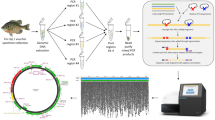
Abstract
We review recent advances in the use of molecular techniques as they apply to monitoring restoration efforts in lakes. Using DNA sequence data, biodiversity can now be assessed to levels previously unattainable using traditional, morphological assessments. In particular, DNA barcoding, the use of small standardised fragments of DNA, has become an increasingly widespread and common approach to identify species. Global initiatives such as the International Barcode of Life (iBOL) have coordinated these efforts and facilitated publically accessible reference databases such as the Barcode of Life Datasystems (BOLD). Such databases can be used for routine identification of specimens as well as for the assessment of community composition and monitoring of changes over time. Through the application of Next Generation Sequencing techniques, multiple samples can be run simultaneously (metabarcoding), greatly automating and streamlining the monitoring process. Reference databases can also be applied to environmental DNA (DNA that is shed into the environment by plants and animals). Here, species can be identified “sight unseen” through analyses of environmental samples (e.g. water, sediment). This latter method has proven useful for the monitoring of exotic fish species, particularly following eradication efforts. Ongoing developments in sequencing technology are likely to further enhance the utility of molecular techniques for assessing and monitoring restoration efforts in New Zealand.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Armstrong K, Ball S (2005) DNA barcodes for biosecurity: invasive species identification. Philos Trans R Soc Lond B Biol Sci 360:1813–1823
Auckland Regional Council (2005) Assessment of trophic state change in selected lakes of the Auckland Region based on rotifer assemblages. Technical Publication 269:31 Auckland Regional Council, Auckland, New Zealand
Baird DJ, Hajibabaei M (2012) Biomonitoring 2.0: a new paradigm in ecosystem assessment made possible by next–generation DNA sequencing. Mol Ecol 21:2039–2044
Banks JC, Hogg ID, Cary SC (2009) The phylogeography of Adelie penguin faecal bacteria. Environ Microbiol 11:577–588
Banks JC, Demetras NJ, Hogg ID, Knox MA, West DW (2016) Monitoring brown trout (Salmo trutta) eradication in a wildlife sanctuary using environmental DNA. N Z Nat Sci 41:1–13
Bianchi F, Acri F, Aubry FB, Berton A, Boldrin A, Camatti E, Cassin D, Comaschi A (2003) Can plankton communities be considered as bio-indicators of water quality in the Lagoon of Venice? Mar Pollut Bull 46:964–971
Biggs J, Ewald N, Valentini A, Gaboriaud C, Dejean T, Griffiths RA, Foster J, Wilkinson JW, Arnell A, Brotherton P, Williams P, Dunn F (2015) Using eDNA to develop a national citizen science-based monitoring programme for the great crested newt (Triturus cristatus). Biol Conserv 183:19–28
Bik HM, Porazinska DL, Creer S, Caporaso JG, Knight R, Thomas WK (2012) Sequencing our way towards understanding global eukaryotic biodiversity. Trends Ecol Evol 27:233–243
Bongers T (1990) The maturity index: an ecological measure of environmental disturbance based on nematode species composition. Oecologia 83:14–19
Burns NM, Rutherford JC, Clayton JS (1999) A monitoring and classification system for New Zealand lakes and reservoirs. Lake Reservoir Manage 15:255–271
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Chapman MA, Lewis MH, Winterbourn MJ (2011) Guide to the freshwater Crustacea of New Zealand. New Zealand Freshwater Sciences Society, Wellington, New Zealand
Civade R, Dejean T, Valentini A et al (2016) Spatial representativeness of environmental DNA metabarcoding signal for fish biodiversity assessment in a natural freshwater system. PLoS One 11:e0157366
