Abstract
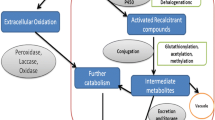
Phenolic compounds are potential pollutants to the environment, and their harmful effects are enormous. The antimicrobial nature of most of these compounds is making them invulnerable to the depuration process. Fungi can be successfully used for degradation as they are less prone to phenol toxicity and actively produce enzymes capable of degrading cyclic ring compounds. They have extraordinary capabilities of phenol removal when compared with other microbes such as bacteria. This chapter explores the applications of fungi in phenol biodegradation, phenol degradation pathways, commonly used fungal species, bioreactor models, immobilization techniques, and entrepreneurial aspects of fungal bioremediation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Acevedo, F., Pizzul, L., Castillo, M. D., Cuevas, R., & Diez, M. C. (2011). Degradation of polycyclic aromatic hydrocarbons by the Chilean white-rot fungus Anthracophyllum discolor. Journal of Hazardous Materials, 185, 212–219.
Anastasi, A., Tigini, V., & Varese, & G.C. (2013). The bioremediation potential of different ecophysiological groups of fungi. In Fungi as bioremediators (Soil biology) (Vol. 32, pp. 29–49). Springer.
Arisoy, M. (1998). Biodegradation of chlorinated organic compounds by white-rot fungi. Bulletin of Environmental Contamination and Toxicology, 60, 872–876.
Basha, K. M., Rajendran, A., & Thangavelu, V. (2010). Recent advances in the biodegradation of phenol: A review. Asian Journal of Experimental Biological Sciences, 1(2), 219–234.
Bhalerao, T. S., & Puranik, P. (2007). Biodegradation of organochlorine pesticide, endosulfan, by a fungal soil isolate, Aspergillus niger. International Biodeterioration and Biodegradation, 59, 315–321.
Cassimjee, K. E., Kadow, M., Wikmark, Y., Humble, M. S., Rothstein, M. L., Rothstein, D. M., & Bäckvall, J. E. (2014). A general protein purification and immobilization method on controlled porosity glass: Biocatalytic applications. Chemical Communications, 50(65), 9134–9137.
Chhaya, U., & Gupte, A. (2013). Possible role of laccase from Fusarium incarnatum UC-14 In bioremediation of Bisphenol A using reverse micelles system. Journal of Hazardous Materials, 254–255, 149–156. https://doi.org/10.1016/j.jhazmat.2013.03.054
Cobas, M., Ferreira, L., Tavares, T., Sanroman, M. A., & Pazos, M. (2013). Development of permeable reactive biobarrier for the removal of PAHs by Trichoderma longibrachiatum. Chemosphere, 91, 711–716. https://doi.org/10.1016/j.chemosphere.2013.01.028
Emergency Planning and Community Right-to-Know Act (EPCRA) Section 313 Chemical list for reporting year 2014. Available from: http://www.epa.gov/toxics-release-inventory-tri-program/tri-chemical-list-ry-2014-including-toxic-chemical-categories
Evans, W. C. (1947). Oxidation of phenol and benzoic acid by some soil bacteria. The Biochemical Journal, 41(3), 373.
Fernandez-Fernandez, M., Sanromán, M. Á., & Moldes, D. (2013). Recent developments and applications of immobilized laccase. Biotechnology Advances, 31(8), 1808–1825.
Gray, C. J., Weissenborn, M. J., Eyers, C. E., & Flitsch, S. L. (2013). Enzymatic reactions on immobilised substrates. Chemical Society Reviews, 42(15), 6378–6405.
Harris, G., & Ricketts, R. (1962). Metaboilsm of phenolic compounds by yeasts. Nature, 195, 473–474.
Harwood, C. S., & Parales, R. E. (1996). The β-ketoadipate pathway and the biology of self-identity. Annual Review of Microbiology, 50(1), 553–590.
Henderson, M. E. K. (1961). The metabolism of aromatic compounds related to lignin by some Hyphomycetes and yeast-like fungi of soil. Journal of General Microbiology, 26, 155–165.
Holladay, D. W., Hancher, C. W., Scott, C. D., & Chilcote, D. D. (1978). Biodegradation of phenolic waste liquors in stirred-tank, packed-bed, and fluidized-bed bioreactors. Journal – Water Pollution Control Federation, 2573–2589.
Huang, Y., Zhang, S. Y., Lv, M. J., & **e, S. G. (2010). Biosorption characteristics of ectomycorrhizal fungal mycelium for anthracene. Biomedical and Environmental Sciences, 23, 378–383.
Jebapriya, G. R., & Gnanadoss, J. J. (2013). Bioremediation of textile dye using white-rot fungi: A review. International Journal of Current Research and Review, 5, 1–13.
Karel, S. F., Libicki, S. B., & Robertson, C. R. (1985). The immobilization of whole cells: Engineering principles. Chemical Engineering Science, 40(8), 1321–1354.
Kilby, B. A. (1948). The bacterial oxidation of phenol to beta-ketoadipic acid. The Biochemical Journal, 43, 1.
Lamar, R. T., & Dietrich, D. M. (1990). In situ depletion of pentachlorophenol from contaminated soil by Phanerochaete spp. Applied and Environmental Microbiology, 56(10), 3093–3100.
Lamar, R. T., & Dietrich, D. M. (1992). Use of lignin-degrading fungi in the disposal of pentachlorophenol-treated wood. Journal of Industrial Microbiology, 9, 181–191.
Lamar, R. T., Davis, M. W., Dietrich, D. M., & Glaser, J. A. (1994). Treatment of a pentachlorophenol- and creosote-contaminated soil using the lignin-degrading fungus Phanerochaetesordida: A field demonstration. Soil Biology and Biochemistry, 26(12), 1603–1616.
Lang, E., E’Uer, G., Kleeberg, I., Martens, R., & Zadrazil, F. (1995). Interaction of white rot fungi and soil microorganisms leading to biodegradation of soil pollutants. In Contaminated Soil’95 (Soil and environment) (Vol. 5, pp. 1277–1278). Springer.
León-Santiesteban, H. H., Wrobel, K., Revah, S., & Tomasini, A. (2016). Pentachlorophenol removal by Rhizopus oryzae CDBB-H-1877 using sorption and degradation mechanisms. Journal of Chemical Technology & Biotechnology, 91, 65–71.
Machado, K. M. G., Matheus, D. R., Monteiro, R. T. R., & Bononi, V. L. R. (2005). Biodegradation of pentachorophenol by tropical basidiomycetes in journal of environmental science and health, part B 153 soils contaminated with industrial residues. World Journal of Microbiology and Biotechnology, 21, 297–301.
Mahiudddin, M., & Fakhruddin, A. N. M. (2012). Degradation of phenol via meta cleavage pathway by Pseudomonas fluorescens PU1. ISRN Microbiology, 2012, 741820.
Mansur, M., Arias, M. E., Copa-Patino, J. L., Flardh, M., & Gonzalez, A. E. (2017). The white-rot fungus Pleurotusostreatus secretes laccase isozymes with different substrate specificities. Mycologia, 95, 1013–1020.
Margot, J., Bennati-Granier, C., Maillard, J., Blanquez, P., Barry, D. A., & Holliger, C. (2013). Bacterial versus fungal laccase: Potential for micropollutant degradation. AMB Express, 3, 63. https://doi.org/10.1186/2191-0855-3-63
Meharg, A. A., Cairney, J. W. G., & Maguire, N. (1997). Mineralization of 2,4-dichlorophenol by ectomycorrhizal fungi in axenic culture and in symbiosis with pine. Chemosphere, 34(12), 2495–2504.
Mouhamadou, B., Faure, M., Sage, L., Marçais, J., Souard, F., & Geremia, R. A. (2013). Potential of autochthonous fungal strains isolated from contaminated soils for degradation of polychlorinated biphenyls. Fungal Biology, 117, 268–274. https://doi.org/10.1016/j.funbio.2013.02.004
Neujahr, H. Y., & Varga, J. M. (1970). Degradation of phenols by intake cells and cell-free preparations of Trichosporoncutaneum. European Journal of Biochemistry, 13, 37–44.
Pointing, S. B. (2001). Feasibility of bioremediation by white-rot fungi. The Applied Microbiology and Biotechnology, 51, 20–33.
Rahman, R. A., Molla, A. H., & Fakhru’l-Razi, A. (2014). Assessment of sewage sludge bioremediation at different hydraulic retention times using mixed fungal inoculation by liquid-state bioconversion. Environmental Science and Pollution Research, 21, 1178–1187. https://doi.org/10.1007/s11356-013-1974-5
Reddy, C. A., & Mathew, Z. (2001). Bioremediation potential of white rot fungi. In British mycological society symposium series (Vol. 23, pp. 52–78).
Rodríguez-Rodríguez, C. E., Castro-Gutiérrez, V., Chin-Pampillo, J. S., & Ruiz-Hidalgo, K. (2013). On-farm biopurificationsystems: Role of white-rot fungi in depuration of pesticide-containing wastewaters. FEMS Microbiology Letters, 345, 1–12. https://doi.org/10.1111/1574-6968.12161
Sack, U., Heinze, T. M., Deck, J., Cerniglia, C. E., Martens, R., Zadrazil, F., & Fritsche, W. (1997). Comparison of phenanthrene and pyrene degradation by different wood-decaying fungi. Applied and Environmental Biology, 63(10), 3919–3925.
Santos, V. L., & Linardi, V. R. (2001). Phenol degradation by yeasts isolated from industrial effluents. Journal of General and Applied Microbiology, 47(4), 213–221.
Singh, L., & Singh, V. P. (2011). Microbial decolourization of textile dyes by the fungus Trichoderma harzianum. Journal of Pure and Applied Microbiology, 6, 1829–1833.
Stainer, R. Y., & Ornston, L. N. (1973). The β-ketoadipate pathway. In Advances in microbial physiology (Vol. 9, pp. 89–151). Academic.
Teramura, Y., Kaneda, Y., Totani, T., & Iwata, H. (2008). Behavior of synthetic polymers immobilized on a cell membrane. Biomaterials, 29(10), 1345–1355.
Tigini, V., Prigione, V., Di, T., Fava, F., & Varese, G. C. (2009). Isolation and characterisation of polychlorinated biphenyl (PCB) degrading fungi from a historically contaminated soil. Microbial Cell Factories, 8(1), 1–14.
Tortella, G. R., Rubilar, O., Gianfreda, L., Valenzuela, E., & Diez, M. C. (2008). Enzymatic characterization of Chilean native wood-rotting fungi for potential use in the bioremediation of polluted environments with chlorophenols. World Journal of Microbiology and Biotechnology, 24, 2805–2818.
Valentín, L., Oesch-Kuisma, H., Steffen, K. T., Kähkönen, M. A., Hatakka, A., & Tuomela, M. (2013). Mycoremediation of wood and soil from an old sawmill area contaminated for decades. Journal of Hazardous Materials, 260, 668–675.
Villegas, L. G. C., Mashhadi, N., Chen, M., Mukherjee, D., Taylor, K. E., & Biswas, N. (2016). A short review of techniques for phenol removal from wastewater. Current Pollution Reports, 2(3), 157–167.
Yordanova, G., Godjevargova, T., Nenkova, R., & Ivanova, D. (2013). Biodegradation of phenol and phenolic derivatives by a mixture of immobilized cells of Aspergillus awamori and Trichosporoncutaneum. Biotechnology & Biotechnological Equipment, 27(2), 3681–3688.
Zaushitsyna, O., Berillo, D., Kirsebom, H., & Mattiasson, B. (2014). Cryostructured and cross-linked viable cells forming monoliths suitable for bioreactor applications. Topics in Catalysis, 57(5), 339–348.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Sasi, R., Zachariah, S., Suchithra, T.V. (2022). The Untapped Potential of Fungi in Phenol Biodegradation. In: Shukla, A.C. (eds) Applied Mycology. Fungal Biology. Springer, Cham. https://doi.org/10.1007/978-3-030-90649-8_19
Download citation
DOI: https://doi.org/10.1007/978-3-030-90649-8_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-90648-1
Online ISBN: 978-3-030-90649-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)