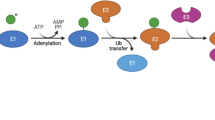
Abstract
Enzymatic post-translational modifications (PTMs) regulate the structure, function, and stability of proteins that are involved in a myriad of vital cellular processes. Incorporation of these modifications onto substrate proteins creates additional complexity to the already expansive proteome, which further hinders efforts for complete functional annotation. Importantly, dysregulation to the activity of PTMs has been implicated in various pathologies such as inflammatory, autoimmune, cardiovascular, and neurodegenerative diseases as well as several types of cancers. Over recent decades, advancements to the experimental techniques employed to study post-translational modifications have considerably expanded the understanding of the functional role they impart on biological processes on both a single-protein basis and a global level. This chapter first provides an overview of the prominent PTM types with a focus on their respective enzymatic regulators, substrate proteins, and their roles in the onset and progression of cancer. The current experimental approaches used to study PTMs such as chemical labeling, unnatural amino acid incorporation, biophysical-based imaging, and mass spectrometry-based proteomics are also highlighted along with a brief discussion of their limitations. Finally, the current trends within the field and a perspective on future work are presented.
Similar content being viewed by others
References
Aebersold R, Agar JN, Amster IJ, Baker MS, Bertozzi CR, Boja ES, Costello CE, Cravatt BF, Fenselau C, Garcia BA, Ge Y, Gunawardena J, Hendrickson RC, Hergenrother PJ, Huber CG, Ivanov AR, Jensen ON, Jewett MC, Kelleher NL, Kiessling LL, Krogan NJ, Larsen MR, Loo JA, Ogorzalek Loo RR, Lundberg E, MacCoss MJ, Mallick P, Mootha VK, Mrksich M, Muir TW, Patrie SM, Pesavento JJ, Pitteri SJ, Rodriguez H, Saghatelian A, Sandoval W, Schlüter H, Sechi S, Slavoff SA, Smith LM, Snyder MP, Thomas PM, Uhlén M, Van Eyk JE, Vidal M, Walt DR, White FM, Williams ER, Wohlschlager T, Wysocki VH, Yates NA, Young NL, Zhang B (2018) How many human proteoforms are there? Nat Chem Biol 14(3):206–214. https://doi.org/10.1038/nchembio.2576
Al-Khouri AM, Ma Y, Togo SH, Williams S, Mustelin T (2005) Cooperative phosphorylation of the tumor suppressor phosphatase and tensin homologue (PTEN) by casein kinases and glycogen synthase kinase 3β. J Biol Chem 280(42):35195–35202. https://doi.org/10.1074/jbc.M503045200
Allen JJ, Li M, Brinkworth CS, Paulson JL, Wang D, Hübner A, Chou W-H, Davis RJ, Burlingame AL, Messing RO, Katayama CD, Hedrick SM, Shokat KM (2007) A semisynthetic epitope for kinase substrates. Nat Methods 4(6):511–516. https://doi.org/10.1038/nmeth1048
Ayrapetov MK, Gursoy-Yuzugullu O, Xu C, Xu Y, Price BD (2014) DNA double-strand breaks promote methylation of histone H3 on lysine 9 and transient formation of repressive chromatin. Proc Natl Acad Sci U S A 111(25):9169–9174. https://doi.org/10.1073/pnas.1403565111
Baskin JM, Prescher JA, Laughlin ST, Agard NJ, Chang PV, Miller IA, Lo A, Codelli JA, Bertozzi CR (2007) Copper-free click chemistry for dynamic in vivo imaging. Proc Natl Acad Sci 104(43):16793–16797. https://doi.org/10.1073/pnas.0707090104
Belardi B, de la Zerda A, Spiciarich DR, Maund SL, Peehl DM, Bertozzi CR (2013) Imaging the glycosylation state of cell surface glycoproteins by two-photon fluorescence lifetime imaging microscopy. Angew Chem Int Ed 52(52):14045–14049. https://doi.org/10.1002/anie.201307512