Cline J, Braman JC, Hogrefe HH (1996) PCR fidelity of Pfu DNA polymerase and other thermostable DNA polymerases. Nucleic Acids Res 24:3546–3551
Cristescu ME (2015) Genetic reconstructions of invasion history. Mol Ecol 24:2212–2225
Darling JA, Mahon AR (2011) From molecules to management: adopting DNA-based methods for monitoring biological invasions in aquatic environments. Environ Res 111:978–988
Deagle BE, Thomas AC, Shaffer AK, Trites AW, Jarman SN (2013) Quantifying sequence proportions in a DNA-based diet study using Ion Torrent amplicon sequencing: which counts count? Mol Ecol Resour 13:620–633
Deiner K, Walser J-C, Mächler E, Altermatt F (2015) Choice of capture and extraction methods affect detection of freshwater biodiversity from environmental DNA. Biol Conserv 183:53–63
Dejean T, Valentini A, Duparc A, Pellier-Cuit S, Pompanon F, Teberlet P, Miaud C (2011) Persistence of environmental DNA in freshwater ecosystems. PLoS One 6:e23398
Dejean T, Valentini A, Miquel C, Taberlet P, Bellemain E, Miaud C (2012) Improved detection of an alien invasive species through environmental DNA barcoding: the example of the American bullfrog Lithobates catesbeianus. J Appl Ecol 49:953–959
Diggle J, Patil J, Wisniewski C (2012) A manual for carp control: the Tasmanian model. Invasive Animals CRC, New Norfolk, TAS, Australia
Dowle E, Pochon X, Banks J, Shearer K, Wood SA (2016) Targeted gene enrichment and high throughput sequencing for environmental biomonitoring: a case study using freshwater macroinvertebrates. Mol Ecol Resour 16(5):1240–1254
Duggan IC (2007) An assessment of the water quality of ten Waikato lakes based on zooplankton community composition. CBER Contract Report No. 60, Prepared for Environment Waikato, Centre for Biodiversity and Ecology Research, University of Waikato, Hamilton, New Zealand
Duggan IC, Green JD, Shiel RJ (2001a) Distribution of rotifers in North Island, New Zealand, and their potential use as bioindicators of lake trophic state. Hydrobiologia 446–447:155–164
Duggan IC, Green JD, Thomasson K (2001b) Do rotifers have potential as bioindicators of lake trophic state? Verh Internat Verein Limnol 27:3497–3502
Duggan IC, Robinson KV, Burns CW, Banks JC, Hogg ID (2012) Identifying invertebrate invasions using morphological and molecular analyses: North American Daphnia ‘pulex’ in New Zealand fresh waters. Aquat Invasions 7:585–590
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200
Environmental Protection Agency (2014) http://www.epa.gov/greatlakes/basicinfo.html. Accessed 3 Sept 2018
Evans NT, Olds BP, Renshaw MA, Turner CR, Li Y, Jerde CL, Mahon AR, Pfrender ME, Lamberti GA, Lodge DM (2016) Quantification of mesocosm fish and amphibian species diversity via environmental DNA metabarcoding. Mol Ecol Resour 16:29–41
Ficetola GF, Miaud C, Pompanon F, Taberlet P (2008) Species detection using environmental DNA from water samples. Biol Lett 4:423–425
Ficetola GF, Pansu J, Bonin A, Coissac E, Giguet-Covex C, De Barba M, Gielly L, Lopes CM, Boyer F (2015) Replication levels, false presences and the estimation of the presence/absence from eDNA metabarcoding data. Mol Ecol Resour 15:543–556
Field KG, Samadpour M (2007) Fecal source tracking, the indicator paradigm, and managing water quality. Water Res 41:3517–3538
Furlan E, Gleeson D (2017) Improving reliability in environmental DNA detection surveys through enhanced quality control. Mar Freshw Res 68:388–395
Furlan E, Gleeson D, Hardy C, Duncan RP (2015) A framework for estimating the sensitivity of eDNA detection. Mol Ecol Resour 16:641–654
Goldberg CS, Pilliod DS, Arkle RS, Waits LP (2011) Molecular detection of vertebrates in stream water: a demonstration using Rocky Mountain tailed frogs and Idaho giant salamanders. PLoS One 6:e22746
