Abstract
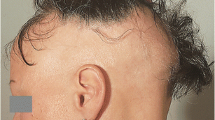
Alopecia areata (AA) is an autoimmune disease characterized by targeting of the hair follicle. Clinically, patients exhibit nonscarring hair loss with varying presentations across all age groups and follow an unpredictable course. Our understanding of the pathogenic mechanisms underlying AA has been greatly enhanced by recent large scale studies of genetic associations of disease. Descriptive studies in humans in tandem with mechanistic experiments in mice have helped define cellular and soluble disease drivers at the level of the end-organ target, the hair follicle, as well as the immune system.In AA, those mechanisms that protect the hair follicle from immune attack and maintain the immune privileged status of this site become disrupted and allow autoreactive cytotoxic immune cells to recognize and respond to self-antigens associated with the hair follicle.Our enhanced understanding of the disease has led to the identification of new therapeutic targets for AA.In particular, targeting of JAK molecules, proximal intermediates that transduce ligand binding signals for a wide variety of cytokine receptors, have shown promise in the treatment of this disease in animals models and humans.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Alkhalifah A, Alsantali A, Wang E, et al. Alopecia areata update: part I. Clinical picture, histopathology, and pathogenesis. J Am Acad Dermatol. 2010;62:177–88. – quiz 189–90. doi: 10.1016/j.jaad.2009.10.032.
Rosenthal T. Aulus cornelius celsus: his contributions to dermatology. Archives of Dermatology. 1961;84:613–18. doi:10.1001/archderm.1961.01580160077013.
McDonagh AJ, Tazi-Ahnini R. Epidemiology and genetics of alopecia areata. Clin Exp Dermatol. 2002;27:405–9.
van der Steen P, Traupe H, Happle R, Boezeman J, Sträter R, Hamm H. The genetic risk for alopecia areata in first degree relatives of severely affected patients. An estimate. Acta Derm Venereol. 1992;72:373–5.
Rodriguez TA, Fernandes KE, Dresser KL, Duvic M, National Alopecia Areata Registry. Concordance rate of alopecia areata in identical twins supports both genetic and environmental factors. J Am Acad Dermatol. 2010;62:525–7. doi:10.1016/j.jaad.2009.02.006.
Jackow C, Puffer N, Hordinsky M, Nelson J, Tarrand J, Duvic M. Alopecia areata and cytomegalovirus infection in twins: genes versus environment? J Am Acad Dermatol. 1998;38:418–25.
Sundberg JP, Silva KA, Li R, Cox GA, King LE. Adult-onset Alopecia areata is a complex polygenic trait in the C3H/HeJ mouse model. J Invest Dermatol. 2004;123:294–7. doi:10.1111/j.0022-202X.2004.23222.x.
Petukhova L, Duvic M, Hordinsky M, et al. Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature. 2010;466:113–7. doi:10.1038/nature09114.
Jagielska D, Redler S, Brockschmidt FF, et al. Follow-up study of the first genome-wide association scan in alopecia areata: IL13 and KIAA0350 as susceptibility loci supported with genome-wide significance. J Invest Dermatol. 2012;132:2192–7. doi:10.1038/jid.2012.129.
Betz RC, Petukhova L, Ripke S, et al. Genome-wide meta-analysis in alopecia areata resolves HLA associations and reveals two new susceptibility loci. Nat Commun. 2015;6:5966. doi:10.1038/NCOMMS6966.
Streilein JW. Ocular immune privilege: therapeutic opportunities from an experiment of nature. Nat Rev Immunol. 2003;3:879–89. doi:10.1038/nri1224.
Medawar PB. Immunity to homologous grafted skin; the fate of skin homografts transplanted to the brain, to subcutaneous tissue, and to the anterior chamber of the eye. Br J Exp Pathol. 1948;29:58–69.
Paus R, Ito N, Takigawa M, Ito T. The hair follicle and immune privilege. J Investig Dermatol Symp Proc. 2003;8:188–94. doi:10.1046/j.1087-0024.2003.00807.x.