Bhattacharya U, Neizer-Ashun F, Mukherjee P, Bhattacharya R (2020) When the chains do not break: the role of USP10 in physiology and pathology. Cell Death Dis 11(12):1033. https://doi.org/10.1038/s41419-020-03246-7
Blackman ML, Royzen M, Fox JM (2008) Tetrazine ligation: fast bioconjugation based on inverse-electron-demand Diels−Alder reactivity. J Am Chem Soc 130(41):13518–13519. https://doi.org/10.1021/ja8053805
Bremang M, Cuomo A, Agresta AM, Stugiewicz M, Spadotto V, Bonaldi T (2013) Mass spectrometry-based identification and characterisation of lysine and arginine methylation in the human proteome. Mol BioSyst 9(9):2231–2247. https://doi.org/10.1039/c3mb00009e
Burnaevskiy N, Fox TG, Plymire DA, Ertelt JM, Weigele BA, Selyunin AS, Way SS, Patrie SM, Alto NM (2013) Proteolytic elimination of N-myristoyl modifications by the Shigella virulence factor IpaJ. Nature 496(7443):106–109. https://doi.org/10.1038/nature12004
Buuh ZY, Lyu Z, Wang RE (2018) Interrogating the roles of post-translational modifications of non-histone proteins. J Med Chem 61(8):3239–3252. https://doi.org/10.1021/acs.jmedchem.6b01817
Caldwell SA, Jackson SR, Shahriari KS, Lynch TP, Sethi G, Walker S, Vosseller K, Reginato MJ (2010) Nutrient sensor O-GlcNAc transferase regulates breast cancer tumorigenesis through targeting of the oncogenic transcription factor FoxM1. Oncogene 29(19):2831–2842. https://doi.org/10.1038/onc.2010.41
Catherman AD, Skinner OS, Kelleher NL (2014) Top down proteomics: facts and perspectives. Biochem Biophys Res Commun 445(4):683–693. https://doi.org/10.1016/j.bbrc.2014.02.041
Charron G, Tsou LK, Maguire W, Yount JS, Hang HC (2011) Alkynyl-farnesol reporters for detection of protein S-prenylation in cells. Mol BioSyst 7(1):67–73. https://doi.org/10.1039/C0MB00183J
Chaube R (2014) Absolute quantitation of post-translational modifications. Front Chem 2. https://doi.org/10.3389/fchem.2014.00058
Chen H, Venkat S, McGuire P, Gan Q, Fan C (2018) Recent development of genetic code expansion for posttranslational modification studies. Molecules 23(7):1662. https://doi.org/10.3390/molecules23071662
Choudhary C, Kumar C, Gnad F, Nielsen Michael L, Rehman M, Walther Tobias C, Olsen Jesper V, Mann M (2009) Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science 325(5942):834–840. https://doi.org/10.1126/science.1175371
Choudhary C, Weinert BT, Nishida Y, Verdin E, Mann M (2014) The growing landscape of lysine acetylation links metabolism and cell signalling. Nat Rev Mol Cell Biol 15(8):536–550. https://doi.org/10.1038/nrm3841
Cicenas J (2008) The potential role of akt phosphorylation in human cancers. Int J Biol Marker 23(1):1–9. https://doi.org/10.1177/172460080802300101
Collins MO, Yu L, Choudhary JS (2007) Analysis of protein phosphorylation on a proteome-scale. Proteomics 7(16):2751–2768. https://doi.org/10.1002/pmic.200700145
Conze T, Carvalho AS, Landegren U, Almeida R, Reis CA, David L, Söderberg O (2010) MUC2 mucin is a major carrier of the cancer-associated sialyl-Tn antigen in intestinal metaplasia and gastric carcinomas. Glycobiology 20(2):199–206. https://doi.org/10.1093/glycob/cwp161
Deng M, Yang X, Qin B, Liu T, Zhang H, Guo W, Lee Seung B, Kim Jung J, Yuan J, Pei H, Wang L, Lou Z (2016) Deubiquitination and activation of AMPK by USP10. Mol Cell 61(4):614–624. https://doi.org/10.1016/j.molcel.2016.01.010
Deng L, Meng T, Chen L, Wei W, Wang P (2020) The role of ubiquitination in tumorigenesis and targeted drug discovery. Signal Transduct Target Ther 5(1):11. https://doi.org/10.1038/s41392-020-0107-0
Doll F, Steimbach RR, Zumbusch A (2019) Direct imaging of protein-specific methylation in mammalian cells. Chembiochem 20(10):1315–1325. https://doi.org/10.1002/cbic.201800787
Dupree EJ, Jayathirtha M, Yorkey H, Mihasan M, Petre BA, Darie CC (2020) A critical review of bottom-up proteomics: the good, the bad, and the future of this field. Proteomes 8(3). https://doi.org/10.3390/proteomes8030014
Fernández-Suárez M, Baruah H, Martínez-Hernández L, **e KT, Baskin JM, Bertozzi CR, Ting AY (2007) Redirecting lipoic acid ligase for cell surface protein labeling with small-molecule probes. Nat Biotechnol 25(12):1483–1487. https://doi.org/10.1038/nbt1355
Ganesan S, Ameer-beg SM, Ng TTC, Vojnovic B, Wouters FS (2006) A dark yellow fluorescent protein (YFP)-based Resonance Energy-Accepting Chromoprotein (REACh) for Förster resonance energy transfer with GFP. Proc Natl Acad Sci 103(11):4089–4094. https://doi.org/10.1073/pnas.0509922103
Gao X, Hannoush RN (2014) Single-cell imaging of Wnt palmitoylation by the acyltransferase porcupine. Nat Chem Biol 10(1):61–68. https://doi.org/10.1038/nchembio.1392
George P, Bali P, Annavarapu S, Scuto A, Fiskus W, Guo F, Sigua C, Sondarva G, Moscinski L, Atadja P, Bhalla K (2005) Combination of the histone deacetylase inhibitor LBH589 and the hsp90 inhibitor 17-AAG is highly active against human CML-BC cells and AML cells with activating mutation of FLT-3. Blood 105(4):1768–1776. https://doi.org/10.1182/blood-2004-09-3413
Grammel M, Hang HC (2013) Chemical reporters for biological discovery. Nat Chem Biol 9(8):475–484. https://doi.org/10.1038/nchembio.1296
Guo H, Zhang B, Nairn AV, Nagy T, Moremen KW, Buckhaults P, Pierce M (2017) O-linked N-acetylglucosamine (O-GlcNAc) expression levels epigenetically regulate colon cancer tumorigenesis by affecting the cancer stem cell compartment via modulating expression of transcriptional factor MYBL1. J Biol Chem 292(10):4123–4137. https://doi.org/10.1074/jbc.M116.763201
Hamamoto R, Saloura V, Nakamura Y (2015) Critical roles of non-histone protein lysine methylation in human tumorigenesis. Nat Rev Cancer 15(2):110–124. https://doi.org/10.1038/nrc3884
Han Z, Chou C-w, Yang X, Bartlett MG, Zheng YG (2017) Profiling cellular substrates of lysine acetyltransferases GCN5 and p300 with orthogonal labeling and click chemistry. ACS Chem Biol 12(6):1547–1555. https://doi.org/10.1021/acschembio.7b00114
Hang HC, Yu C, Kato DL, Bertozzi CR (2003) A metabolic labeling approach toward proteomic analysis of mucin-type O-linked glycosylation. Proc Natl Acad Sci 100(25):14846–14851. https://doi.org/10.1073/pnas.2335201100
Hong V, Steinmetz NF, Manchester M, Finn MG (2010) Labeling live cells by copper-catalyzed alkyne−Azide click chemistry. Bioconjug Chem 21(10):1912–1916. https://doi.org/10.1021/bc100272z
Islam K, Zheng W, Yu H, Deng H, Luo M (2011) Expanding cofactor repertoire of protein lysine methyltransferase for substrate labeling. ACS Chem Biol 6(7):679–684. https://doi.org/10.1021/cb2000567
Jaggi M, Rao PS, Smith DJ, Wheelock MJ, Johnson KR, Hemstreet GP, Balaji KC (2005) E-cadherin phosphorylation by protein kinase D1/protein kinase Cμ is associated with altered cellular aggregation and motility in prostate cancer. Cancer Res 65(2):483–492
