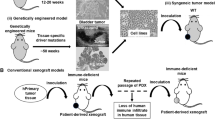
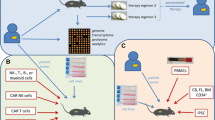
Abstract
Despite the developments in anti-cancer therapeutic approaches during the previous decades, cancer is still distinguished as one of the major health problems and a leading cause of death worldwide. Recent innovations in the field of cancer immunotherapy, such as immune checkpoint inhibition and chimeric antigen receptor T-cell therapy, have induced tumor regression in heavily pretreated patients with high-risk tumor subtypes and unresponsive to previous conventional therapies. However, there is so much room to improve the response rates and safety profiles of these immunotherapeutic agents. Preclinical models are a crucial part of cancer research and drug discovery mechanisms. Their capacity to provide a realistic model of human cancer genetic and expression profile, tumor microenvironment, and tumor cells’ response to immunotherapies has tremendously added to our understanding of the tumorigenesis mechanisms and immunotherapeutic agent mechanism of action. Moreover, so many unknown tumor-specific immunological antigens and components can be identified and targeted in mono- or combination therapies to enhance the response rates. The prospective advances in the field of immunotherapy would be highly dependent on our in-depth comprehension of the dynamics and interactions between the tumor cells and their hosts’ immunological components. Many different cancer models have been employed in cancer experiments and brought about their unique advantages along with some limitations. The research on preclinical models has always sought to model the inter- and intratumoral heterogeneity and the interactions in the tumor microenvironment and to enhance the producibility and predictability of the results. In this chapter, we comprehensively review the administered preclinical models in the cancer research experiment, with an emphasis on immunotherapy experiments, from cancer cell lines to non-human primate animal models.
Similar content being viewed by others
References
Agarwal S, Hynes PG, Tillman HS, Lake R, Abou-Kheir WG, Fang L, Casey OM, Ameri AH, Martin PL, Yin JJ, Iaquinta PJ, Karthaus WR, Clevers HC, Sawyers CL, Kelly K (2015) Identification of different classes of luminal progenitor cells within prostate tumors. Cell Rep 13(10):2147–2158. https://doi.org/10.1016/j.celrep.2015.10.077
Alison MR (2020) The cellular origins of cancer with particular reference to the gastrointestinal tract. Int J Exp Pathol 101(5):132–151. https://doi.org/10.1111/iep.12364
Aref AR, Campisi M, Ivanova E, Portell A, Larios D, Piel BP, Mathur N, Zhou C, Coakley RV, Bartels A, Bowden M, Herbert Z, Hill S, Gilhooley S, Carter J, Cañadas I, Thai TC, Kitajima S, Chiono V et al (2018) 3D microfluidic: ex vivo culture of organotypic tumor spheroids to model immune checkpoint blockade. Lab Chip 18(20):3129–3143. https://doi.org/10.1039/C8LC00322J
Aung A, Kumar V, Theprungsirikul J, Davey SK, Varghese S (2020) An engineered tumor-on-a-chip device with breast cancer–immune cell interactions for assessing T-cell recruitment. Cancer Res 80(2):263–275. https://doi.org/10.1158/0008-5472.CAN-19-0342
Balakrishnan A, Goodpaster T, Randolph-Habecker J, Hoffstrom BG, Jalikis FG, Koch LK, Berger C, Kosasih PL, Rajan A, Sommermeyer D, Porter PL, Riddell SR (2017) Analysis of ROR1 protein expression in human cancer and normal tissues. Clin Cancer Res 23(12):3061–3071. https://doi.org/10.1158/1078-0432.CCR-16-2083