Billingham RE, Boswell T. Studies on the problem of corneal homografts. Proc R Soc Lond B Biol Sci. 1953;141:392–406. doi:10.1098/rspb.1953.0049.
Barker CF, Billingham RE. Analysis of local anatomic factors that influence the survival times of pure epidermal and full-thickness skin homografts in guinea pigs. Ann Surg. 1972;176:597–604.
Billingham RE, Silvers WK. A biologist’s reflections on dermatology. J Invest Dermatol. 1971;57:227–40.
Reynolds AJ, Lawrence C, Cserhalmi-Friedman PB, et al. Trans-gender induction of hair follicles. Nature. 1999;402:33–4. doi:10.1038/46938.
Harrist TJ, Ruiter DJ, Mihm MC, Bhan AK. Distribution of major histocompatibility antigens in normal skin. Br J Dermatol. 1983;109:623–33. doi:10.1111/j.1365-2133.1983.tb00540.x.
Christoph T, Müller-Röver S, Audring H, Tobin DJ, Hermes B, Cotsarelis G, Rückert R, Paus R. The human hair follicle immune system: cellular composition and immune privilege. Br J Dermatol. 2000;142:862–73.
Ito T, Ito N, Saatoff M, et al. Maintenance of hair follicle immune privilege is linked to prevention of NK cell attack. J Invest Dermatol. 2008;128:1196–206. doi:10.1038/sj.jid.5701183.
Apte RS, Sinha D, Mayhew E, et al. Cutting edge: role of macrophage migration inhibitory factor in inhibiting NK cell activity and preserving immune privilege. J Immunol. 1998;160:5693–6.
Foitzik K, Lindner G, Mueller-Roever S, et al. Control of murine hair follicle regression (catagen) by TGF-β1 in vivo. FASEB J. 2000;14:752–60. doi:10.1096/fj.1530-6860.
Slominski A, Wortsman J, Mazurkiewicz JE, et al. Detection of proopiomelanocortin- derived antigens in normal and pathologic human skin. J Lab Clin Med. 1993;122:658–66.
Nanninga PB, Ghanem GE, Lejeune FJ, et al. Evidence for alpha-MSH binding sites on human scalp hair follicles: preliminary results. Pigment Cell Res. 1991;4:193–8.
Wang X, Marr AK, Breitkopf T, et al. Hair follicle mesenchyme-associated PD-L1 regulates T-cell activation induced apoptosis: a potential mechanism of immune privilege. J Invest Dermatol. 2014;134:736–45. doi:10.1038/jid.2013.368.
Paus R, Slominski A, Czarnetzki BM. Is alopecia areata an autoimmune-response against melanogenesis-related proteins, exposed by abnormal MHC class I expression in the anagen hair bulb? Yale J Biol Med. 1993;66:541–54.
Moresi JM, Horn TD. Distribution of Langerhans cells in human hair follicle. J Cutan Pathol. 1997;24:636–40. doi:10.1111/j.1600-0560.1997.tb01095.x.
Bröcker EB, Echternacht-Happle K, Hamm H, Happle R. Abnormal expression of class I and class II major histocompatibility antigens in alopecia areata: modulation by topical immunotherapy. J Invest Dermatol. 1987;88:564–8.
Todes-Taylor N, Turner R, Wood GS, et al. T cell subpopulations in alopecia areata. J Am Acad Dermatol. 1984;11:216–23.
**ng L, Dai Z, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043–9. doi:10.1038/nm.3645.
Sundberg JP, Cordy WR, King LE. Alopecia areata in aging C3H/HeJ mice. J Invest Dermatol. 1994;102:847–56. doi:10.1111/1523-1747.ep12382416.
Carroll JM, McElwee KJ, E King L, et al. Gene array profiling and immunomodulation studies define a cell-mediated immune response underlying the pathogenesis of alopecia areata in a mouse model and humans. J Invest Dermatol. 2002;119:392–402. doi:10.1046/j.1523-1747.2002.01811.x.
Gilhar A, Keren A, Shemer A, et al. Autoimmune disease induction in a healthy human organ: a humanized mouse model of alopecia areata. J Invest Dermatol. 2013;133:844–7. doi:10.1038/jid.2012.365.