Jarvius M, Paulsson J, Weibrecht I, Leuchowius K-J, Andersson A-C, Wählby C, Gullberg M, Botling J, Sjöblom T, Markova B, Östman A, Landegren U, Söderberg O (2007) In situ detection of phosphorylated platelet-derived growth factor receptor β using a generalized proximity ligation method*. Mol Cell Proteomics 6(9):1500–1509. https://doi.org/10.1074/mcp.M700166-MCP200
Jiao J, Han R, Hancock WW, Beier UH (2017) Proximity ligation assay to quantify Foxp3 acetylation in regulatory T cells. In: Krämer OH (ed) HDAC/HAT function assessment and inhibitor development: methods and protocols. Methods in molecular biology. Springer, New York, pp 287–293
Kazemi Z, Chang H, Haserodt S, McKen C, Zachara NE (2010) O-linked β-N-acetylglucosamine (O-GlcNAc) regulates stress-induced heat shock protein expression in a GSK-3β-dependent manner. J Biol Chem 285(50):39096–39107. https://doi.org/10.1074/jbc.M110.131102
Kim SC, Sprung R, Chen Y, Xu Y, Ball H, Pei J, Cheng T, Kho Y, **ao H, **ao L, Grishin NV, White M, Yang XJ, Zhao Y (2006) Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol Cell 23(4):607–618. https://doi.org/10.1016/j.molcel.2006.06.026
Kim W, Bennett EJ, Huttlin EL, Guo A, Li J, Possemato A, Sowa ME, Rad R, Rush J, Comb MJ, Harper JW, Gygi SP (2011) Systematic and quantitative assessment of the ubiquitin-modified proteome. Mol Cell 44(2):325–340. https://doi.org/10.1016/j.molcel.2011.08.025
Kolb HC, Finn MG, Sharpless KB (2001) Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed 40(11):2004–2021. https://doi.org/10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5
Kovacs JJ, Murphy PJM, Gaillard S, Zhao X, Wu JT, Nicchitta CV, Yoshida M, Toft DO, Pratt WB, Yao TP (2005) HDAC6 regulates Hsp90 acetylation and chaperone-dependent activation of glucocorticoid receptor. Mol Cell 18(5):601–607. https://doi.org/10.1016/j.molcel.2005.04.021
Kulathu Y, Komander D (2012) Atypical ubiquitylation — the unexplored world of polyubiquitin beyond Lys48 and Lys63 linkages. Nat Rev Mol Cell Biol 13(8):508–523. https://doi.org/10.1038/nrm3394
Laughlin ST, Baskin JM, Amacher SL, Bertozzi CR (2008) In vivo imaging of membrane-associated glycans in develo** zebrafish. Science 320(5876):664–667. https://doi.org/10.1126/science.1155106
Leutert M, Entwisle SW, Villén J (2021) Decoding post-translational modification crosstalk with proteomics. Mol Cell Proteomics 20:100129. https://doi.org/10.1016/j.mcpro.2021.100129
Li W, He Y, Yang J, Hu G, Lin Y, Ran T, Peng B, **e B, Huang M, Gao X, Huang H, Zhu HH, Ye F, Liu W (2021) Profiling PRMT methylome reveals roles of hnRNPA1 arginine methylation in RNA splicing and cell growth. Nat Commun 12(1):1946. https://doi.org/10.1038/s41467-021-21963-1
Lin W, Gao L, Chen X (2015) Protein-specific imaging of posttranslational modifications. Curr Opin Chem Biol 28:156–163. https://doi.org/10.1016/j.cbpa.2015.07.020
Lin DC, Xu L, Chen Y, Yan H, Hazawa M, Doan N, Said JW, Ding LW, Liu LZ, Yang H, Yu S, Kahn M, Yin D, Koeffler HP (2015) Genomic and functional analysis of the E3 ligase PARK2 in glioma. Cancer Res 75(9):1815–1827. https://doi.org/10.1158/0008-5472.CAN-14-1433
Liu L, Zhao X, Zhao L, Li J, Yang H, Zhu Z, Liu J, Huang G (2016) Arginine methylation of SREBP1a via PRMT5 promotes de novo lipogenesis and tumor growth. Cancer Res 76(5):1260–1272. https://doi.org/10.1158/0008-5472.CAN-15-1766
Lyu Z, Zhao Y, Buuh ZY, Gorman N, Goldman AR, Islam MS, Tang HY, Wang RE (2021) Steric-free bioorthogonal labeling of acetylation substrates based on a fluorine-thiol displacement reaction. J Am Chem Soc 143(3):1341–1347. https://doi.org/10.1021/jacs.0c05605
Ma J, Hart GW (2014) O-GlcNAc profiling: from proteins to proteomes. Clin Proteomics 11(1):8. https://doi.org/10.1186/1559-0275-11-8
Mendonsa AM, Bandyopadhyay C, Gumbiner BM (2020) p120-catenin phosphorylation status alters E-cadherin mediated cell adhesion and ability of tumor cells to metastasize. PLoS One 15(6):e0235337. https://doi.org/10.1371/journal.pone.0235337
Mitri Z, Constantine T, O’Regan R (2012) The HER2 receptor in breast cancer: pathophysiology, clinical use, and new advances in therapy. Chemother Res Pract 2012:743193. https://doi.org/10.1155/2012/743193
Mongiardi MP, Savino M, Bartoli L, Beji S, Nanni S, Scagnoli F, Falchetti ML, Favia A, Farsetti A, Levi A, Nasi S, Illi B (2015) Myc and Omomyc functionally associate with the Protein Arginine Methyltransferase 5 (PRMT5) in glioblastoma cells. Sci Rep 5(1):15494. https://doi.org/10.1038/srep15494
Narayan S, Bader GD, Reimand J (2016) Frequent mutations in acetylation and ubiquitination sites suggest novel driver mechanisms of cancer. Genome Med 8(1):55. https://doi.org/10.1186/s13073-016-0311-2
Narita T, Weinert BT, Choudhary C (2019) Functions and mechanisms of non-histone protein acetylation. Nat Rev Mol Cell Biol 20(3):156–174. https://doi.org/10.1038/s41580-018-0081-3
Neumann H, Hancock SM, Buning R, Routh A, Chapman L, Somers J, Owen-Hughes T, van Noort J, Rhodes D, Chin JW (2009) A method for genetically installing site-specific acetylation in recombinant histones defines the effects of H3 K56 acetylation. Mol Cell 36(1):153–163. https://doi.org/10.1016/j.molcel.2009.07.027
Nihira Naoe T, Ogura K, Shimizu K, North Brian J, Zhang J, Gao D, Inuzuka H, Wei W (2017) Acetylation-dependent regulation of MDM2 E3 ligase activity dictates its oncogenic function. Sci Signal 10(466):eaai8026. https://doi.org/10.1126/scisignal.aai8026
Olsen JV, Blagoev B, Gnad F, Macek B, Kumar C, Mortensen P, Mann M (2006) Global, in vivo, and site-specific phosphorylation dynamics in signaling networks. Cell 127(3):635–648. https://doi.org/10.1016/j.cell.2006.09.026
Ordureau A, Münch C, Harper JW (2015) Quantifying ubiquitin signaling. Mol Cell 58(4):660–676. https://doi.org/10.1016/j.molcel.2015.02.020
Ozaki T, Nakagawara A (2011) p53: the attractive tumor suppressor in the cancer research field. J Biomed Biotechnol 2011:603925. https://doi.org/10.1155/2011/603925
Peters AHFM, O’Carroll D, Scherthan H, Mechtler K, Sauer S, Schöfer C, Weipoltshammer K, Pagani M, Lachner M, Kohlmaier A, Opravil S, Doyle M, Sibilia M, Jenuwein T (2001) Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107(3):323–337. https://doi.org/10.1016/S0092-8674(01)00542-6
Pinho SS, Reis CA (2015) Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer 15(9):540–555. https://doi.org/10.1038/nrc3982
Powers MV, Clarke PA, Workman P (2009) Death by chaperone: HSP90, HSP70 or both? Cell Cycle 8(4):518–526. https://doi.org/10.4161/cc.8.4.7583
Prescher JA, Dube DH, Bertozzi CR (2004) Chemical remodelling of cell surfaces in living animals. Nature 430(7002):873–877. https://doi.org/10.1038/nature02791
Ruffner H, Joazeiro CAP, Hemmati D, Hunter T, Verma IM (2001) Cancer-predisposing mutations within the RING domain of BRCA1: loss of ubiquitin protein ligase activity and protection from radiation hypersensitivity. Proc Natl Acad Sci 98(9):5134. https://doi.org/10.1073/pnas.081068398
Sannino S, Brodsky JL (2017) Targeting protein quality control pathways in breast cancer. BMC Biol 15(1):109. https://doi.org/10.1186/s12915-017-0449-4
Sato Y, Mukai M, Ueda J, Muraki M, Stasevich TJ, Horikoshi N, Kujirai T, Kita H, Kimura T, Hira S, Okada Y, Hayashi-Takanaka Y, Obuse C, Kurumizaka H, Kawahara A, Yamagata K, Nozaki N, Kimura H (2013) Genetically encoded system to track histone modification in vivo. Sci Rep 3(1):2436. https://doi.org/10.1038/srep02436
Saxon E, Bertozzi CR (2000) Cell surface engineering by a modified Staudinger reaction. Science 287(5460):2007–2010. https://doi.org/10.1126/science.287.5460.2007
Schjoldager KT, Narimatsu Y, Joshi HJ, Clausen H (2020) Global view of human protein glycosylation pathways and functions. Nat Rev Mol Cell Biol 21(12):729–749. https://doi.org/10.1038/s41580-020-00294-x
Senft D, Qi J, Ronai ZA (2018) Ubiquitin ligases in oncogenic transformation and cancer therapy. Nat Rev Cancer 18(2):69–88. https://doi.org/10.1038/nrc.2017.105
Sharma K, D’Souza Rochelle CJ, Tyanova S, Schaab C, Wiśniewski Jacek R, Cox J, Mann M (2014) Ultradeep human phosphoproteome reveals a distinct regulatory nature of Tyr and Ser/Thr-based signaling. Cell Rep 8(5):1583–1594. https://doi.org/10.1016/j.celrep.2014.07.036
Sharma BS, Prabhakaran V, Desai AP, Bajpai J, Verma RJ, Swain PK (2019) Post-translational modifications (PTMs), from a cancer perspective: an overview. Oncogen 2(3):12. https://doi.org/10.35702/onc.10012
Shi Y, Lan F, Matson C, Mulligan P, Whetstine JR, Cole PA, Casero RA, Shi Y (2004) Histone demethylation mediated by the nuclear amine oxidase homolog LSD1. Cell 119(7):941–953. https://doi.org/10.1016/j.cell.2004.12.012
Shrestha D, Jenei A, Nagy P, Vereb G, Szöllősi J (2015) Understanding FRET as a research tool for cellular studies. Int J Mol Sci 16(4):6718–6756. https://doi.org/10.3390/ijms16046718
Singh V, Ram M, Kumar R, Prasad R, Roy BK, Singh KK (2017) Phosphorylation: implications in cancer. Protein J 36(1):1–6. https://doi.org/10.1007/s10930-017-9696-z
Slawson C, Hart GW (2011) O-GlcNAc signalling: implications for cancer cell biology. Nat Rev Cancer 11(9):678–684. https://doi.org/10.1038/nrc3114
Sletten EM, Bertozzi CR (2009) Bioorthogonal chemistry: fishing for selectivity in a sea of functionality. Angew Chem Int Ed 48(38):6974–6998. https://doi.org/10.1002/anie.200900942
Söderberg O, Gullberg M, Jarvius M, Ridderstråle K, Leuchowius K-J, Jarvius J, Wester K, Hydbring P, Bahram F, Larsson L-G, Landegren U (2006) Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat Methods 3(12):995–1000. https://doi.org/10.1038/nmeth947
Strahl BD, Briggs SD, Brame CJ, Caldwell JA, Koh SS, Ma H, Cook RG, Shabanowitz J, Hunt DF, Stallcup MR, Allis CD (2001) Methylation of histone H4 at arginine 3 occurs in vivo and is mediated by the nuclear receptor coactivator PRMT1. Curr Biol 11(12):996–1000. https://doi.org/10.1016/S0960-9822(01)00294-9
Sun Y, Jiang X, Xu Y, Ayrapetov MK, Moreau LA, Whetstine JR, Price BD (2009) Histone H3 methylation links DNA damage detection to activation of the tumour suppressor Tip60. Nat Cell Biol 11(11):1376–1382. https://doi.org/10.1038/ncb1982
Suraritdechachai S, Lakkanasirorat B, Uttamapinant C (2022) Molecular probes for cellular imaging of post-translational proteoforms. RSC Chem Biol 3:201. https://doi.org/10.1039/D1CB00190F
Sykes SM, Mellert HS, Holbert MA, Li K, Marmorstein R, Lane WS, McMahon SB (2006) Acetylation of the p53 DNA-binding domain regulates apoptosis induction. Mol Cell 24(6):841–851. https://doi.org/10.1016/j.molcel.2006.11.026
