimsley PG, Dai YP, Tapner M, Chesterman CN, Owensby DA (1997) Soluble low density lipoprotein receptor-related protein (LRP) circulates in human plasma. J Biol Chem 272:23946–23951
Ramachandran GN, Kartha G (1955) Structure of collagen. Nature 176:593–595
Reddy P, Slack JL, Davis R, Cerretti DP, Kozlosky CJ, Blanton RA, Shows D, Peschon JJ, Black RA (2000) Functional analysis of the domain structure of tumor necrosis factor-alpha converting enzyme. J Biol Chem 275:14608–14614
Richards FM, Tape CJ, Jodrell DI, Murphy G (2012) Anti-tumour effects of a specific anti-ADAM17 antibody in an ovarian cancer model in vivo. PLoS One 7:e40597
Rosenblum G, Van den Steen PE, Cohen SR, Grossmann JG, Frenkel J, Sertchook R, Slack N, Strange RW, Opdenakker G, Sagi I (2007) Insights into the structure and domain flexibility of full-length pro-matrix metalloproteinase-9/gelatinase B. Structure 15:1227–1236
Rousseau S, Papoutsopoulou M, Symons A, Cook D, Lucocq JM, Prescott AR, O’Garra A, Ley SC, Cohen P (2008) TPL2-mediated activation of ERK1 and ERK2 regulates the processing of pre-TNF alpha in LPS-stimulated macrophages. J Cell Sci 121:149–154
Roy-Beaudry M, Martel-Pelletier J, Pelletier JP, M’Barek KN, Christgau S, Shipkolye F, Moldovan F (2003) Endothelin 1 promotes osteoarthritic cartilage degradation via matrix metalloprotease 1 and matrix metalloprotease 13 induction. Arthritis Rheum 48:2855–2864
Sabatini M, Lesur C, Thomas M, Chomel A, Anract P, de Nanteuil G, Pastoureau P (2005) Effect of inhibition of matrix metalloproteinases on cartilage loss in vitro and in a guinea pig model of osteoarthritis. Arthritis Rheum 52:171–180
Sadler JE (1998) Biochemistry and genetics of von Willebrand factor. Annu Rev Biochem 67:395–424
Sagane K, Hayakawa K, Kai J, Hirohashi T, Takahashi E, Miyamoto N, Ino M, Oki T, Yamazaki K, Nagasu T (2005) Ataxia and peripheral nerve hypomyelination in ADAM22-deficient mice. BMC Neurosci 6:33
Sagane K, Ishihama Y, Sugimoto H (2008) LGI1 and LGI4 bind to ADAM22, ADAM23 and ADAM11. Int J Biol Sci 4:387–396
Sandy JD (2006) A contentious issue finds some clarity: on the independent and complementary roles of aggrecanase activity and MMP activity in human joint aggrecanolysis. Osteoarthritis Cartilage 14:95–100
Sandy JD, Neame PJ, Boynton RE, Flannery CR (1991) Catabolism of aggrecan in cartilage explants. Identification of a major cleavage site within the interglobular domain. J Biol Chem 266:8683–8685
Sandy JD, Flannery CR, Neame PJ, Lohmander LS (1992) The structure of aggrecan fragments in human synovial fluid. Evidence for the involvement in osteoarthritis of a novel proteinase which cleaves the Glu373-Ala374 bond of the interglobular domain. J Clin Invest 89:1512–1516
Scheller J, Ohnesorge N, Rose-John S (2006) Interleukin-6 trans-signalling in chronic inflammation and cancer. Scand J Immunol 63:321–329
Shieh HS, Mathis KJ, Williams JM, Hills RL, Wiese JF, Benson TE, Kiefer JR, Marino MH, Carroll JN, Leone JW, Malfait AM, Arner EC, Tortorella MD, Tomasselli A (2008) High resolution crystal structure of the catalytic domain of ADAMTS-5 (aggrecanase-2). J Biol Chem 283:1501–1507
Smookler DS, Mohammed FF, Kassiri Z, Duncan GS, Mak TW, Khokha R (2006) Tissue inhibitor of metalloproteinase 3 regulates TNF-dependent systemic inflammation. J Immunol 176:721–725
Soejima K, Matsumoto M, Kokame K, Yagi H, Ishizashi H, Maeda H, Nozaki C, Miyata T, Fujimura Y, Nakagaki T (2003) ADAMTS-13 cysteine-rich/spacer domains are functionally essential for von Willebrand factor cleavage. Blood 102:3232–3237
Stanton H, Rogerson FM, East CJ, Golub SB, Lawlor KE, Meeker CT, Little CB, Last K, Farmer PJ, Campbell IK, Fourie AM, Fosang AJ (2005) ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434:648–652
Stanton H, Melrose J, Little CB, Fosang AJ (2011) Proteoglycan degradation by the ADAMTS family of proteinases. Biochim Biophys Acta 1812:1616–1629
Sternlicht MD, Werb Z (2001) How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol 17:463–516
Struglics A, Larsson S, Pratta MA, Kumar S, Lark MW, Lohmander LS (2006) Human osteoarthritis synovial fluid and joint cartilage contain both aggrecanase- and matrix metalloproteinase-generated aggrecan fragments. Osteoarthritis Cartilage 14:101–113
Takeda S (2009) Three-dimensional domain architecture of the ADAM family proteinases. Semin Cell Dev Biol 20:146–152
Takeda S, Igarashi T, Mori H, Araki S (2006) Crystal structures of VAP1 reveal ADAMs’ MDC domain architecture and its unique C-shaped scaffold. EMBO J 25:2388–2396
Takeda S, Takeya H, Iwanaga S (2012) Snake venom metalloproteinases: structure, function and relevance to the mammalian ADAM/ADAMTS family proteins. Biochim Biophys Acta 1824:164–176
Tallant C, Garcia-Castellanos R, Seco J, Baumann U, Gomis-Rüth FX (2006) Molecular analysis of ulilysin, the structural prototype of a new family of metzincin metalloproteases. J Biol Chem 281:17920–17928
Tan K, Duquette M, Liu JH, Dong Y, Zhang R, Joachimiak A, Lawler J, Wang JH (2002) Crystal structure of the TSP-1 type 1 repeats: a novel layered fold and its biological implication. J Cell Biol 159:373–382
Tape CJ, Willems SH, Dombernowsky SL, Stanley PL, Fogarasi M, Ouwehand W, McCafferty J, Murphy G (2011) Cross-domain inhibition of TACE ectodomain. Proc Natl Acad Sci USA 108:5578–5583
Tochowicz A, Goettig P, Evans R, Visse R, Shitomi Y, Palmisano R, Ito N, Richter K, Maskos K, Franke D, Svergun D, Nagase H, Bode W, Itoh Y (2011) The dimer interface of the membrane type 1 matrix metalloproteinase hemopexin domain: crystal structure and biological functions. J Biol Chem 286:7587–7600
Tortorella MD, Burn TC, Pratta MA, Abbaszade I, Hollis JM, Liu R, Rosenfeld SA, Copeland RA, Decicco CP, Wynn R, Rockwell A, Yang F, Duke JL, Solomon K, George H, Bruckner R, Nagase H, Itoh Y, Ellis DM, Ross H, Wiswall BH, Murphy K, Hillman MC Jr, Hollis GF, Arner EC (1999) Purification and cloning of aggrecanase-1: a member of the ADAMTS family of proteins. Science 284:1664–1666
Tortorella MD, Arner EC, Hills R, Easton A, Korte-Sarfaty J, Fok K, Wittwer A, Liu RQ, Malfait AM (2004) alpha 2-macroglobulin is a novel substrate for adamts-4 and represents an endogenous inhibitor of these enzymes. J Biol Chem 279(17):17554–17561
Troeberg L, Nagase H (2012) Proteases involved in cartilage matrix degradation in osteoarthritis. Biochim Biophys Acta 1824:133–145
Troeberg L, Tanaka M, Wait R, Shi YE, Brew K, Nagase H (2002) E. coli expression of TIMP-4 and comparative kinetic studies with TIMP-1 and TIMP-2: insights into the interactions of TIMPs and matrix metalloproteinase 2 (gelatinase A). Biochemistry 41:15025–15035
Troeberg L, Fushimi K, Khokha R, Emonard H, Ghosh P, Nagase H (2008) Calcium pentosan polysulfate is a multifaceted exosite inhibitor of aggrecanases. FASEB J 22:3515–3524
Troeberg L, Mulloy B, Ghosh P, Lee MH, Murphy G, Nagase H (2012) Pentosan polysulfate increases affinity between ADAMTS-5 and TIMP-3 through formation of an electrostatically driven trimolecular complex. Biochem J 443(1):307–315
Tsukada H, Pourmotabbed T (2002) Unexpected crucial role of residue 272 in substrate specificity of fibroblast collagenase. J Biol Chem 277:27378–27384
Van den Steen PE, Van AI, Hvidberg V, Piccard H, Fiten P, Jacobsen C, Moestrup SK, Fry S, Royle L, Wormald MR, Wallis R, Rudd PM, Dwek RA, Opdenakker G (2006) The hemopexin and O-glycosylated domains tune gelatinase B/MMP-9 bioavailability via inhibition and binding to cargo receptors. J Biol Chem 281:18626–18637
Visse R, Nagase H (2003) Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res 92:827–839
Wang P, Tortorella M, England K, Malfait AM, Thomas G, Arner EC, Pei D (2004) Proprotein convertase furin interacts with and cleaves pro-ADAMTS4 (Aggrecanase-1) in the trans-Golgi network. J Biol Chem 279:15434–15440
Weber S, Saftig P (2012) Ectodomain shedding and ADAMs in development. Development 139:3693–3709
Wei S, Kashiwagi M, Kota S, **e Z, Nagase H, Brew K (2005) Reactive site mutations in tissue inhibitor of metalloproteinase-3 disrupt inhibition of matrix metalloproteinases but not tumor necrosis factor-alpha-converting enzyme. J Biol Chem 280:32877–32882
Welgus HG, Jeffrey JJ, Stricklin GP, Roswit WT, Eisen AZ (1980) Characteristics of the action of human skin fibroblast collagenase on fibrillar collagen. J Biol Chem 255:6806–6813
White JM (2003) ADAMs: modulators of cell-cell and cell-matrix interactions. Curr Opin Cell Biol 15:598–606
Willems SH, Tape CJ, Stanley PL, Taylor NA, Mills IG, Neal DE, McCafferty J, Murphy G (2010) Thiol isomerases negatively regulate the cellular shedding activity of ADAM17. Biochem J 428:439–450
Williamson RA, Martorell G, Carr MD, Murphy G, Docherty AJP, Freedman RB, Feeney J (1994) Solution structure of the active domain of tissue inhibitor of metalloproteinases-2. A new member of the OB fold protein family. Biochemistry 33:11745–11759
Wingfield PT, Sax JK, Stahl SJ, Kaufman J, Palmer I, Chung V, Corcoran ML, Kleiner DE, Stetler-Stevenson WG (1999) Biophysical and functional characterization of full-length, recombinant human tissue inhibitor of metalloproteinases-2 (TIMP-2) produced in Escherichia coli – Comparison of wild type and amino-terminal alanine appended variant with implications for the mechanism of TIMP functions. J Biol Chem 274:21362–21368
Wisniewska M, Goettig P, Maskos K, Belouski E, Winters D, Hecht R, Black R, Bode W (2008) Structural determinants of the ADAM inhibition by TIMP-3: crystal structure of the TACE-N-TIMP-3 complex. J Mol Biol 381:1307–1319
Wu YI, Munshi HG, Sen R, Snipas SJ, Salvesen GS, Fridman R, Stack MS (2004) Glycosylation broadens the substrate profile of membrane type 1 matrix metalloproteinase. J Biol Chem 279:8278–8289
Xu P, Derynck R (2010) Direct activation of TACE-mediated ectodomain shedding by p38 MAP kinase regulates EGF receptor-dependent cell proliferation. Mol Cell 37:551–566
Yamamoto K, Troeberg L, Scilabra SD, Pelosi M, Murphy CL, Strickland DK, Nagase H (2013) LRP-1-mediated endocytosis regulates extracellular activity of ADAMTS-5 in articular cartilage. FASEB J 27:511–521
Zhang Q, Thomas SM, Lui VW, ** S, Siegfried JM, Fan H, Smithgall TE, Mills GB, Grandis JR (2006) Phosphorylation of TNF-alpha converting enzyme by gastrin-releasing peptide induces amphiregulin release and EGF receptor activation. Proc Natl Acad Sci U S A 103:6901–6906
Acknowledgements
We thank Ngee Han Lim, Alan Lyons and Rob Visse for preparation of the figures. This work is supported by grant from Arthritis Research UK, European FP7 LIVIMODE programme, NIH/NIAMS grant AR40994 (to HN), and Cancer Research UK and Medical Research Council (to GM).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Wien
About this chapter
Cite this chapter
Nagase, H., Murphy, G. (2013). Metalloproteinases in Cartilage Matrix Breakdown: The Roles in Rheumatoid Arthritis and Osteoarthritis. In: Brix, K., Stöcker, W. (eds) Proteases: Structure and Function. Springer, Vienna. https://doi.org/10.1007/978-3-7091-0885-7_13
Download citation
DOI: https://doi.org/10.1007/978-3-7091-0885-7_13
Published:
Publisher Name: Springer, Vienna
Print ISBN: 978-3-7091-0884-0
Online ISBN: 978-3-7091-0885-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)