Bauer S, Groh V, Wu J, Steinle A, Phillips JH, Lanier LL, Spies T. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science. 1991;285:727–9.
Groh V, Rhinehart R, Randolph-Habecker J, Topp MS, Riddell SR, Spies T. Costimulation of CD8alphabeta T cells by NKG2D via engagement by MIC induced on virus-infected cells. Nat Immunol. 2001;2:255–60. doi:10.1038/85321.
McElwee KJ, Freyschmidt-Paul P, Hoffmann R, et al. Transfer of CD8(+) cells induces localized hair loss whereas CD4(+)/CD25(-) cells promote systemic alopecia areata and CD4(+)/CD25(+) cells blockade disease onset in the C3H/HeJ mouse model. J Invest Dermatol. 2005;124:947–57. doi:10.1111/j.0022-202X.2005.23692.x.
Tembhre MK, Sharma VK. T-helper and regulatory T-cell cytokines in the peripheral blood of patients with active alopecia areata. Br J Dermatol. 2013;169:543–8. doi:10.1111/bjd.12396.
Kaufman G, d’Ovidio R, Kaldawy A, et al. An unexpected twist in alopecia areata pathogenesis: are NK cells protective and CD49b + T cells pathogenic? Exp Dermatol. 2010;19:e347–9. doi:10.1111/j.1600-0625.2010.01106.x.
Bertolini M, Zilio F, Rossi A, et al. Abnormal interactions between perifollicular mast cells and CD8+ T-cells may contribute to the pathogenesis of alopecia areata. PLoS One. 2014;9:e94260. doi:10.1371/journal.pone.0094260.
Wong GH, Clark-Lewis I, McKimm-Breschkin JL, Schrader JW. Interferon-gamma-like molecule induces Ia antigens on cultured mast cell progenitors. Proc Natl Acad Sci U S A. 1982;79:6989–93.
Banovac K, Neylan D, Leone J, et al. Are the mast cells antigen presenting cells? Immunol Invest. 1989;18:901–6.
Rumsaeng V, Cruikshank WW, Foster B, et al. Human mast cells produce the CD4+ T lymphocyte chemoattractant factor, IL-16. J Immunol. 1997;159:2904–10.
Castellana D, Paus R, Perez-Moreno M. Macrophages contribute to the cyclic activation of adult hair follicle stem cells. PLos Biol. 2014;12:e1002002. doi:10.1371/journal.pbio.1002002.
Gilhar A, Kam Y, Assy B, Kalish RS. Alopecia areata induced in C3H/HeJ mice by interferon-gamma: evidence for loss of immune privilege. J Invest Dermatol. 2005;124:288–9. doi:10.1111/j.0022-202X.2004.23580.x.
Sundberg JP, Silva KA, Edwards K, et al. Failure to induce alopecia areata in C3H/HeJ mice with exogenous interferon gamma. J Exp Anim Sci. 2007;43:265–70. doi:10.1016/j.jeas.2006.10.005.
Freyschmidt Paul P, McElwee KJ, Hoffmann R, et al. Interferon-γ-deficient mice are resistant to the development of alopecia areata. Br J Dermatol. 2006;155:515–21. doi:10.1111/j.1365-2133.2006.07377.x.
Agesta N, Zabala R, Diaz-Perez JL. Alopecia areata during interferon alpha-2b/ribavirin therapy. Dermatology. 2002;205:300–1. doi:10.1159/000065841.
Taliani G, Biliotti E, Capanni M, et al. Reversible alopecia universalis during treatment with PEG-interferon and ribavirin for chronic hepatitis C. J Chemother. 2005;17:212–4. doi:10.1179/joc.2005.17.2.212.
Ghoreishi M, Martinka M, Dutz JP. Type 1 interferon signature in the scalp lesions of alopecia areata. Br J Dermatol. 2010;163:57–62. doi:10.1111/j.1365-2133.2010.09775.x.
Meresse B, Chen Z, Ciszewski C, et al. Coordinated induction by IL15 of a TCR- independent NKG2D signaling pathway converts CTL into lymphokine-activated killer cells in celiac disease. Immunity. 2004;21:357–66. doi:10.1016/j.immuni.2004.06.020.
Freyschmidt-Paul P, McElwee KJ, Hoffmann R, et al. Reduced expression of interleukin-2 decreases the frequency of alopecia areata onset in C3H/HeJ mice. J Invest Dermatol. 2005;125:945–51. doi:10.1111/j.0022-202X.2005.23888.x.
Abadie V, Jabri B. IL-15: a central regulator of celiac disease immunopathology. Immunol Rev. 2014;260:221–34. doi:10.1111/imr.12191.
Meresse B, Malamut G, Cerf-Bensussan N. Celiac disease: an immunological jigsaw. Immunity. 2012;36:907–19. doi:10.1016/j.immuni.2012.06.006.
Waldmann TA. The biology of IL-15: implications for cancer therapy and the treatment of autoimmune disorders. J Investig Dermatol Symp Proc. 2013;16:S28–30. doi:10.1038/jidsymp.2013.8.
Leung MC, Sutton CW, Fenton DA, Tobin DJ. Trichohyalin is a potential major autoantigen in human alopecia areata. J Proteome Res. 2010;9:5153–63. doi:10.1021/pr100422u.
Delamere FM, Sladden MM, Dobbins HM, Leonardi-Bee J. Interventions for alopecia areata. Cochrane Database Syst Rev. 2008;2:CD004413. doi:10.1002/14651858.CD004413.pub2.
Alkhalifah A, Alsantali A, Wang E, et al. Alopecia areata update: part II. Treatment. J Am Acad Dermatol. 2010;62:191–202. – quiz 203–4. doi:10.1016/j.jaad.2009.10.031.
Rokhsar CK, Shupack JL, Vafai JJ, Washenik K. Efficacy of topical sensitizers in the treatment of alopecia areata. J Am Acad Dermatol. 1998;39:751–61.
Happle R. Antigenic competition as a therapeutic concept for alopecia areata. Arch Dermatol Res. 1980;267:109–14.
Wasyłyszyn T, Kozłowski W, Zabielski SL. Changes in distribution pattern of CD8 lymphocytes in the scalp in alopecia areata during treatment with diphencyprone. Arch Dermatol Res. 2007;299:231–7. doi:10.1007/s00403-007-0759-4.
Happle R, Klein HM, Macher E. Topical immunotherapy changes the composition of the peribulbar infiltrate in alopecia areata. Arch Dermatol Res. 1986;278:214–8.
Ali J, Jane EC, James CC, Julian MW, Madeleine D, Vera P, Maria H, David N, Raphael C, Angela MC. Molecular signatures define alopecia areata subtypes and transcriptional biomarkers. doi: http://dx.doi.org/10.1016/j.ebiom.2016.03.036.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Questions
Questions
-
1.
A class of medications that has recently shown promise in AA:
-
A.
NSAIDs
-
B.
TNF inhibitors
-
C.
Interferons
-
D.
JAK inhibitors
-
E.
ACE inhibitors
-
A.
-
2.
These cells are not commonly found in the peribulbar infiltrate in skin biopsies sections of AA lesions:
-
A.
Plasma cells
-
B.
CD4 T cells
-
C.
CD8 T cells
-
D.
NK cells
-
E.
Macrophages
-
A.
-
3.
Activation of this pathway is associated with enhanced immune responses to the hair follicle:
-
A.
Interferon-γ
-
B.
IL-2
-
C.
TGF-β
-
D.
all of the above
-
E.
A and B only
-
A.
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Jabbari, A., Petukhova, L., Christiano, A.M. (2017). Alopecia Areata. In: Gaspari, A., Tyring, S., Kaplan, D. (eds) Clinical and Basic Immunodermatology. Springer, Cham. https://doi.org/10.1007/978-3-319-29785-9_29
Download citation
DOI: https://doi.org/10.1007/978-3-319-29785-9_29
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-29783-5
Online ISBN: 978-3-319-29785-9
eBook Packages: MedicineMedicine (R0)