Tang Y, Zhao W, Chen Y, Zhao Y, Gu W (2008) Acetylation is indispensable for p53 activation. Cell 133(4):612–626. https://doi.org/10.1016/j.cell.2008.03.025
Teo CF, Ingale S, Wolfert MA, Elsayed GA, Nöt LG, Chatham JC, Wells L, Boons G-J (2010) Glycopeptide-specific monoclonal antibodies suggest new roles for O-GlcNAc. Nat Chem Biol 6(5):338–343. https://doi.org/10.1038/nchembio.338
Ubersax JA, Ferrell JE Jr (2007) Mechanisms of specificity in protein phosphorylation. Nat Rev Mol Cell Biol 8(7):530–541. https://doi.org/10.1038/nrm2203
Verveer PJ, Wouters FS, Reynolds AR, Bastiaens PI (2000) Quantitative imaging of lateral ErbB1 receptor signal propagation in the plasma membrane. Science 290(5496):1567–1570. https://doi.org/10.1126/science.290.5496.1567
Wagner SA, Beli P, Weinert BT, Nielsen ML, Cox J, Mann M, Choudhary C (2011) A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. Mol Cell Proteomics 10(10):M111.013284–M013111.013284. https://doi.org/10.1074/mcp.m111.013284
Wang L, Schultz PG (2002) Expanding the genetic code. Chem Commun 1:1–11. https://doi.org/10.1039/B108185N
Wang WW, Zeng Y, Wu B, Deiters A, Liu WR (2016) A chemical biology approach to reveal Sirt6-targeted histone H3 sites in nucleosomes. ACS Chem Biol 11(7):1973–1981. https://doi.org/10.1021/acschembio.6b00243
Wu Q, Schapira M, Arrowsmith CH, Barsyte-Lovejoy D (2021) Protein arginine methylation: from enigmatic functions to therapeutic targeting. Nat Rev Drug Discov 20(7):509–530. https://doi.org/10.1038/s41573-021-00159-8
Xu H, Wang Y, Lin S, Deng W, Peng D, Cui Q, Xue Y (2018) PTMD: a database of human disease-associated post-translational modifications. Genomics Proteomics Bioinformatics 16(4):244–251. https://doi.org/10.1016/j.gpb.2018.06.004
Yang Y-Y, Ascano JM, Hang HC (2010) Bioorthogonal chemical reporters for monitoring protein acetylation. J Am Chem Soc 132(11):3640–3641. https://doi.org/10.1021/ja908871t
Yates JR, Ruse CI, Nakorchevsky A (2009) Proteomics by mass spectrometry: approaches, advances, and applications. Annu Rev Biomed Eng 11(1):49–79. https://doi.org/10.1146/annurev-bioeng-061008-124934
Yun M, Wu J, Workman JL, Li B (2011) Readers of histone modifications. Cell Res 21(4):564–578. https://doi.org/10.1038/cr.2011.42
Zhang X, Li R, Chen Y, Zhang S, Wang W, Li F (2016) Applying DNA rolling circle amplification in fluorescence imaging of cell surface glycans labeled by a metabolic method. Chem Sci 7(9):6182–6189. https://doi.org/10.1039/C6SC02089E
Zhao S, Xu W, Jiang W, Yu W, Lin Y, Zhang T, Yao J, Zhou L, Zeng Y, Li H, Li Y, Shi J, An W, Hancock Susan M, He F, Qin L, Chin J, Yang P, Chen X, Lei Q, **ong Y, Guan K-L (2010) Regulation of cellular metabolism by protein lysine acetylation. Science 327(5968):1000–1004. https://doi.org/10.1126/science.1179689
Acknowledgments
This work was supported by Temple University (start-up funds) and the National Institutes of Health Grants (5R35GM133468). Partial support from the National Science Foundation (CHE-2144075) is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Cohen, C., Lewis, T.R., Wang, R.E. (2022). The Role of Post-Translational Modifications in Cancer. In: Rezaei, N. (eds) Handbook of Cancer and Immunology. Springer, Cham. https://doi.org/10.1007/978-3-030-80962-1_288-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80962-1_288-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80962-1
Online ISBN: 978-3-030-80962-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences