Abstract
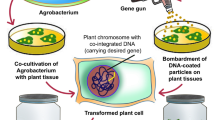
Sugarcane (Saccharum officinarum) is an important cash crop cultivated across the world. Conventional breeding methods are used to cross different Saccharum spp. to develop sugarcane hybrids with high sucrose content and for other novel traits including increased tolerance to various biotic and abiotic stresses. Two major factors that limit conventional breeding method are that it is highly time consuming and difficulty in getting the desirable trait in the hybrid. These limitations can be overcome by genetic transformation method in which specific gene(s) are used to generate stable transgenic lines expressing specific trait. Compared to conventional breeding methods, generation of stable lines takes less time. In addition, complications associated with backcross and testcross during breeding program can be avoided. There are several reports since 1990s mentioning generation of transgenic sugarcane by different methods of transformation such as electroporation, particle bombardment method, and Agrobacterium-mediated transformation. Transient expression systems have also been developed in sugarcane. Nevertheless, all transformation methodologies have their own limitation which hinders the stable expression of the transformed gene. Here, we discuss about the complications and factors affecting efficiency of genetic transformation in sugarcane.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ali S, Hassan SW, Razi-ud-Din S, Shah SZR (2004) Micropropagation of sugarcane through bud culture. Sarhad J. Agric 20:79–82
Anderson DJ, Birch RG (2012) Minimal handling and super-binary vectors facilitate efficient, Agrobacterium-mediated transformation of sugarcane (Saccharum spp. hybrid). Trop Plant Biol 5:183–192
Arencibia AD, Carmona ER (2006) Sugarcane (Saccharum spp.) Methods Mol Biol 344:227–235
Arencibia A, Molina P, Gutierrez C, Fuentes A, Greenidge V, Menendez E, de la Riva G, Selman-Housein G (1992) Regeneration of transgenic sugarcane (Saccharum officinarum L.) plants from intact meristematic tissue transformed by electroporation. Biotecnol Apl 9:156–165
Arencibia A, Molina PR, de la Riva G, Selman-Housein G (1995) Production of transgenic sugarcane (Saccharum officinarum L.) plants by intact cell electroporation. Plant Cell Rep 14:305–309
Arencibia A, Vazquez R, Prieto D, Tellez P, Carmona E, Coego A, Hernandez L, de la Riva G, Selman-Housein G (1997) Transgenic sugarcane plants resistant to stem-borer attack. Mol Breed 3:247–255
Arencibia AD, Carmona ER, Tellez P, Chan MT, SM Y, Trujillo LE, Oramas P (1998) An efficient protocol for sugarcane (Saccharum spp. L.) transformation mediated by Agrobacterium tumefaciens. Transgenic Res 7:1–10
Arvinth S, Arun S, Selvakesavan RK, Srikanth J, Mukunthan N, Kumar PA, Premachandran MN, Subramonian N (2010) Genetic transformation and pyramiding of aprotinin-expressing sugarcane with Cry1Ab for shoot borer (Chiloinfuscatellus) resistance. Plant Cell Rep 29:383–395
Asad S, Amoozegar MA, Pourbabaee AA, Sarbolouki MN, Dastgheib SM (2007) Decolorization of textile dyes by newly isolated halophilic and halotolerant bacteria. Bioresour Technol 98:2082–2088
Bevan M (1984) Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res 12:8711–8721
Bower R, Birch RG (1992) Transgenic sugarcane plants via microprojectile bombardment. Plant J 2:409–416
Bower R, Elliott AR, Potier BAM, Birch RG (1996) High-efficiency, microprojectile-mediated co-transformation of sugarcane, using visible or selectable markers. Mol Breed 2:239–249
Cheavegatti-Gianotto A, de Abreu HMC, Arruda P, Bespalhok-Filho JC, Burnquist WL, Creste S et al (2011) Sugarcane (Saccharum X officinarum): a reference study for the regulation of genetically modified cultivars in Brazil. Trop Plant Biol 4:62–89
Chen WH, Gartland KMA, Davey MR, Sotak R, Gartland JS, Mulligan BJ, Power JB, Cocking EC (1987) Transformation of sugarcane protoplasts by direct uptake of a selectable chimeric gene. Plant Cell Rep 6:297–301
Cheng M, Jarret RL, Li Z, **ng A, Demski JW (1996) Production of fertile transgenic peanut (Arachishypogaea L.) plants using Agrobacterium tumefaciens. Plant Cell Rep 15:653–657
Cheng M, Fry JE, Pang S, Zhou H, Hironaka CM, Duncan DR, Conner TW, Wan Y (1997) Genetic transformation of wheat mediated by Agrobacterium tumefaciens. Plant Physiol 115:971–980
Chowdhury MKU, Vasil IK (1992) Stably transformed herbicide resistant callus of sugarcane via microprojectile bombardment of cell suspension cultures and electroporation of protoplasts. Plant Cell 11:494–498
Christou P, McCabe DE, Swain WF (1988) Stable transformation of soybean callus by DNA-coated gold particles. Plant Physiol 87:671–674
Christy LA, Arvinth S, Saravanakumar M, Kanchana M, Mukunthan N, Srikanth J, George T, Subramonian N (2009) Engineering sugarcane cultivars with bovine pancreatic trypsin inhibitor (aprotinin) gene for protection against top borer (Scripophaga excerptalis Walker). Plant Cell Rep 28:175–184
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Dafny-Yelin M, Tzfira T (2007) Delivery of multiple transgenes to plant cells. Plant Physiol 145:1118–1128
De La Riva GA, Gonzalez-Cabrera J, Vazquez-Padron R, Ayra-Pardo C (1998) Agrobacterium tumefaciens: a natural tool for plant transformation. Electron J Biotechnol 1:24–25
Dong S, Delucca P, Geijskes RJ, Ke J, Mayo K, Mai P, Sainz M, Caffall K et al (2014a) Advances in Agrobacterium-mediated sugarcane transformation and stable transgene expression. Sugar Tech 16:366–371
Dong H, Zhao Y, Wang Y, Li H (2014b) Recombinant proteins expressed in lettuce. Indian J Biotechnol 13:427–436
Elliott AR, Campbell JA, Dugdale B, Brettell RIS, Grof CPL (1999) Green-fluorescent protein facilitates rapid in vivo detection of genetically transformed plant cells. Plant Cell Rep 18:707–714
Enriquez-Obregon GA, Vazquez-Padron RI, Prieto-Samsonov DL, Perez M, Selman-Housein G (1997) Genetic transformation of sugarcane by Agrobacterium tumefaciens using antioxidants compounds. Biotecnol Apl 14:169–174
Enriquez-Obregon GA, Vazquez-padron RI, Prieto-sansonov DL, de la Riva GA, Selman-Housein G (1998) Herbicide resistant sugarcane (Saccharum officinarum L.) plants by Agrobacterium-mediated transformation. Planta 206:20–27
Falco MC, Neto AT, Mendes BMJ, Arias FJZ (1996) Isolation and cultivation of protoplasts. Bras Fisiol Veg 8:175–179
Franks T, Birch RG (1991) Gene transfer into intact sugarcane cells using microprojectile bombardment. Funct Plant Biol 18:471–480
Gallo-Meagher M, Irvine JE (1993) Effects of tissue type and promoter strength on transient GUS expression in sugarcane following particle bombardment. Plant Cell Rep 12:666–670
Gallo-Meagher M, Irvine JE (1996) Herbicide resistant sugarcane containing the bar gene. Crop Sci 36:1367–1374
Gambley RL, Ford R, Smith GR (1993) Microprojectile transformation of sugarcane meristems and regeneration of shoots expressing β-glucuronidase. Plant Cell Rep 12:343–346
Gao SJ, Damaj MB, Park JW, Beyene G, Buenrostro-Nava MT, Molina J, Wang X, Ciomperlik JJ, Manabayeva SA, Alvarado VY, Rathore KS, Scholthof HB, Mirkov TE (2013) Enhanced transgene expression in sugarcane by co-expression of virus-encoded RNA silencing suppressors. PloS One 14:e66046
Gao S, Yang Y, Wang C, Guo J, Zhou D, Wu Q, Su Y, Xu L, Que Y (2016) Transgenic sugarcane with a Cry1Ac gene exhibited better phenotypic traits and enhanced resistance against sugarcane borer. PLoS One 11:e0153929
Geijskes RJ, Wang L, Lakshmanan P, Mckeon MG, Berding N, Swain RS et al (2003) Smartsett™ seedlings: tissue cultured seed plants for the Australian sugar industry. Sugarcane Int 25:13–17
Gelvin SB (2003) Agrobacterium-mediated plant transformation: the biology behind the “gene-jockeying” tool. Microbiol Mol Biol Rev 67:16–37
Grzebelus E, Skop L (2014) Effect of β-lactam antibiotics on plant regeneration in carrot protoplast cultures. In Vitro Cell Dev Biol Plant 50:568–575
Guo JL, Ling H, Wu QB, Xu LP, Que YX (2014) The choice of reference genes for assessing gene expression in sugarcane under salinity and drought stresses. Sci Rep 4:7042
Hauptmann RM, Vasil V, Ozias-Skins P, Tabaeizadeh Z, Rogers SG, Fraley RT, Horsch RB, Vasil IK (1988) Evaluation of selectable markers for obtaining stable transformation in the Gramineae. Plant Physiol 86:602–606
Heinz DJ, Mee GW (1969) Plant differentiation from callus tissue of Saccharum species. Crop Sci 9:346–348
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6:271–282
Hiei Y, Komari T, Kubo T (1997) Transformation of rice mediated by Agrobacterium tumefaciens. Plant Mol Biol 35:205–218
Hille J, Verheggen F, Roelvink P, Franssen H, van Kammen A, Zabel P (1986) Bleomycin resistance: a new dominant selectable marker for plant cell transformation. Plant Mol Biol 7:171–176
Ingelbrecht IL, Irvine JE, Mirkov TE (1999) Post transcriptional gene silencing in transgenic sugarcane. Dissection of homology-dependent virus resistance in a monocot that has a complex polyploid genome. Plant Physiol 119:1187–1198
Ishida Y, Saito H, Ohta S, Hiei Y, Komari T, Kumashiro T (1996) High efficiency transformation of maize (Zea mays L.) mediated by Agrobacterium tumefaciens. Nat Biotechnol 14:745–750
Jackson MA, Anderson DJ, Birch RG (2013) Comparison of Agrobacterium and particle bombardment using whole plasmid or minimal cassette for production of high-expressing, low-copy transgenic plants. Transgenic Res 22:143–151
Jain M, Chengalrayan K, Abouzid A, Gallo M (2007) Prospecting the utility of a PMI/mannose selection system for the recovery of transgenic sugarcane (Saccharum spp. hybrid) plants. Plant Cell Rep 26:581–590
Joyce P, Kuwahata M, Turner N, Lakshmanan P (2010) Selection system and co cultivation medium are important determinants of Agrobacterium-mediated transformation of sugarcane. Plant Cell Rep 29:173–183
Joyce P, Hermann S, Connell A, Dinh Q, Shumbe L, Lakshmanan P (2014) Field performance of transgenic sugarcane produced using Agrobacterium and biolistics methods. Plant Biotechnol J 12:411–424
Kalunke RM, Kolge AM, Babu KH, Prasad DT (2009) Agrobacterium mediated transformation of sugarcane for borer resistance using Cry1Aa3 gene and one-step regeneration of transgenic plants. Sugar Tech 11:355–359
Karami O (2008) Factors affecting Agrobacterium-mediated transformation of plants. Transgenic Plant J 2:127–137
Kaur A, Gill MS, Gill R, Gosal SS (2007) Standardization of different parameters for ‘particle gun’ mediated genetic transformation of sugarcane (Saccharum officinarum L.) Indian J Biotechnol 6:31–34
Khamrit R, Jaisil P, Bunnag S (2012) Callus induction, regeneration and transformation of sugarcane (Saccharum officinarum L.) with chitinase gene using particle bombardment. Afr J Biotechnol 11:6612–6618
Klein TM, Harper EC, Svab Z, Sanford JC, Fromm ME, Maliga P (1988) Stable genetic transformation of intact Nicotiana cells by the particle bombardment process. Proc Natl Acad Sci 85:8502–8505
Komori T, Imayama T, Kato N, Ishida Y, Ueki J, Komari T (2007) Current status of binary vectors and super-binary vectors. Plant Physiol 145:1155–1160
Kumar T, Uzma KMR, Abbas Z, Ali GM (2014) Genetic improvement of sugarcane for drought and salinity stress tolerance using Arabidopsis vacuolar pyrophosphatase (AVP1) gene. Mol Biotechnol 56:199–209
Lakshmanan P, Geijskes RJ, Aitken KS, Grof CLP, Bonnett GD, Smith GR (2005) Sugarcane biotechnology: the challenges and opportunities. In Vitro Cell Dev Biol Plant 41:345–363
Lee LY, Gelvin SB (2008) T-DNA binary vectors and systems. Plant Physiol 146:325–332
Leibbrandt NB, Snyman SJ (2001) Initial field testing of transgenic glufosinate-ammonium resistant sugarcane. S Afr Sugar Technol Proc 75:105–108
Lutz KA, Azhagiri AK, Tungsuchat-Huang T, Maliga P (2007) A guide to choosing vectors for transformation of the plastid genome of higher plants. Plant Physiol 145:1201–1210
Manfroi E, Yamazaki-Lau E, Grando MF, Roesler EA (2015) Acetosyringone, pH and temperature effects on transient genetic transformation of immature embryos of Brazilian wheat genotypes by Agrobacterium tumefaciens. Genet Mol Biol 38:470–476
Manickavasagam M, Ganapathi A, Anbazhagan VR, Sudhakar B, Selvaraj N, Vasudevan A, Kasthurirengan S (2004) Agrobacterium-mediated genetic transformation and development of herbicide-resistant sugarcane (Saccharum species hybrids) using axillary buds. Plant Cell Rep 23:134–143
Mayavan S, Subramanyam K, Arun M, Rajesh M, Dev GK, Sivanandhan G, Jaganath B, Manickavasagam M, Selvaraj N, Ganapathi A (2013) Agrobacterium tumefaciens-mediated in planta seed transformation strategy in sugarcane. Plant Cell Rep 32:1557–1574
Mayavan S, Subramanyam K, Jaganath B, Sathish D, Manickavasagam M, Ganapathi A (2015) Agrobacterium-mediated in planta genetic transformation of sugarcane setts. Plant Cell Rep 34:1835–1848
Mohan C (2016) Genome editing in sugarcane: challenges ahead. Front Plant Sci 7:1542
Molina P, Menendez E, Arencibia A, Fuentes A, Alfonso J, Coll Y, Greenidge V, Perez S, Selman-Housein G (1993) Transient gene expression in sugarcane protoplasts after electroporation and polyethylene glycol treatment. Biotecnol Apl 10:171–175
Nandakumar R, Chen L, Rogers SMD (2004) Factors affecting the Agrobacterium-mediated transient transformation of the wetland monocot, Typhalatifolia. Plant Cell Tissue Organ Cult 79:31–38
Nasir IA, Tabassum B, Qamar Z, Javed MA, Tariq M, Farooq AM, Butt SJ, Qayyum A, Husnain T (2014) Herbicide-tolerant sugarcane (Saccharum officinarum L.) plants: an unconventional method of weed removal. Turk J Biol 38:439–449
Naureby B, Billing K, Wyndaele R (1997) Influence of the antibiotic timentin on plant regeneration compared to carbenicillin and cefotaxime in concentration suitable for elimination of Agrobacterium tumefaciens. Plant Sci 123:169–177
Naz S, Ali A, Iqbal J (2008) Phenolic content in vitro cultures of chick pea (Cicerarietinum L.) during callogenesis and organogenesis. Pak J Bot 40:2525–2539
Nutt KA, Allsopp PG, McGhie TK, Shepherd KM, Joyce PA (1999) Transgenic sugarcane with increased resistance to canegrubs. In: Conference of the Australian Society of Sugar Cane Technologists Proceedings, vol 21. pp 171–176
Que Q, Elumalai S, Li X, Zhong H, Nalapalli S, Schweiner M, Fei X, Nuccio M, Kelliher T, Gu W, Chen Z (2014) Maize transformation technology development for commercial event generation. Front Plant Sci 5:379
Rajeevkumar S, Anunanthini P, Ramalingam S (2015) Epigenetic silencing in transgenic plants. Front Plant Sci 6:693
Rashid H, Yoki S, Toriyama K, Hinata K (1996) Transgenic plant production mediated by Agrobacterium in indica rice. Plant Cell Rep 15:727–730
Rathus C, Birch RG (1992) Stable transformation of callus from electroporated sugarcane protoplasts. Plant Sci 82:81–89
Sanford JC, Klein TM, Wolf ED, Allen N (1987) Delivery of substances into cells and tissues using a particle bombardment process. Part Sci Technol 5:27–37
Seema G, Pande HP, Lal J, Madan VK (2001) Plantlet regeneration of sugarcane varieties and transient GUS expression in calli by electroporation. Sugar Tech 3:27–33
Shrawat AK, Lorz H (2006) Agrobacterium-mediated transformation of cereals: a promising approach crossing barriers. Plant Biotechnol J 4:575–603
Singh RK, Kumar P, Tiwari NN, Rastogi J, Singh SP (2013) Current status of sugarcane transgenic: an overview. Adv Genet Eng 2:2
Snyman SJ, Meyer GM, Carson DL, Botha FC (1996) Establishment of embryogenic callus and transient gene expression in selected sugarcane varieties. S Afr J Bot 62:29–37
Snyman SJ, Leibbrandt NB, Botha FC (1998) Buster resistant sugarcane. S Afr Sugar Technol Proc 72:140–142
Solangi SK, Qureshi ST, Khan IA, Raza S (2016) Establishment of in vitro callus in sugarcane (Saccharum officinarum L.) varieties influenced by different auxins. Afr J Biotechnol 15:1541–1550
Song ZY, Tian JL, WZ F, Li L, LH L, Zhou L, Shan Z, Tang GX, Shou HX (2013) Screening Chinese soybean genotypes for Agrobacterium-mediated genetic transformation suitability. J Zhejiang Univ Sci B 14:289–298
Sood P, Bhattacharya A, Sood A (2011) Problems and possibilities of monocot transformation. Biol Plant 55:1–15
Sunilkumar G, Rathore KS (2001) Transgenic cotton: factors influencing Agrobacterium-mediated transformation and regeneration. Mol Breed 8:37–52
Taylor PW, Ko HL, Adkins SW, Rathus C, Birch RG (1992) Establishment of embryogenic callus and high protoplast yielding suspension cultures of sugarcane (Saccharum spp. hybrids). Plant Cell Tissue Organ Cult 28:69–78
Tingay S, McElroy D, Kalla R, Fieg S, Wang M, Thornton S, Brettell R (1997) Agrobacterium tumefaciens-mediated barley transformation. Plant J 11:1369–1376
Tzfira T, Citovsky V (2006) Agrobacterium-mediated genetic transformation of plants: biology and biotechnology. Curr Opin Biotechnol 17:147–154
Vacher C, Kossler TM, Hochberg ME, Arthur E (2011) Weis impact of inter-specific hybridization between crops and weedy relatives on the evolution of flowering time in weedy phenotypes. PLoS One 6:e14649
Vyver CVD (2010) Genetic transformation of the euploid Saccharum officinarum via direct and indirect embryogenesis. Sugar Tech 12:21–25
Weng LX, Deng HH, Xu JL, Li Q, Zhang YQ, Jiang ZD, Li QW, Chen JW, Zhang LH (2011) Transgenic sugarcane plants expressing high levels of modified Cry1Ac provide effective control against stem borers in field trials. Transgenic Res 20:759–772
Xu LP, Que YX, Xu JS, Fang SR, Zhang MQ, Chen YQ, Chen RK (2008) Establishment of genetic transformation system and obtaining transgenic sugarcane (var. badila) transformed with RS gene. Sugar Tech 10:128–132
Zhao ZY, Cai T, Tagliani L, Miller M, Wang N, Pang H, Rudert M, Schroeder S, Hondred D, Seltzer J, Pierce D (2000) Agrobacterium-mediated sorghum transformation. Plant Mol Biol 44:789–798
Zhao ZY, Gu W, Cai T, Tagliani L, Hondred D, Bond D, Schroeder S, Rudert M, Pierce D (2001) High throughput genetic transformation mediated by Agrobacterium tumefaciens in maize. Mol Breed 8:323–333
Zucchi MI, Arizona H, Morais VA, Fungaro MHP, Vieira MLC (2002) Genetic instability of sugarcane plants derived from meristem cultures. Genet Mol Biol 25:91–96
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Anunanthini, P., Kumar, S.R., Sathishkumar, R. (2017). Factors Affecting Genetic Transformation Efficiency in Sugarcane. In: Mohan, C. (eds) Sugarcane Biotechnology: Challenges and Prospects. Springer, Cham. https://doi.org/10.1007/978-3-319-58946-6_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-58946-6_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-58945-9
Online ISBN: 978-3-319-58946-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)