Goldberg CS, Sepulveda A, Ray A, Baumgardt J, Waits LP (2013) Environmental DNA as a new method for early detection of New Zealand mudsnails (Potamopyrgus antipodarum). Freshw Sci 32:792–800
Griffin DW, Lipp EK, McLaughlin MR, Rose JB (2001) Marine recreation and public health microbiology: quest for the ideal indicator. Bioscience 51:817–826
Gu W, Swihart RK (2004) Absent or undetected? Effects of non-detection of species occurrence on wildlife–habitat models. Biol Conserv 116:195–203
Haas BJ, Gevers D, Earl AM, Feldgarden M, Ward DV, Giannoukos G, Ciulla D, Tabbaa D, Highlander SK, Sodergren E, Methé B, DeSantis TZ; Human Microbiome Consortium, Petrosino JF, Knight R, Birren BW (2011) Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res 21:494–504
Hajibabaei M, Shokralla S, Zhou X, Singer GA, Baird DJ (2011) Environmental barcoding: a next-generation sequencing approach for biomonitoring applications using river benthos. PLoS One 6:e17497
Hänfling B, Handley L, Read DS, Hahn C, Li J, Nichols P, Blackman RC, Oliver A, Winfield IJ (2016) Environmental DNA metabarcoding of lake fish communities reflects long-term data from established survey methods. Mol Ecol 25:3101–3119
Hebert PDN, Cywinska A, Ball SL, de Waard JR (2003a) Biological identifications through DNA barcodes. Proc Biol Sci 270:313–321
Hebert PDN, Ratnasingham S, de Waard JR (2003b) Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proc Biol Sci 270(Suppl 1):S96–S99
Hicks BJ, Watson NRN (1985) Seasonal changes in abundance of brown trout (Salmo trutta) and rainbow trout (S. gairdnerii) assessed by drift diving in the Rangitikei River, New Zealand. N Z J Mar Freshw Res 19:1–10
Hicks BJ, Brijs J, Daniel A, Morgan DKJ, Ling N (2015) Biomass estimation of invasive fish. In: Collier KJ, Gainger NPJ (eds) New Zealand invasive fish management handbook. The University of Waikato, Hamilton, New Zealand, pp 116–122
Hofreiter M, Mead JI, Martin P, Poinar HN (2003) Molecular caving. Curr Biol 13:R693–R695
Hogg ID, Larose C, de Lafontaine Y, Doe KG (1998) Genetic evidence for a Hyalella species complex within the Great Lakes-St. Lawrence River drainage basin: implications for ecotoxicology and conservation biology. Can J Zool 76:1134–1152
Hogg ID, Stevens MI, Schnabel KE, Chapman MA (2006) Deeply divergent lineages among populations of the widespread New Zealand amphipod Paracalliope fluviatilis revealed using allozyme and mitochondrial DNA analyses. Freshw Biol 51:236–248
Jerde CL, Mahon AR, Chadderton WL, Lodge DM (2011) “Sight-unseen” detection of rare aquatic species using environmental DNA. Conserv Lett 4:150–157
Jerde CL, Chadderton WL, Mahon AR, Renshaw MA, Corush J, Budny ML, Mysorekar S, Lodge DM (2013) Detection of Asian carp DNA as part of a Great Lakes basin-wide surveillance program. Can J Fish Aquat Sci 70:522–526
Laramie MB, Pilliod DS, Goldberg CS (2015) Characterizing the distribution of an endangered salmonid using environmental DNA analysis. Biol Conserv 183:29–37
Lear G, Lewis GD (2009) Impact of catchment land use on bacterial communities within stream biofilms. Ecol Indic 9:848–855
Lindeque PK, Parry HE, Harmer RA et al (2013) Next generation sequencing reveals the hidden diversity of zooplankton assemblages. PLoS One 8:e81327
Lodge DM, Turner CR, Jerde CL, Barnes MA, Chadderton L, Egan SP, Feder JL, Mahon AR, Pfrender ME (2012) Conservation in a cup of water: estimating biodiversity and population abundance from environmental DNA. Mol Ecol 21:2555–2558
Machida R, Hashiguchi Y, Nishida M, Nishida S (2009) Zooplankton diversity analysis through single-gene sequencing of a community sample. BMC Genomics 10:438
Mahon AR, Jerde CL, Galaska M, Bergner JL, Chadderton WL, Lodge DM, Hunter ME, Nico LG (2013) Validation of eDNA surveillance sensitivity for detection of Asian carp in controlled and field experiments. PLoS One 8:e58316
Mahon AR, Nathan LR, Jerde CL (2014) Meta-genomic surveillance of invasive species in the bait trade. Conserv Genet Resour 6:563–567
McDowall RM (2003) Impacts of introduced salmonids on native galaxiids in New Zealand upland streams: a new look at an old problem. Trans Am Fish Soc 132:229–238
McInerney PJ, Rees GN, Gawne B, Suter P, Watson G, Stoffels RJ (2016) Invasive willows drive instream community structure. Freshw Biol 61:1379–1391
McIntosh AR, McHugh PA, Dunn NR, Goodman J, Howard SW, Jellyman PG, O’Brien LK, Nyström P, Woodford DJ (2010) The impact of trout on galaxiid fishes in New Zealand. N Z J Ecol 34:195–206
Metzker ML (2010) Sequencing technologies—the next generation. Nat Rev Genet 11:31–46
Mills EL, Leach JH, Carlton JT, Secor CL (1993) Exotic species in the Great Lakes: a history of biotic crises and anthropogenic introductions. J Great Lakes Res 19:1–54
Minamoto T, Yamanaka H, Takahara T, Honjo MN, Kawabata Z (2012) Surveillance of fish species composition using environmental DNA. Limnology 13:193–197
Moyer GR, Díaz-Ferguson E, Hill JE, Shea C (2014) Assessing environmental DNA detection in controlled lentic systems. PLoS One 9:e103767
Murray DC, Bunce M, Cannell BL, Oliver R, Houston J, White NE, Barrero RA, Bellgard MI, Haile J (2011) DNA-based faecal dietary analysis: a comparison of qPCR and high throughput sequencing approaches. PLoS One 6:e2577
Nathan LR, Jerde CL, Budny ML, Mahon AR (2014) The use of environmental DNA in invasive species surveillance of the Great Lakes commercial bait trade. Conserv Biol 29:430–439
National Oceanic and Atmospheric Administration (2014) http://www.glerl.noaa.gov/pr/ourlakes/economy.html. Accessed 3 Dec 2014
Nolte V, Pandey RV, Jost S, Medinger R, Ottenwälder B, Boenigk J, Schlötterer C (2010) Contrasting seasonal niche separation between rare and abundant taxa conceals the extent of protist diversity. Mol Ecol 19:2908–2915
Olds BP, Jerde CL, Renshaw MA et al (2016) Estimating species richness using environmental DNA. Ecol Evol 6:4214–4226
Porazinska DL, Sung WAY, Giblin-Davis RM, Thomas WK (2010) Reproducibility of read numbers in high-throughput sequencing analysis of nematode community composition and structure. Mol Ecol Resour 10:666–676
Quail MA, Smith M, Coupland P, Otto TD, Harris SR, Connor TR, Bertoni A, Swerdlow HP, Gu Y (2012) A tale of three next generation sequencing platforms: comparison of Ion Torrent, Pacific Biosciences and Illumina MiSeq sequencers. BMC Genomics 13:341
Quince C, Lanzen A, Davenport RJ, Turnbaugh PJ (2011) Removing noise from pyrosequenced amplicons. BMC Bioinformatics 12:38
Ratnasingham S, Hebert PDN (2007) BOLD: the barcode of life data system (http://www.barcodinglife.org). Mol Ecol Notes 7:355–364
Ratnasingham S, Hebert PDN (2013) A DNA-based registry for all animal species: the barcode index number (BIN) system. PLoS One 8:e66213
Rees HC, Maddison BC, Middleditch DJ, Patmore JRM, Gough KC (2014) The detection of aquatic animal species using environmental DNA—a review of eDNA as a survey tool in ecology. J Appl Ecol 51:1450–1459
Schloss PD, Westcott SL, Ryabin T et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Schmidt BR, Kéry M, Ursenbacher S, Hyman OJ, Collins JP (2013) Site occupancy models in the analysis of environmental DNA presence/absence surveys: a case study of an emerging amphibian pathogen. Methods Ecol Evol 4:646–653
Scott TM, Rose JB, Jenkins TM, Farrah SR, Lukasik J (2002) Microbial source tracking: current methodology and future directions. Appl Environ Microbiol 68:5796–5803
Scriver M, Marinich A, Wilson C, Freeland J (2015) Development of species-specific environmental DNA (eDNA) markers for invasive aquatic plants. Aquat Bot 122:27–31
Sevilla RG, Diez A, Norén M et al (2007) Primers and polymerase chain reaction conditions for DNA barcoding teleost fish based on the mitochondrial cytochrome b and nuclear rhodopsin genes. Mol Ecol Notes 7:730–734
Shaw JL, Clarke ALJ, Wedderburn SD, Barnes TC, Weyricha LS, Cooper A (2016) Comparison of environmental DNA metabarcoding and conventional fish survey methods in a river system. Biol Conserv 197:131–138
Sigsgaard EE, Carl H, Møller PR, Thomsen PF (2015) Monitoring the near-extinct European weather loach in Denmark based on environmental DNA from water samples. Biol Conserv 183:46–52
Spear SF, Groves JD, Williams LA, Waits LP (2015) Using environmental DNA methods to improve detectability in a hellbender (Cryptobranchus alleganiensis) monitoring program. Biol Conserv 183:38–45
Stark JD (1993) Performance of the macroinvertebrate community index: effects of sampling method, sample replication, water depth, current velocity, and substratum on index values. N Z J Mar Freshw Res 27:463–478
Stewart-Oaten A, Murdoch WW, Parker KR (1986) Environmental impact assessment: “Pseudoreplication” in time? Ecology 67:929–940
Takahara T, Minamoto T, Doi H (2015) Effects of sample processing on the detection rate of environmental DNA from the common carp (Cyprinus carpio). Biol Conserv 183:64–69
Thomsen PF, Willerslev E (2015) Environmental DNA – an emerging tool in conservation for monitoring past and present biodiversity. Biol Conserv 183:4–18
Thomsen P, Kielgast JOS, Iversen LL, Wiuf C, Rasmussen M, Gilbert MT, Orlando L, Willerslev E (2012) Monitoring endangered freshwater biodiversity using environmental DNA. Mol Ecol 21:2565–2573
Townsend CR (1996) Invasion biology and ecological impacts of brown trout Salmo trutta in New Zealand. Biol Conserv 78:13–22
Valentini A, Taberlet P, Miaud C et al (2016) Next-generation monitoring of aquatic biodiversity using environmental DNA metabarcoding. Mol Ecol 25:929–942
Wilcox TM, Mckelvey KS, Young MK, Jane SF, Lowe WH, Whiteley AR, Schwartz MK (2013) Robust detection of rare species using environmental DNA: the importance of primer specificity. PLoS One 8:e59520
Willerslev E, Cappellini E, Boomsma W et al (2007) Ancient biomolecules from deep ice cores reveal a forested Southern Greenland. Science 317:111–114
Wilson IG (1997) Inhibition and facilitation of nucleic acid amplification. Appl Environ Microbiol 63:3741–3751
Wittmann ME, Cooke RM, Rothlisberger JD, Rutherford ES, Zhang H, Mason DM, Lodge DM (2014) Use of structured expert judgment to forecast invasions by bighead and silver carp in Lake Erie. Conserv Biol 29:187–197
Wood SA, Smith KF, Banks J, Tremblay L, Rhodes L, Mountfort D, Cary SC, Pochon X (2013) Molecular tools for environmental monitoring of New Zealand’s aquatic habitats: past, present and the future. N Z J Mar Freshw Res 47:90–119
Yoccoz NG, Nichols JD, Boulinier T (2001) Monitoring of biological diversity in space and time. Trends Ecol Evol 16:446–453
Yu DW, Ji Y, Emerson BC, Wang X, Ye C, Yang C, Ding Z (2012) Biodiversity soup: metabarcoding of arthropods for rapid biodiversity assessment and biomonitoring. Methods Ecol Evol 3:613–623
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Hogg, I.D., Banks, J.C., Woods, S.M. (2018). Biodiversity Genomics: Monitoring Restoration Efforts Using DNA Barcoding and Environmental DNA. In: Hamilton, D., Collier, K., Quinn, J., Howard-Williams, C. (eds) Lake Restoration Handbook. Springer, Cham. https://doi.org/10.1007/978-3-319-93043-5_12
Download citation
DOI: https://doi.org/10.1007/978-3-319-93043-5_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-93042-8
Online ISBN: 978-3-319-93043-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)