Barretina J, Caponigro G, Stransky N, Venkatesan K, Margolin AA, Kim S, Wilson CJ, Lehár J, Kryukov GV, Sonkin D, Reddy A, Liu M, Murray L, Berger MF, Monahan JE, Morais P, Meltzer J, Korejwa A, Jané-Valbuena J et al (2012) The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 483(7391):603–607. https://doi.org/10.1038/NATURE11003
Bower R, Green VL, Kuvshinova E, Kuvshinov D, Karsai L, Crank ST, Stafford ND, Greenman J (2017) Maintenance of head and neck tumor on-chip: gateway to personalized treatment? Future Sci OA 3(2):FSO174–FSO174. https://doi.org/10.4155/fsoa-2016-0089
Brehm MA, Kenney LL, Wiles MV, Low BE, Tisch RM, Burzenski L, Mueller C, Greiner DL, Shultz LD (2019) Lack of acute xenogeneic graft- versus-host disease, but retention of T-cell function following engraftment of human peripheral blood mononuclear cells in NSG mice deficient in MHC class I and II expression. FASEB J 33(3):3137–3151. https://doi.org/10.1096/fj.201800636R
Calbo J, van Montfort E, Proost N, van Drunen E, Beverloo HB, Meuwissen R, Berns A (2011) A functional role for tumor cell heterogeneity in a mouse model of small cell lung cancer. Cancer Cell 19(2):244–256. https://doi.org/10.1016/j.ccr.2010.12.021
Carrel A, Burrows MT (1911) Cultivation in vitro of malignant tumors. J Exp Med 13(5):571–575. https://doi.org/10.1084/jem.13.5.571
Carrel A, Lindbergh CA (1935) The culture of whole organs. Science (New York, N.Y.) 81(2112):621–623. https://doi.org/10.1126/science.81.2112.621
Chaicharoenaudomrung N, Kunhorm P, Noisa P (2019) Three-dimensional cell culture systems as an in vitro platform for cancer and stem cell modeling. World J Stem Cells 11(12):1065–1083. https://doi.org/10.4252/wjsc.v11.i12.1065
Clevers H (2016) Modeling development and disease with organoids. Cell 165(7):1586–1597. https://doi.org/10.1016/j.cell.2016.05.082
Cunningham TJ, Tabacchi M, Eliane J-P, Tuchayi SM, Manivasagam S, Mirzaalian H, Turkoz A, Kopan R, Schaffer A, Saavedra AP, Wallendorf M, Cornelius LA, Demehri S (2017) Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy. J Clin Invest 127(1):106–116. https://doi.org/10.1172/JCI89820
Deng J, Wang ES, Jenkins RW, Li S, Dries R, Yates K, Chhabra S, Huang W, Liu H, Aref AR, Ivanova E, Paweletz CP, Bowden M, Zhou CW, Herter-Sprie GS, Sorrentino JA, Bisi JE, Lizotte PH, Merlino AA et al (2018) CDK4/6 inhibition augments antitumor immunity by enhancing T-cell activation. Cancer Discov 8(2):216–233. https://doi.org/10.1158/2159-8290.CD-17-0915
Dewi FN, Cline JM (2021) Nonhuman primate model in mammary gland biology and neoplasia research. Lab Anim Res 37(1):3. https://doi.org/10.1186/s42826-020-00053-1
Drexler HG, Matsuo AY, MacLeod RA (2000) Continuous hematopoietic cell lines as model systems for leukemia-lymphoma research. Leuk Res 24(11):881–911. https://doi.org/10.1016/s0145-2126(00)00070-9
Ericsson AC, Crim MJ, Franklin CL (2013) A brief history of animal modeling. Mo Med 110(3):201–205. https://pubmed.ncbi.nlm.nih.gov/23829102
Gambacorti-Passerini C, le Coutre P, Mologni L, Fanelli M, Bertazzoli C, Marchesi E, Di Nicola M, Biondi A, Corneo GM, Belotti D, Pogliani E, Lydon NB (1997) Inhibition of the ABL kinase activity blocks the proliferation of BCR/ABL+ leukemic cells and induces apoptosis. Blood Cells Mol Dis 23(3):380–394. https://doi.org/10.1006/bcmd.1997.0155
Han MG, Jang B-S, Kang MH, Na D, Kim IA (2021) PI3Kγδ inhibitor plus radiation enhances the antitumour immune effect of PD-1 blockade in syngenic murine breast cancer and humanised patient-derived xenograft model. Eur J Cancer (Oxford, England: 1990) 157:450–463. https://doi.org/10.1016/j.ejca.2021.08.029
Harrison RG (1908) Embryonic transplantation and development of the nervous system. Anat Rec 2(9):385–410. https://doi.org/10.1002/ar.1090020902
Hason M, Bartůněk P (2019) Zebrafish models of cancer-new insights on modeling human cancer in a non-mammalian vertebrate. Genes 10(11):935. https://doi.org/10.3390/genes10110935
He X, Yin X, Wu J, Wickström SL, Duo Y, Du Q, Qin S, Yao S, **g X, Hosaka K, Wu J, Jensen LD, Lundqvist A, Salter AI, Bräutigam L, Tao W, Chen Y, Kiessling R, Cao Y (2020) Visualization of human T lymphocyte-mediated eradication of cancer cells in vivo. Proc Natl Acad Sci U S A 117(37):22910–22919. https://doi.org/10.1073/pnas.2009092117
Hijaz M, Das S, Mert I, Gupta A, Al-Wahab Z, Tebbe C, Dar S, Chhina J, Giri S, Munkarah A, Seal S, Rattan R (2016) Folic acid tagged nanoceria as a novel therapeutic agent in ovarian cancer. BMC Cancer 16:220. https://doi.org/10.1186/s12885-016-2206-4
Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE (2010) Reconstituting organ-level lung functions on a chip. Science (New York, N.Y.) 328(5986):1662–1668. https://doi.org/10.1126/science.1188302
Hwang HH, You S, Ma X, Kwe L, Victorine G, Lawrence N, Wan X, Shen H, Zhu W, Chen S (2020) High throughput direct 3D bioprinting in multiwell plates. Biofabrication 13:025007. https://doi.org/10.1088/1758-5090/ab89ca
Jenkins RW, Aref AR, Lizotte PH, Ivanova E, Stinson S, Zhou CW, Bowden M, Deng J, Liu H, Miao D, He MX, Walker W, Zhang G, Tian T, Cheng C, Wei Z, Palakurthi S, Bittinger M, Vitzthum H et al (2018) Ex vivo profiling of PD-1 blockade using Organotypic tumor spheroids. Cancer Discov 8(2):196–215. https://doi.org/10.1158/2159-8290.CD-17-0833
Jeon JS, Bersini S, Gilardi M, Dubini G, Charest JL, Moretti M, Kamm RD (2015) Human 3D vascularized organotypic microfluidic assays to study breast cancer cell extravasation. Proc Natl Acad Sci U S A 112(1):214–219. https://doi.org/10.1073/pnas.1417115112
Lanotte M, Martin-Thouvenin V, Najman S, Balerini P, Valensi F, Berger R (1991) NB4, a maturation inducible cell line with t(15;17) marker isolated from a human acute promyelocytic leukemia (M3). Blood 77(5):1080–1086
Lee S-H, Cho S-Y, Yoon Y, Park C, Sohn J, Jeong J-J, Jeon B-N, Jang M, An C, Lee S, Kim YY, Kim G, Kim S, Kim Y, Lee GB, Lee EJ, Kim SG, Kim HS, Kim Y et al (2021) Bifidobacterium bifidum strains synergize with immune checkpoint inhibitors to reduce tumour burden in mice. Nat Microbiol 6(3):277–288. https://doi.org/10.1038/s41564-020-00831-6
Ling K, Huang G, Liu J, Zhang X, Ma Y, Lu T, Xu F (2015) Bioprinting-based high-throughput fabrication of three-dimensional MCF-7 human breast cancer cellular spheroids. Engineering 1(2):269–274. https://doi.org/10.15302/J-ENG-2015062
Liu W, Pan H-F, Wang Q, Zhao Z-M (2018) The application of transgenic and gene knockout mice in the study of gastric precancerous lesions. Pathol Res Pract 214(12):1929–1939. https://doi.org/10.1016/j.prp.2018.10.022
Liu X, Bao X, Hu M, Chang H, Jiao M, Cheng J, **e L, Huang Q, Li F, Li C-Y (2020) Inhibition of PCSK9 potentiates immune checkpoint therapy for cancer. Nature 588(7839):693–698. https://doi.org/10.1038/s41586-020-2911-7
Mathur D, Root AR, Bugaj-Gaweda B, Bisulco S, Tan X, Fang W, Kearney JC, Lucas J, Guffroy M, Golas J, Rohde CM, Stevens C, Kamperschroer C, Kelleher K, Lawrence-Henderson RF, Upeslacis E, Yao J, Narula J, LaVallie ER et al (2020) A novel GUCY2C-CD3 T-cell engaging bispecific construct (PF-07062119) for the treatment of gastrointestinal cancers. Clin Cancer Res 26(9):2188–2202. https://doi.org/10.1158/1078-0432.CCR-19-3275
Neal JT, Li X, Zhu J, Giangarra V, Grzeskowiak CL, Ju J, Liu IH, Chiou S-H, Salahudeen AA, Smith AR, Deutsch BC, Liao L, Zemek AJ, Zhao F, Karlsson K, Schultz LM, Metzner TJ, Nadauld LD, Tseng Y-Y et al (2018) Organoid modeling of the tumor immune microenvironment. Cell 175(7):1972–1988.e16. https://doi.org/10.1016/j.cell.2018.11.021
Nguyen M, De Ninno A, Mencattini A, Mermet-Meillon F, Fornabaio G, Evans SS, Cossutta M, Khira Y, Han W, Sirven P, Pelon F, Di Giuseppe D, Bertani FR, Gerardino A, Yamada A, Descroix S, Soumelis V, Mechta-Grigoriou F, Zalcman G et al (2018) Dissecting effects of anti-cancer drugs and cancer-associated fibroblasts by on-Chip reconstitution of immunocompetent tumor microenvironments. Cell Rep 25(13):3884–3893.e3. https://doi.org/10.1016/j.celrep.2018.12.015
Nusinow DP, Szpyt J, Ghandi M, Rose CM, McDonald ER 3rd, Kalocsay M, Jané-Valbuena J, Gelfand E, Schweppe DK, Jedrychowski M, Golji J, Porter DA, Rejtar T, Wang YK, Kryukov GV, Stegmeier F, Erickson BK, Garraway LA, Sellers WR, Gygi SP (2020) Quantitative proteomics of the cancer cell line encyclopedia. Cell 180(2):387–402.e16. https://doi.org/10.1016/j.cell.2019.12.023
Olson B, Li Y, Lin Y, Liu ET, Patnaik A (2018) Mouse models for cancer immunotherapy research. Cancer Discov 8(11):1358–1365. https://doi.org/10.1158/2159-8290.CD-18-0044
Polacheck WJ, Charest JL, Kamm RD (2011) Interstitial flow influences direction of tumor cell migration through competing mechanisms. Proc Natl Acad Sci U S A 108(27):11115–11120. https://doi.org/10.1073/pnas.1103581108
Regev A, Teichmann SA, Lander ES, Amit I, Benoist C, Birney E, Bodenmiller B, Campbell P, Carninci P, Clatworthy M, Clevers H, Deplancke B, Dunham I, Eberwine J, Eils R, Enard W, Farmer A, Fugger L, Göttgens B et al (2017) The human cell atlas. elife 6. https://doi.org/10.7554/ELIFE.27041
Scherer WF, Syverton JT, Gey GO (1953) Studies on the propagation in vitro of poliomyelitis viruses. IV. Viral multiplication in a stable strain of human malignant epithelial cells (strain HeLa) derived from an epidermoid carcinoma of the cervix. J Exp Med 97(5):695–710. https://doi.org/10.1084/jem.97.5.695
Shi C, Chen X, Tan D (2019) Development of patient-derived xenograft models of prostate cancer for maintaining tumor heterogeneity. Transl Androl Urol 8(5):519–528. https://doi.org/10.21037/tau.2019.08.31
Solit DB, Garraway LA, Pratilas CA, Sawai A, Getz G, Basso A, Ye Q, Lobo JM, She Y, Osman I, Golub TR, Sebolt-Leopold J, Sellers WR, Rosen N (2006) BRAF mutation predicts sensitivity to MEK inhibition. Nature 439(7074):358. https://doi.org/10.1038/NATURE04304
Sun W, Luo Z, Lee J, Kim HJ, Lee KJ, Tebon P, Feng Y, Dokmeci MR, Sengupta S, Khademhosseini A (2019) Organ-on-a-Chip for cancer and immune Organs modeling. Adv Healthc Mater 8(4):e1801363. https://doi.org/10.1002/ADHM.201801363
Tannenbaum J, Bennett BT (2015) Russell and Burch’s 3Rs then and now: the need for clarity in definition and purpose. J Am Assoc Lab Anim Sci 54(2):120–132. https://pubmed.ncbi.nlm.nih.gov/25836957
Taraseviciute A, Tkachev V, Ponce R, Turtle CJ, Snyder JM, Liggitt HD, Myerson D, Gonzalez-Cuyar L, Baldessari A, English C, Yu A, Zheng H, Furlan SN, Hunt DJ, Hoglund V, Finney O, Brakke H, Blazar BR, Berger C et al (2018) Chimeric antigen receptor T cell-mediated neurotoxicity in nonhuman primates. Cancer Discov 8(6):750–763. https://doi.org/10.1158/2159-8290.CD-17-1368
Taylor MW (2014) A history of cell culture BT - viruses and man: a history of interactions (MW Taylor (ed), pp. 41–52). Springer International Publishing. https://doi.org/10.1007/978-3-319-07758-1_3
Townsend EC, Murakami MA, Christodoulou A, Christie AL, Köster J, DeSouza TA, Morgan EA, Kallgren SP, Liu H, Wu S-C, Plana O, Montero J, Stevenson KE, Rao P, Vadhi R, Andreeff M, Armand P, Ballen KK, Barzaghi-Rinaudo P et al (2016) The public repository of xenografts enables discovery and randomized phase II-like trials in mice. Cancer Cell 29(4):574–586. https://doi.org/10.1016/j.ccell.2016.03.008
Verma B, Wesa A (2020) Establishment of humanized mice from peripheral blood mononuclear cells or cord blood CD34+ hematopoietic stem cells for immune-oncology studies evaluating new therapeutic agents. Curr Protoc Pharmacol 89(1):e77. https://doi.org/10.1002/cpph.77
Vickerman V, Kamm RD (2012) Mechanism of a flow-gated angiogenesis switch: early signaling events at cell-matrix and cell-cell junctions. Integr Biol 4(8):863–874. https://doi.org/10.1039/c2ib00184e
Virumbrales-Muñoz M, Chen J, Ayuso J, Lee M, Abel EJ, Beebe DJ (2020) Organotypic primary blood vessel models of clear cell renal cell carcinoma for single-patient clinical trials. Lab Chip 20(23):4420–4432. https://doi.org/10.1039/d0lc00252f
Wadosky KM, Wang Y, Zhang X, Goodrich DW (2019) Generation of tumor organoids from genetically engineered mouse models of prostate cancer. J Vis Exp 148. https://doi.org/10.3791/59710
Wang M, Yao L-C, Cheng M, Cai D, Martinek J, Pan C-X, Shi W, Ma A-H, De Vere White RW, Airhart S, Liu ET, Banchereau J, Brehm MA, Greiner DL, Shultz LD, Palucka K, Keck JG (2018a) Humanized mice in studying efficacy and mechanisms of PD-1-targeted cancer immunotherapy. FASEB J 32(3):1537–1549. https://doi.org/10.1096/fj.201700740R
Wang Y, Shi W, Kuss M, Mirza S, Qi D, Krasnoslobodtsev A, Zeng J, Band H, Band V, Duan B (2018b) 3D bioprinting of breast cancer models for drug resistance study. ACS Biomater Sci Eng 4(12):4401–4411. https://doi.org/10.1021/acsbiomaterials.8b01277
Wang L, Chen H, Fei F, He X, Sun S, Lv K, Yu B, Long J, Wang X (2019) Patient-derived heterogeneous xenograft model of pancreatic cancer using zebrafish larvae as hosts for comparative drug assessment. J Vis Exp 146. https://doi.org/10.3791/59507
Wimalachandra DC, Li Y, Liu J, Shikha S, Zhang J, Lim Y-C, Zhang Y (2019) Microfluidic-based immunomodulation of immune cells using Upconversion nanoparticles in simulated blood vessel-tumor system. ACS Appl Mater Interfaces 11(41):37513–37523. https://doi.org/10.1021/acsami.9b15178
Zhao X, Xu Z, **ao L, Shi T, **ao H, Wang Y, Li Y, Xue F, Zeng W (2021) Review on the vascularization of organoids and organoids-on-a-Chip. Front Bioeng Biotechnol 9:637048. https://doi.org/10.3389/fbioe.2021.637048
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this entry
Cite this entry
Pezeshki, P.S., Rezaei, N. (2023). Preclinical Cancer Models for the Evaluation of Immunotherapies: From Cell Lines to Animal Models. In: Rezaei, N. (eds) Handbook of Cancer and Immunology. Springer, Cham. https://doi.org/10.1007/978-3-030-80962-1_206-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80962-1_206-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80962-1
Online ISBN: 978-3-030-80962-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences